Abstract
Seven compounds, namely aminobenzoates A to D (
para-Aminobenzoic acid (PABA), a vital intermediate product in the folate synthesis of bacteria, fungi, and plants, is essential for cell growth and survival. 1 In 1940, Woods discovered that PABA was able to reverse the bacteriostatic action of sulfonamides, and this finding leads to the recognition of the biological importance of PABA. 2,3 Owing to the structure similarity and higher concentration, sulfonamides can replace PABA in the synthesis of dihydrofolate in vivo, which can block the synthesis of dihydrofolate and folate in microorganisms and terminate life prolongation. 4 The potassium salt of PABA (under the trade name Potaba) was approved to treat certain fibrotic skin disorders by increasing oxygen consumption in the skin, such as scleroderma and Peyronie’s disease. 5,6 Therefore, it is necessary to continue exploring PABA analogs for adding more therapeutical options.
Strain SP301 was constructed for an initial purpose to obtain novel ansamycins from Streptomyces sp. S010 by ansa gene heterologous expression in Streptomyces sp. LZ35.
7
Streptomyces sp. S010 and LZ35 were 3-amino-5-hydroxy benzoic acid positive strains which have the potential to produce new ansamycins. Our research group did a thorough study of strain LZ35, and a series of novel compounds were isolated from it.
