Abstract
Two undescribed triterpenoid saponins together with 5 known ones were isolated from the root bark of Haplocoelum congolanum Hauman. Their structures were elucidated by spectroscopic methods including one-dimensional and two-dimensional nuclear magnetic resonance experiments in combination with mass spectrometry as 3-O-(4-O-[3-hydroxy-3-methylglutaryl])-α-l-arabinopyranosyl-(1→3)-α-l-rhamnopyranosyl-(1→2)-[β-d-glucopyranosyl-(1→4)]-α-l-arabinopyranosyloleanolic acid and 3-O-α-l-arabinofuranosyl-(1→3)-α-l-rhamnopyranosyl-(1→2)-[β-d-glucopyranosyl-(1→4)]-α-l-arabinopyranosyloleanolic acid.
The Sapindaceae family includes more than 2 000 species distributed in 3 subfamilies as Sapindoideae, Dodonaeoideae, and Aceroideae. 1,2 These plants are known to contain triterpenoid saponins. 3 In a continuation of our studies on natural saponins from the plants of this family, 4,5 we decided to examine the saponins from the root bark of Haplocoelum congolanum Hauman, belonging to the Sapindoideae subfamily. No information was available about its therapeutic or medicinal properties. Previous chemical studies of H. congolanum led to the isolation and characterization of a triterpene saponin mixture having hederagenin or oleanolic acid as aglycone with glucose, arabinose, rhamnose, and xylose as sugars. 6 In the present paper, we report the isolation and structure elucidation of 2 undescribed triterpene saponins together with 5 known ones. Their structures were elucidated by spectroscopic methods including 600 MHz one-dimensional (1D) and two-dimensional (2D) experiments (1H, 13C, heteronuclear single-quantum coherence [HSQC], heteronuclear multiple bond correlation [HMBC], correlated spectroscopy [COSY], total correlated spectroscopy [TOCSY], rotating-frame Overhauser spectroscopy [ROESY]) in combination with high-resolution electroscopy ionization mass spectroscopy (HR-ESIMS) and by comparison of their physical and spectral data with literature values.
The saponin fraction obtained from the 80% aqueous ethanolic extract of the root bark of H. congolanum was fractionated by repeated medium-pressure liquid chromatography (MPLC) and semipreparative high-performance liquid chromatography (HPLC) on RP-18 silica gel yielding 2 undescribed compounds

Saponins from root bark of Haplocoelum congolanum.
Compounds
Compound
13C NMR and 1H NMR Spectroscopic Data of the Aglycone Moieties for Compounds 1 and 2, in CD3OD (δ in ppm) a .

nd, not determined;NMR, nuclear magnetic resonance.
aOverlapped proton NMR signals are reported without designated multiplicity.
The analysis of the 1H NMR spectrum of the sugar part of
13C NMR and 1H NMR Spectroscopic Data of the Sugar Moieties for Compounds 1 and 2, in CD3OD (δ in ppm) a .
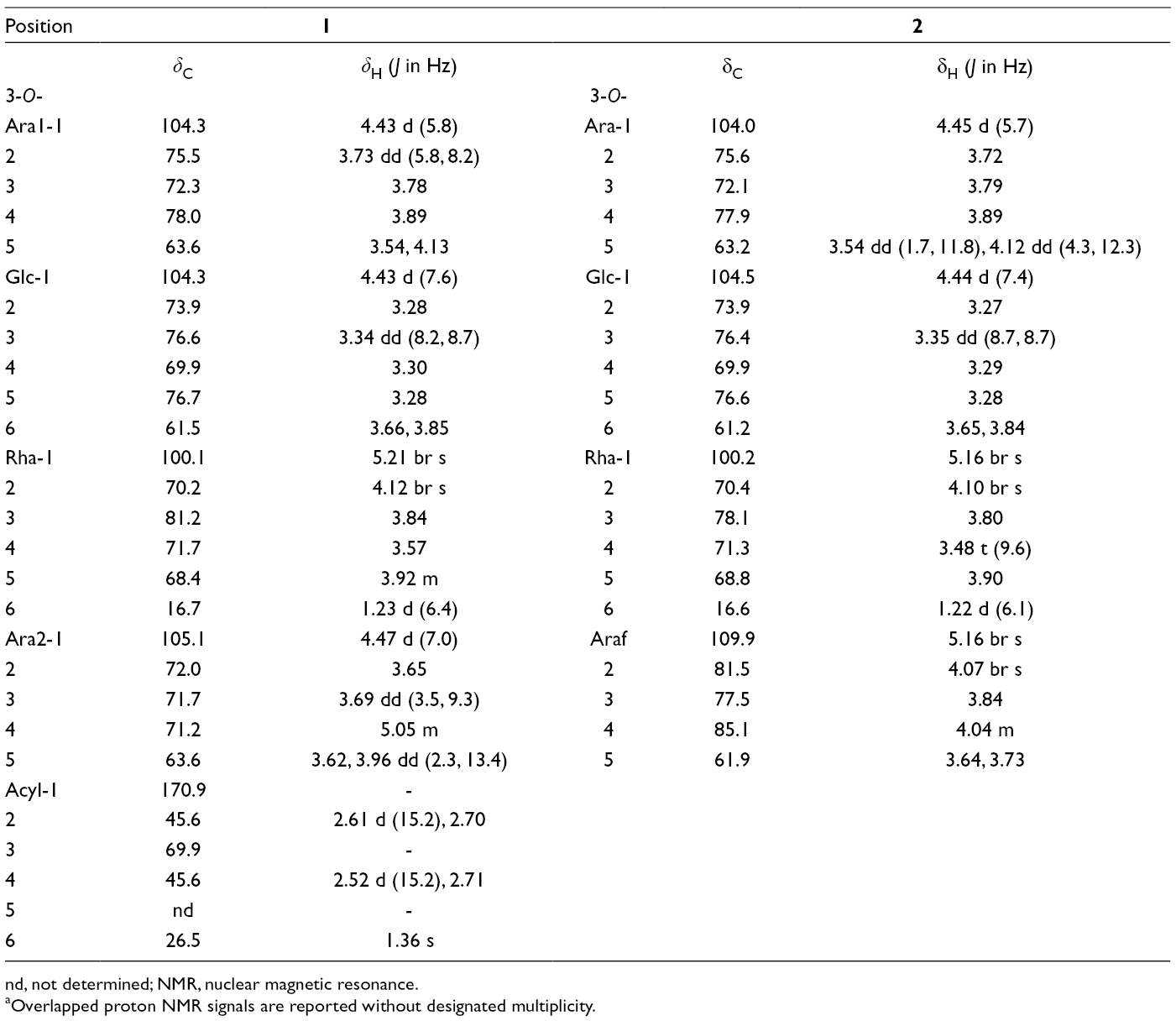
nd, not determined;NMR, nuclear magnetic resonance.
aOverlapped proton NMR signals are reported without designated multiplicity.
Furthermore, the TOCSY spectrum showed a correlation at δ
H / δ
