Abstract
Two previously undescribed triterpenoid saponins together with 4 known ones were isolated from the stem bark of Pentaclethra eetveldeana De Wild. & Th. Dur. Their structures were elucidated by spectroscopic methods including 1D and 2D NMR experiments in combination with mass spectrometry as 3-O-β-D-glucopyranosyl-(1→2)-[β-D-glucopyranosyl-(1→3)]-β-D-glucopyranosyl-(1→4)-β-D-glucopyranosyl-(1→3)-α-L-rhamnopyranosyl-(1→2)-[β-D-glucopyranosyl-(1→4)]-α-L-arabinopyranosyloleanolic acid and 3-O-β-D-glucopyranosyl-(1→2)-[β-D-glucopyranosyl-(1→3)]-β-D-glucopyranosyl-(1→4)-β-D-glucopyranosyl-(1→3)-α-L-rhamnopyranosyl-(1→2)-[β-D-glucopyranosyl-(1→4)]-α-L-arabinopyranosylhederagenin.
The genus Pentaclethra (Fabaceae) is represented by only 3 species (Pentaclethra eetveldeana, P. macrophylla, and P. macroloba). 1 P. macroloba is known to the local people in Brazil as antidote against snakebites 1 from which triterpenoid saponins have been isolated and characterized. 1,2 P. eetveldeana is a tree of 15 to 30 m of tropical African forest. It is used in Zairian traditional medicine for the treatment of hemorrhoids and various diseases such as malaria and epilepsy. 3 Previous phytochemical studies on P. eetveldeana showed the presence of saponins in the seeds 4 with oleanolic acid or hederagenin as aglycone and glucose, rhamnose, and arabinose as sugar units. In a continuation of our studies on natural saponins from the plants of the Fabaceae family, 5,6 we decided to examine the saponins from the stem bark of P. eetveldeana De Wild. & Th. Dur. In this paper, we report the isolation and structure elucidation of 2 previously undescribed triterpene saponins together with 4 known ones. Their structures were elucidated by spectroscopic methods including 600 MHz 1D and 2D experiments (1H, 13C, HSQC, HMBC, COSY, TOCSY, ROESY) in combination with HR-ESIMS and by comparison of their physical and spectral data with literature values.
The saponin fraction obtained from the 80% aqueous methanolic extract of the stem bark of P. eetveldeana was fractionated by vacuum liquid chromatography (VLC) on reverse-phase RP-18 silica gel and medium-pressure liquid chromatography (MPLC) yielding 2 previously undescribed saponins

Structure of compounds 1 and
Compounds
Compound