8
-13
However, instead of obtaining new ansamycins from this effort, 7 compounds were isolated from Streptomyces sp. SP301 at the first time. Here, we showed the fermentation, isolation, and structure elucidation of 7 compounds, namely aminobenzoates A to D (
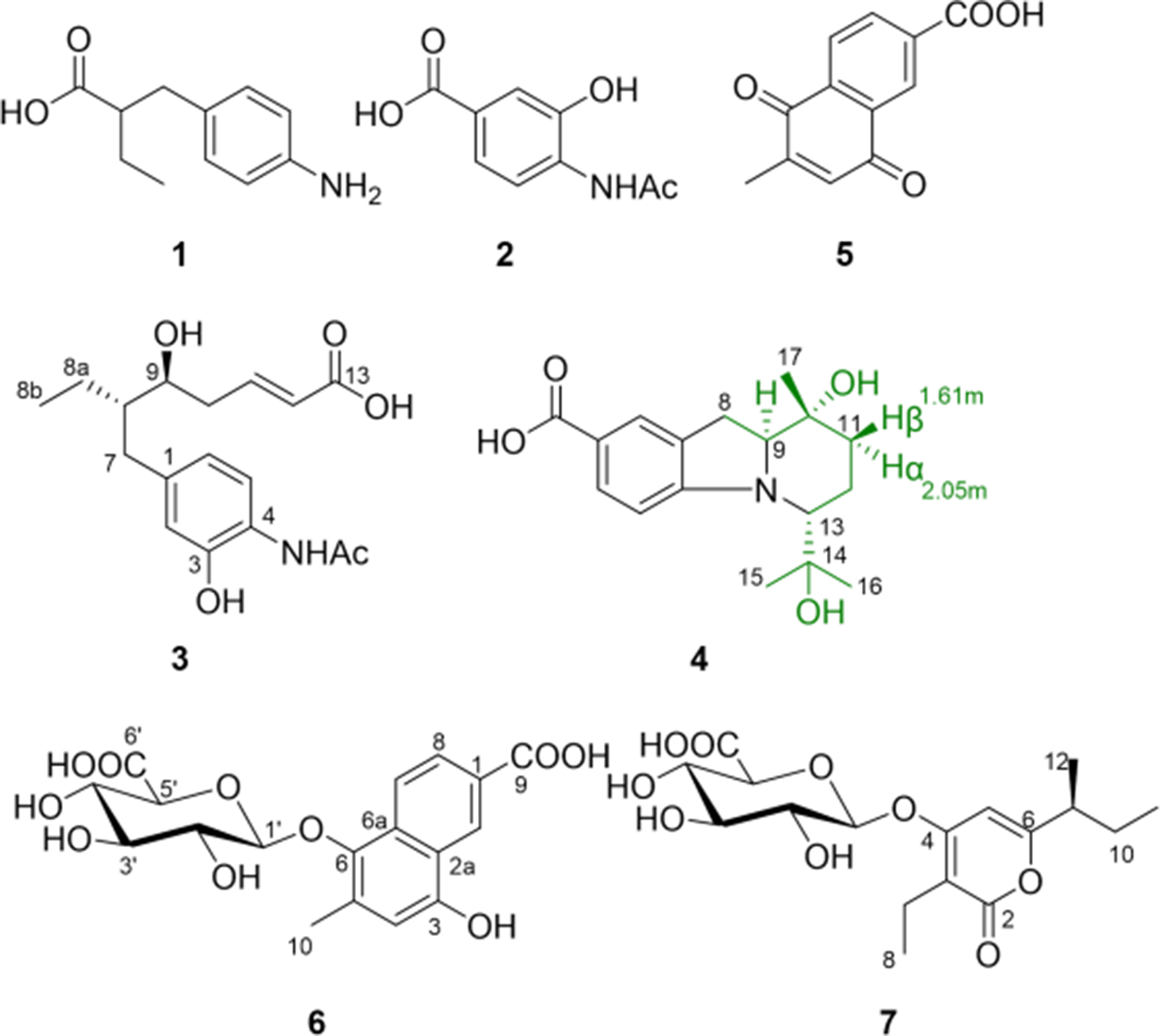
Structures of compounds 1 to
According to the literatures search and database comparison, we found that aminobenzoate A (
The high-resolution Mass Spectrometry of aminobenzoate C (
1H NMR Spectroscopic Data of Compounds of 1 to

13C NMR Spectroscopic Data of Compounds 1 to

Aminobenzoate D (
The HR-ESIMS of naphthalenecarboxylate (
The molecular formula of glycosylatelactone A (
The antimicrobial activities of compounds
In summary, we isolated and characterized 7 compounds from Streptomyces sp. SP301 strain, including aminobenzoates A to D (
Aminobenzoates A to D (
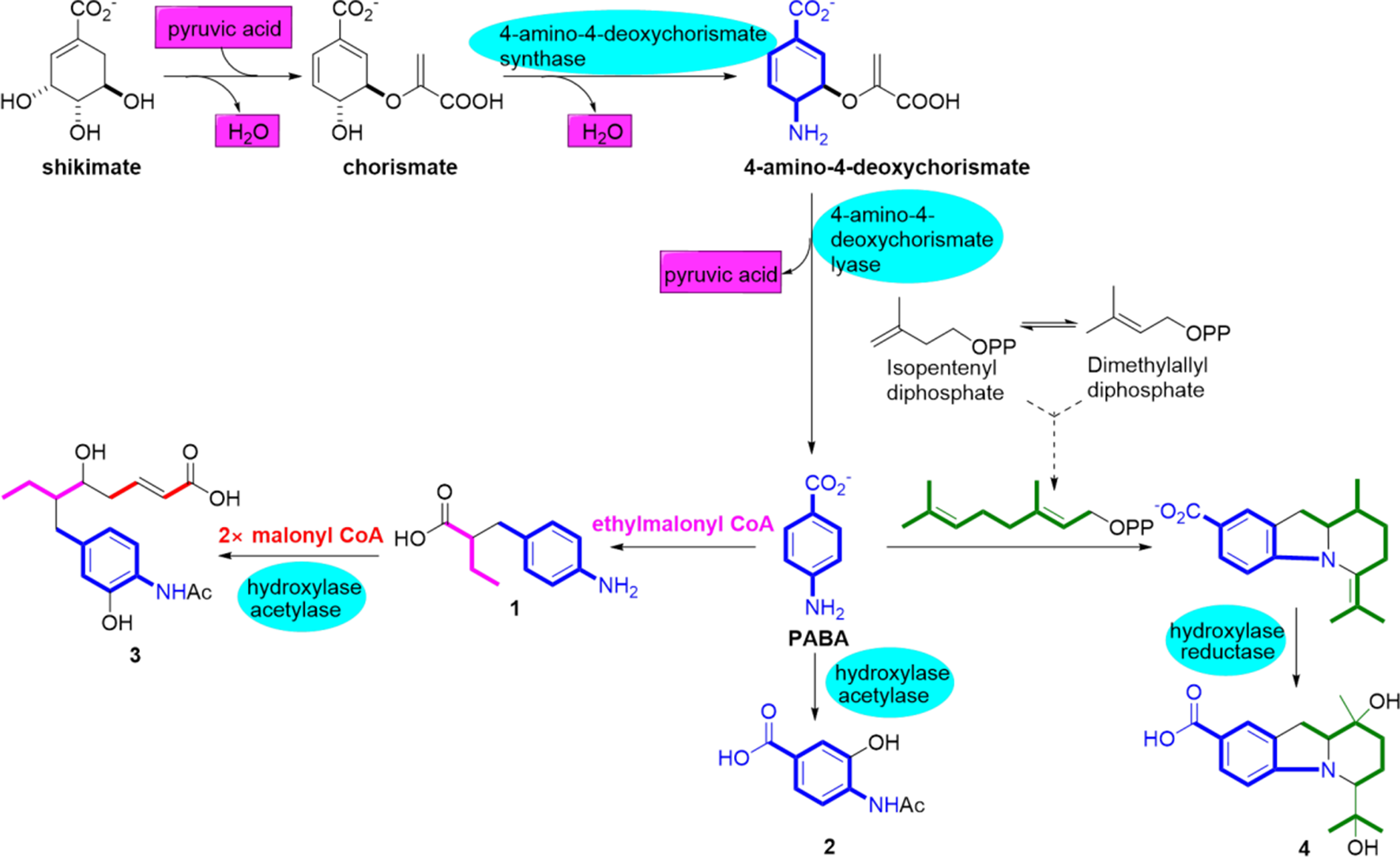
Proposed biosynthetic pathway for compounds 1 to
Shikimic acid can be turned into PABA via the shikimate pathway, which is a significant metabolic pathway in plants, fungi, and microorganisms that can be used for the biosynthesis of aromatic compounds.