H 4.47 (Ara2 H-1)/ 5.05, which suggested an acylation. We observed the presence in 1D- and 2D-NMR spectra (Table 2) of additional signals of a 3-hydroxy-3-methylglutaryl moiety (dicrotalic acid), which were in good agreement with literature data.
14,15
The location of dicrotalic acid at C-4 position of Ara2 was ascertained by observation of a deshielded 13C NMR chemical shift at δ
C71.2 (Ara2 C-4) instead of 67.5 to 68.7, or 69.2 for a free Ara 4-position as described in the literature.
5,16
Furthermore, all the NMR data of acylated Ara2 C-4 were in good agreement with literature values for an acylated Ara at C-4 position.
16
This result allowed to confirm that the dicrotalic acid was linked at the C-4 position of Ara2. Thus, the structure of
Compound
From the crude 80% ethanolic extract of the root bark of H. congolanum, we isolated 2 undescribed compounds and 5 known ones by successive MPLC and semipreparative HPLC. The sequences Rha-(1→2)-[Glc-(1→4)]-Ara- and Xyl-(1→3)-Rha-(1→2)-[Glc-(1→4)]-Ara- linked at C-3 of oleanolic acid or hederagenin was not considered as a chemotaxonomic marker in this family because they were also found in the Ranunculaceae family. All known triterpenoid saponins were also isolated from the genus Anemone, in the Ranunculaceae family. The group 3-hydroxy-3-methylglutaric acid was not unusual in the Sapindaceae family: it was encountered only once linked to a phenolic glycoside from Eurycorymbus cavaleriei,
17
but was found for the first time linked at a sugar of a triterpenoid saponin in this family. The sequence Araf-3Rha-2Ara-3oleanolic acid in compound
Experimental
General Procedures
Optical rotation values were recorded on AA-10R automatic polarimeter. NMR spectra: Spectra were performed using a Varian INOVA 600 at the operating frequency of 600 MHz. For details, see experimental part. 5 HR-ESIMS (positive-ion mode) and ESIMS (positive-ion mode) were carried out on a Bruker micrOTOF mass spectrometer. GC analysis was carried out on a thermoquest gas chromatograph using a DB-1701 cap. column (30 m × 0.25 mm, i.d) (J and W Scientific); detection by FID; detector temperature, 250°C, injection temperature, 230°C, initial temperature was maintained at 80°C for 5 minutes and then raised to 270°C at 15 °C/min; carrier gas, He. Thin-layer chromatography (TLC) and high-performance TLC employed precoated Si gel plates 60 F254 (Merck) (CHCl3–MeOH–H2O, 70/30/5 and 60/32/7). The spray reagent for saponins was vanillin reagent (2% mixture of concentrated H2SO4 solution and 1% vanillin in EtOH). Isolations were carried out using a MPLC system (Alltech pump, Büchi column [460 × 15 mm], Büchi precolumn [110 × 15 mm], Silica gel 60 [Merck, 15-40 μm]). HPLC was performed on a 1260 Agilent instrument, equipped with a degasser, a quaternary pump, an autosampler, an UV detector at 210 nm. Semipreparative separation was carried out on a C-18 column (250 mm × 10 mm id, 5 µm; Phenomenex LUNA) at room temperature and protected by a guard column. Eluent: (A) 0.01% (v/v) aqueous trifluoroacetic acid and (B) acetonitrile, 3 mL/min, detection at 210 nm. Gradient: 35% B to 45% B for 20 minutes, 45% B for 10 minutes.
