Abstract
The glycosylation of piceid and resveratroloside was investigated using glucosyltransferase from Phytolacca americana expressed in Escherichia coli. The enzyme glucosylated piceid at its 5- and 4′-positions to give resveratrol 3,5-diglucoside and resveratrol 3,4′-diglucoside, respectively. Resveratroloside was glucosylated to resveratrol 3,4′-diglucoside by the enzyme.
Cultured plant cells have been studied as useful agents for biotransformation reactions because of their potential to produce specific secondary metabolites such as flavors, pigments, and agrochemicals. 1 The reactions involved in the biotransformation of organic compounds by cultured plant cells include oxidation, reduction, hydroxylation, esterification, methylation, isomerization, hydrolysis, and glycosylation. Glycosylation is a characteristic biotransformation reaction in cultured plant cells because glycosyltransferases are widespread in plants. Also, it has been the subject of increasing attention, because a one-step enzymatic glycosylation is more convenient than a chemical one, which requires tedious steps such as protection-deprotection of the hydroxy groups of the sugar moieties. 2 -7 In addition, the glycosylation of bioactive compounds can enhance their water-solubility, physicochemical stability, intestinal absorption, and biological half-life, and improve their bio- and pharmacological properties. We recently reported that glucosyltransferase from Phytolacca americana (PaGT) expressed in Escherichia coli had high potential for the glucosylation of quercetin. 8
Herein, we report, for the first time, the glucosylation of piceid (resveratrol 3-O-β-d-glucoside) (
The plasmids prepared with pQE30, including cDNA of PaGT, were transformed into E. coli M15 cells. The expressed glucosyltransferase, PaGT, was used for the glucosylation reaction. Analysis by high performance liquid chromatography (HPLC) of the reaction mixture containing piceid (
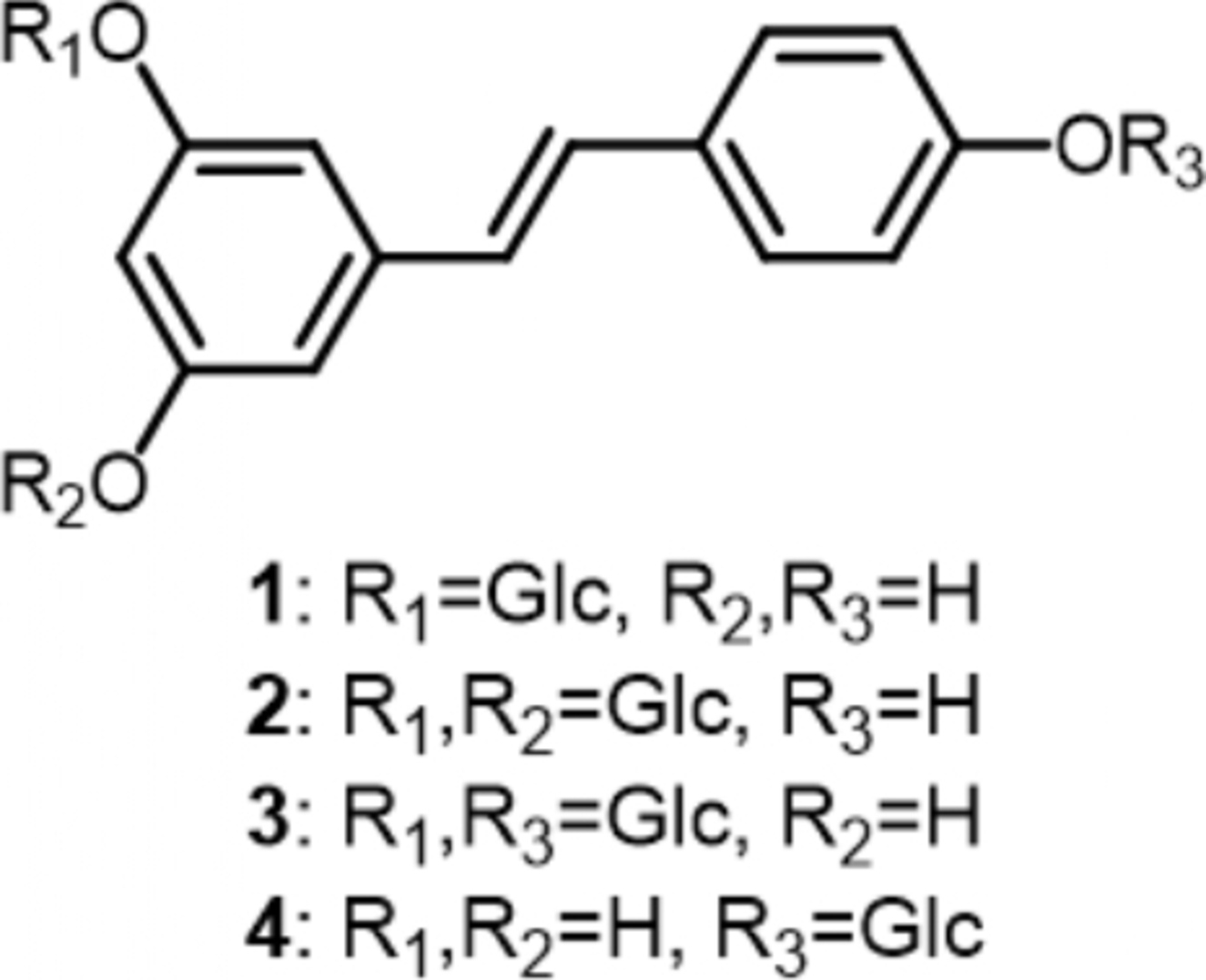
Chemical structures of substrates 1 and 4, and products 2 and 3.
When resveratroloside (
Thus, it was demonstrated that PaGT expressed in E. coli can glucosylate piceid at its 5- and 4′-positions to give resveratrol 3,5-diglucoside and resveratrol 3,4′-diglucoside, respectively, and resveratroloside at its 3-position to afford resveratrol 3,4′-diglucoside. Studies on the glucosylation of other stilbene compounds by glucosyltransferase (PaGT) expressed in E. coli are now in progress.
Experimental
General
Piceid used as substrate was purchased from Wako Pure Chemicals Co. and was used without further purification. The cultured plant cells of P. americana were subcultured at 4-week intervals on solid medium containing 2% glucose, 1 ppm 2,4-dichlorophenoxyacetic acid, and 1% agar (adjusted to pH 5.7) in the dark.
Glycosylation Procedure
cDNA of PaGT was cloned into pQE30, and the resulting plasmids were transformed into E. coli M15 cells. The expression and purification of PaGT were performed as described previously. 8 The purified enzyme solution was dialyzed with 50 mM Tris-HCl (pH 7.2) containing 5 mM dithiothreitol, and stored at –80°C. Glucosylation reactions were performed at 35°C for 24 hours in 5 mL of 50 mM potassium phosphate buffer (pH 7.2) supplemented with 50 µM substrate, 100 µM UDP-glucose, and 5 µM enzyme. The reaction was stopped by adding 1.5% trifluoroacetic acid; the reaction mixture was analyzed by HPLC. The reaction mixture was extracted with n-BuOH. The n-BuOH fraction was concentrated by evaporation and the residue was dissolved in water. The water fraction was applied to Diaion HP20, washed with water, and eluted with methanol. The methanol solution was subjected to preparative HPLC [column: CrestPak C18S; solvent: CH3CN:H2O = 15:85 (v/v); flow rate: 1.0 mL/min; and column temperature: 40°C].
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
