Abstract
On the basis of EO composition and the principal compounds characterizing the volatile fraction of rosemary, 3 principal chemotypes can be identified 18 : chemotype I, with a high content of 1,8-cineole; chemotype II, with a content of camphor >20%; chemotype III, with a content of verbenone >15%. Conventionally the chemotypes reported above are also called cineoliferum, camphoriferum, and verbenoniferum, respectively. Jordan et al 16 reported high variability in rosemary EO composition in wild populations based on several environmental factors. The qualitative and quantitative composition and yield of the EO are influenced by the place of origin, 19 environmental conditions, 20 plant development stage, 21 and harvest time. 22 More recently, studies have reported that even the soil type and its composition could influence the EO profile of aromatic plant in the Mediterranean area. 23,24 Chemical compositional and temporal variations in rosemary EO have been reported in southern Spain. 25 In this case, the chemical variability of the EO profile was deeply influenced by geographical area. 26
To the best of our knowledge, no previous studies have explored the monthly variation in EO composition of
The seasonal EO yield, water content, and DW from rosemary leaves and branch and the plant origin are reported in Tables 1 and 2. The EO yield of all 5
Seasonal Essential Oil (EO) Yield, Water Content (%), and Dry Weight (g) of

n.s., not significant.
Probability of significant ANOVA results was also reported.
aThe analysis of variance (ANOVA) table shows the results of a 2-way ANOVA performed using season (A) and accession (B) as factors. Means separation was performed by Fisher’s least significant difference (LSD) procedure (
Climatic Condition of the Sampling Site.

Spring, summer, autumn, and winter average temperatures (average, Av. Temp; maximum, Av. Max. Temp; minimum, Av. Min. Temp), maximum and minimum temperatures (Max. Temp, Min. Temp, respectively), and rain precipitation mm were obtained from the 1991 to 2016 data series. In table are reported the average value ± the standard deviation.
The rosemary EO composition has been reported by several research studies.
16,29-31
A total of 27 compounds, belonging to 6 chemical groups (hydrocarbon monoterpene, alcohols, aldehydes, ketones, esters, hydrocarbon sesquiterpene), were identified as reported in Table 4. Seven compounds represent the predominant chemical fraction of rosemary EO: α-pinene, camphene, 1,8-cineole, borneol, camphor, verbenone, bornyl acetate (Table 4). The identified principal compounds are in agreement with the Sardinian rosemary “α-pinene\borneol\bornyl acetate\verbenone” chemotype previously described by Pintore et al
1
and later by Angioni et al.
31
Accession and season significantly affect the EO composition of
The Main Constituents of Essential Oil of Rosemary as Affected by Growing Location and Harvest Season.

RI, retention index on HP5 column.
Average values of 12 months and standard deviations are reported for each accession. In addition, the average seasonal values and standard deviations are also reported. In bold are reported the most produced compounds.
As shown in Table 4, 15 compounds are produced differently based on the genotype. β-Pinene, γ-terpinene, α-terpinolene, and bornyl acetate significantly varied during the 4 seasons. β-Pinene and bornyl acetate showed the maximum production during spring, while the lowest was found in autumn (3.1 and 0.8; 6.8 and 4.8, respectively); γ-terpinene and terpinolene showed the highest production during summer (0.8 and 0.7, respectively), while the minimum values (0) were found in winter and autumn, respectively.
In addition, relative percentages of α-terpinolene and bornyl acetate are influenced by both accession and seasonal conditions. According to these data, it seems that the accession and environmental conditions affect the EO composition of both the principal and minor compounds.
Data on the volatile fraction obtained by internal normalization of the FID chromatograms were subjected to multivariate analysis. The raw matrix composed of the volatile organic compounds was detected and all the samples were subjected to principal component analysis (PCA). Analyzing the loading plot is possible to evaluate which variables mainly contain the information able to discriminate the samples (the farther from zero) and, in contrast, which variables are mainly involved in increasing the noise of the model (the closer to zero). Myrcene, camphor, geraniol, α-fenchene,