13C NMR and 1H NMR Spectroscopic Data of the Aglycone Moieties for Compounds 1 and 2, in CD3OD (δ in ppm) a .

nd: not determined.
Overlapped proton NMR signals are reported without designated multiplicity.
The analysis of the 1H NMR spectrum of the sugar part of
13C NMR and 1H NMR Spectroscopic Data of the Sugar Moieties for Compounds 1 and 2, in CD3OD (δ in ppm) a .

Overlapped proton NMR signals are reported without designated multiplicity.
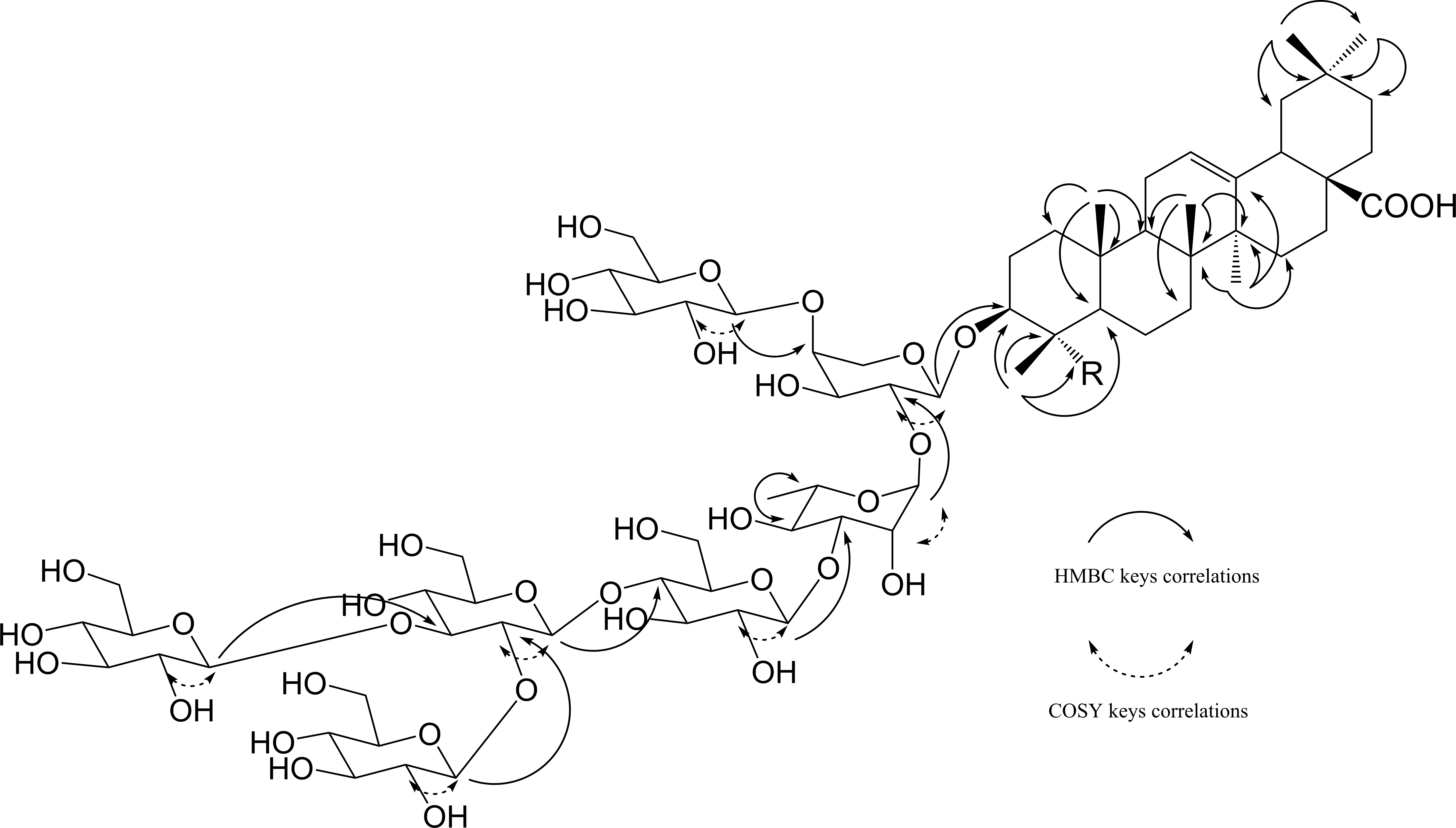
HMBC and COSY correlations for compounds 1 and 2.
Compound
From the crude ethanolic extract of the stem bark of P eetveldeana, we isolated 2 previously undescribed monodesmosidic triterpenoid saponins and 4 known ones by successive MPLC. As it was reported, structural analogs were isolated from P. macroloba.
1,2
Some of them shared the same 6 sugar sequence Glc-(1→2)-Glc-(1→4)-Glc1-(1→3)-Rha-(1→2)-[Glc-(1→4)]-Ara- linked at C-3 of oleanolic acid and hederagenin as in
Experimental
General Procedures
Optical rotation values were recorded on AA-10R automatic polarimeter. NMR spectra were performed using a Varian INOVA 600 at the operating frequency of 600 MHz. For details, see the section Experimental part of Ref. 11. 11 HR-ESIMS (positive-ion mode) and ESIMS (positive-ion mode) were carried out on a Bruker micrOTOF mass spectrometer. GC analysis was carried out on a thermoquest gas chromatograph using a DB-1701 cap. column (30 m × 0.25 mm, i.d.) (J and W Scientific); detection by FID; detector temperature 250°C, injection temperature 230°C; initial temperature was maintained at 80°C for 5 minutes and then raised to 270°C at 15°C/min; carrier gas He. TLC and HPTLC are performed using silica gel plates (Merck) (CHCl3-MeOH-H2O, 70/30/5 and 60/32/7). The spray reagent for saponins was vanillin reagent (2% mixture of conc. H2SO4 soln. and 1% vanillin in EtOH). Isolations were carried out using an MPLC system (Alltech pump, Büchi column [460 × 15 mm], Büchi precolumn [110 × 15 mm], silica gel 60 [Merck, 15-40 μm]).
Plant Material
The stem bark of P. eetveldeana De Wild. & Th. Dur. (Fabaceae) was collected on the campus of the University of Kinshasa (Democratic Republic of Congo) in a small forest. It was identified by comparison with the reference Herbal P. Compere 698 kept in the Herbarium of INERA/University of Kinshasa.