Plant Material
Haplocoelum congolanum Hauman. root bark was collected from Yangambi (Democratic Republic of Congo) and identified by H. Breyne. A voucher specimen (No. 2845) is deposited in the herbarium of the Laboratory of Botanic of National University of Zaïre in Kinshasa and the National Botanical Garden in Brussels.
Extraction and Isolation
Dried powdered root bark (800 g) of H. congolanum was macerated in a 80% ethanolic solution and refluxed for 3 hours. After evaporation of the solvent under vacuum, the obtained residue was treated by successive protocols as dissolution in methanol, filtration, precipitation in diethyl ether, dialysis, and treatment by charcoal
6
to give 8.5 g of a crude saponin mixture. An aliquot (4 × 150 mg) of this residue was subjected to MPLC (1.5 × 46 cm, 2.5 mL/min) over silica gel using a CHCl3-MeOH-H2O gradient (80/20/2, 70/30/5, 60/32/7) to give 22 fractions Fr. 1 to Fr. 22. Fr. 3 and Fr. 4 were combined to give
Acid Hydrolysis and GC Analysis
Each compound (3 mg) was hydrolyzed with 2 N aq. CF3COOH (5 mL) for 3 hours at 95°C. After extraction with CH2Cl2 (3 × 5 mL), the aqueous layer was repeatedly evaporated to dryness with MeOH until neutral, and then analyzed by TLC over silica gel (CHCl3-MeOH-H2O 8/5/1) by comparison with authentic samples. Furthermore, the residue of sugars was dissolved in anhydrous pyridine (100 µL), and L-cysteine methyl ester hydrochloride (0.06 mol/L) was added. The mixture was stirred at 60°C for 1 hour, then 150 µL of hexamethyl-disilazane-trimethylchlorosilane (3:1) was added, and the mixture was stirred at 60°C for another 30 minutes. The precipitate was centrifuged off, and the supernatant was concentrated under N2 stream. The residue was partitioned between n-hexane and H2O (0.1 mL each), and the hexane layer (1 µL) was analyzed by GC.
12
The absolute configurations were determined by comparing the retention times with thiazolidine derivatives prepared in a similar way from standard sugars (Sigma-Aldrich): L-rhamnose, D-glucose, and L-arabinose for
3-O-(4-O-[3-Hydroxy-3-Methylglutaryl])-α -l-Arabinopyranosyl-(1→3)-α -l-Rhamnopyranosyl-(1→2)-[β -d-Glucopyranosyl-(1→4) ]-α-l- Arabinopyranosyloleanolic Acid (1)
White, amorphous powder.
[α]25 D: +23 (c 0.3, MeOH)
1H NMR and 13C NMR (CD3OD, 600 MHz and 150 MHz): Tables 1 and 2.
HR-ESIMS (positive-ion mode) m/z 1195.5872 [M+Na]+ (calculated for C58H92O24Na, 1195.5876) ; ESIMS (positive-ion mode) m/z 1195 [M+Na]+.
3-O-α-l-Arabinofuranosyl-(1→3)-α -l-Rhamnopyranosyl-(1→2)-[β -d-Glucopyranosyl-(1→4)]-α-l-Arabinopyranosyloleanolic Acid (2)
White, amorphous powder.
[α]25 D: – 12 (c 0.1, MeOH)
1H NMR and 13C NMR (CD3OD, 600 MHz and 150 MHz): Tables 1 and 2.
HR-ESIMS (positive-ion mode) m/z 1051.2016 [M+Na]+ (calculated for C52H84O20Na, 1051.2011) ; ESIMS (positive-ion mode) m/z 1051 [M+Na]+.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed no financial support for the research, authorship, and/or publication of this article.