Principal component analysis of volatile organic compounds from
In conclusion, the harvesting time of different
Experimental
Sampling Sites, Plant Material, and Essential Oil Extraction
Chemicals and Reagents
Unless stated otherwise, all chemicals and reagents were supplied by Sigma (Dorset, UK). For the following terpenoid compounds commercial reference standards were used: α-pinene, verbenene, β-pinene, myrcene, α-phellandrene, α-terpinene,
Gas Chromatography Mass Spectrometry Analysis
Gas chromatography (GC)-mass spectrometry (MS) analysis of the EO in hexane (dilution ratio 1:100) was carried out using an Agilent 7890 GC equipped with a Gerstel MPS autosampler, coupled to an Agilent 7000C MSD detector. Chromatographic separation was performed on a HP-5MS capillary column (30 m × 0.25 mm, film thickness 0.17 μm), using the following temperature program: 60°C held for 3 minutes, then increased to 210°C at a rate of 4°C/min, then held at 210°C for 15 minutes, before increasing to 300°C at a rate of 10°C/min, and finally held at 300°C for 15 minutes. Helium was used as the carrier gas at a constant flow of 1 mL/min.
A mixture of aliphatic hydrocarbons (C9-C23; Sigma-Aldrich, USA) in
For data analysis, a Mass Hunter Workstation B.06.00 SP1 was used. Identification of the individual components was performed by comparison with the co-injected pure compounds (see the section “Chemicals and Reagents”) and by matching the MS fragmentation patterns and retention indices with the built-in libraries or literature data or commercial mass spectral libraries (NIST/EPA/NIH 2008; HP1607 purchased from Agilent Technologies).
Gas Chromatography-Flame Ionization Detector Analysis
GC analysis of the EO in hexane (dilution ratio 1:100) was conducted using an Agilent 4890N instrument equipped with a flame ionization detector (FID) and a HP-5 capillary column (30 m × 0.25 mm, film thickness 0.17 µm). The column temperature program was the same as described above for the GC-MS analysis.
The constituent’s quantification in the
Meteorological Conditions
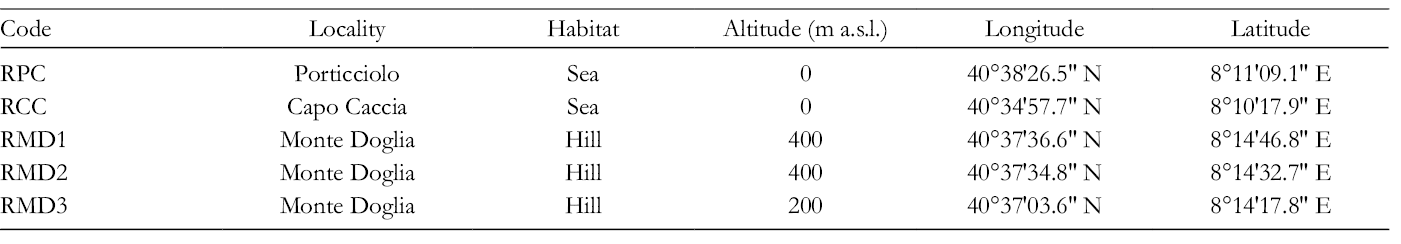
Altitude level, geographic localization, and climatic conditions were recorded (Tables 1 and 2). Meteorological data were provided by “Settore Idrografico della Regione Sardegna.” Seasonal meteorological parameters were obtained for the monthly data. Monthly precipitation and temperatures (average, maximum, and minimum) of historical series (1991-2016) were considered (Table 2).
Statistical Analysis and Principal Component Analysis
Correlations of EO production and composition and seasonal trends were calculated. All data were subjected to analysis of variance. All meteorological variables were standardized before the statistical analysis. Correlations were carried out by JMP 7 software (SAS Institute, Cary, NC, USA). GLM was run to evaluate the effect of the variables.
The multivariate analysis of samples was carried out subjecting the GC-FID data to PCA. The relative percentages of VOCs data of each EO were used to define a matrix
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ Note
Sara Melito and Giacomo Luigi Petretto contributed equally to this work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