Extraction and Isolation
A total of 221 g of dried and pulverized stem bark was macerated in 350 mL of 80% aqueous MeOH during 1 week and then refluxed for 1h 30. The extract was then filtered and washed with 300 mL of 80% aq. MeOH and left above a water bath to evaporate at 50°C in a ventilated atmosphere. The hydro-alcoholic extract was dissolved in MeOH (150 mL) and saponin was precipitated in Et2O (450 mL), filtered, and dried. This procedure was repeated again, yielding 11 g of a whitish crude saponin mixture (CSM). An aliquot of CSM (1.5 g) was fractionated by VLC on normal phase silica gel (40-60 μm) using as eluent CHCl3-MeOH-H2O (60/32/7) to give 7 fractions Fr.1 to Fr.7 (150 mL ×7). Fr.2 (190 mg) was fractionated by MPLC over silica gel eluted with a CHCl3-MeOH-H2O gradient (70/30/5, 60/32/7), 2.5 mL/min, to give 12 fractions Fr.2.1 to Fr.2.12. Fr.2.11 (11 mg) was purified by VLC over RP-18 silica gel (75-200 μm) using as eluent H2O/EtOH (linear gradient 100/0-0/100) to give compound
Acid Hydrolysis and GC Analysis
Each compound (3 mg) was hydrolyzed with 2 N aq. CF3COOH (5 mL) for 3 hours at 95°C. After extraction with CH2Cl2 (3 × 5 mL), the aq. layer was repeatedly evaporated to dryness with MeOH until neutral and then analyzed by TLC over silica gel (CHCl3-MeOH-H2O 8/5/1) by comparison with authentic samples. Furthermore, the residue of sugars was dissolved in anhydrous pyridine (100 µL), and L-cysteine methyl ester hydrochloride (0.06 mol/L) was added. The mixture was stirred at 60°C for 1 hour, then 150 µL of HMDS-TMCS (hexamethyl-disilazane-trimethylchlorosilane 3:1) was added, and the mixture was stirred at 60°C for another 30 minutes. The precipitate was centrifuged off, and the supernatant was concentrated under N2 stream. The residue was partitioned between n-hexane and H2O (0.1 mL each), and the hexane layer (1 µL) was analyzed by GC.
9
The absolute configurations were determined by comparing the retention times with thiazolidine derivatives prepared in a similar way from standard sugars (Sigma-Aldrich): l-rhamnose, d-glucose, and l-arabinose for
3-O-β-D-Glucopyranosyl-(1→2)-[β-D-Glucopyranosyl-(1→3)]-β-D-Glucopyranosyl-(1→4)-β-D-Glucopyranosyl-(1→3)-α-L-Rhamnopyranosyl-(1→2)-[β-D-Glucopyranosyl-(1→4)]-α-L-Arabinopyranosyloleanolic Acid (1)
White, amorphous powder.
1H NMR and 13C NMR (CD3OD, 600 and 150 MHz): Tables 1 and 2.
HR-ESIMS (positive-ion mode) m/z 1567.7140 [M+Na]+ (calcd. 1567.7144 for C71H116O36Na); ESIMS (positive-ion mode) m/z 1567 [M+Na]+.
3-O-β-D-Glucopyranosyl-(1→2)-[β-D-Glucopyranosyl-(1→3)]-β-D-Glucopyranosyl-(1→4)-β-D-Glucopyranosyl-(1→3)-α-L-Rhamnopyranosyl-(1→2)-[β-D-Glucopyranosyl-(1→4)]-α-L-Arabinopyranosylhederagenin (2)
White, amorphous powder.
1H NMR and 13C NMR (CD3OD, 600 and 150 MHz): Tables 1 and 2.
HR-ESIMS (positive-ion mode) m/z 1583.7098 [M+Na]+ (calcd. 1583.7093 for C71H116O37Na); ESIMS (positive-ion mode) m/z 1583 [M+Na]+.
Supplemental Material
Supporting information - Supplemental material for Triterpenoid Saponins From the Stem Bark of Pentaclethra eetveldeana
Supplemental material, Supporting information for Triterpenoid Saponins From the Stem Bark of Pentaclethra eetveldeana by David Pertuit, Mpuza Kapundu, Anne-Claire Mitaine-Offer, Tomofumi Miyamoto, Chiaki Tanaka, Clément Delaude, and Marie-Aleth Lacaille-Dubois in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