18
Chorismate, the anionic form of chorismic acid, plays an important role as a branch point and synthetic precursor for the generation of most aromatic compounds in plants and microorganisms, such as various aromatic amino acids, quinones, salicylic acids, the folate precursor para-aminobenzoate, polyketones, polyenes, and terpenoids.
19
Chorismate is transformed into PABA by the enzymes 4-amino-4-deoxychorismate synthase and 4-amino-4-deoxychorismate lyase.
20
para-Aminobenzoic acid is used as a common starter unit by aminobenzoates A to D (
Experimental
General
Optical rotations were carried out using an Anton Paar MCP200 automatic polarimeter. The UV spectra were obtained on a TU-1810 spectrophotometer (Beijing Purkinje General Instrument Co., Ltd.). NMR spectra were recorded on a Bruker DRX-600 NMR spectrometer (Bruker Daltonics Inc., Billerica, MA, United States) with tetramethylsilane as an internal standard. HR-ESIMS were measured on an LTQ-Orbitrap XL. Sephadex LH-20 was obtained from GE Amersham Biosciences (25-100 μm; Piscataway, NJ, United States) and LiChroprep RP-18 for column chromatography (CC) from Merck (40-63 μm; Darmstadt, Germany). Semipreparative High Performance Liquid Chromatography (HPLC) was performed on an Agilent 1200 equipped with a ZORBAX Eclipse XDC18 5 µm column (9.4 × 250 mm).
Strain, Fermentation, and Extraction
Streptomyces sp. SP301 was separated from No.1 soil of Nanjing arboretum, China. The mutant strain SP301 was cultured in 30 L ISP3 medium (oatmeal 30 g, saline salt 1 mL, agar 20 g, pH 7.2) for 10 days at 28°C. The cultures (a total volume of 30 L) were chopped, diced, and extracted 3 times overnight with an equal volume of EtOAc/MeOH/AcOH 80:15:5 (v/v/v) at room temperature and partitioned between ddH2O and EtOAc until the EtOAc layer was colorless. Then, the EtOAc extract was dried with Na2SO4, and the solvent was removed under vacuum at 38°C. The EtOAc extract was partitioned with light petroleum (PE) and MeOH until the PE layer was colorless. The MeOH solution was concentrated under vacuum at 38°C to obtain the MeOH extract (7.5 g).
Isolation and Purification of Compounds 1 to 7
The MeOH extract (7.5 g) was subjected to MPLC over RP-18 silica gel (140 g), and 24 subfractions with 200 mL for each gradient were obtained from the elution with 30%, 50%, 70%, and 100% MeOH, respectively. In accordance with TLC results, 10 fractions, Frs. A to J, were obtained.
Fr. B (155 mg) was subjected to CC over Sephadex LH-20 (80 g) eluted with MeOH to obtain 7 fractions (Frs. B1-B7). Fr. B4 (38 mg) was purified by preparative HPLC (eluted with 12% acetonitrile, 4 mL/min, UV 320 nm) to yield
Aminobenzoate A (1)
1H (CD3OD, 400 MHz): Table 1.
13C NMR (CD3OD, 100 MHz): Table 2.
HR-ESIMS: m/z 194.1178 [M+H]+ (calcd. for C11H16NO2 +, 194.1176), m/z 216.0998 [M+Na]+ (calcd. for C11H15NO2Na+, 216.0995).
Aminobenzoate B (2)
1H (CD3OD, 400 MHz): Table 1.
13C NMR (CD3OD, 100 MHz): Table 2.
HR-ESIMS: m/z 196.0605 [M+H]+ (calcd. for C9H10NO4 +, 196.0604), m/z 218.0426 [M+Na]+ (calcd. for C9H9NO4Na+, 218.0424)..
Aminobenzoate C (3)
1H (CD3OD, 400 MHz): Table 1.
13C NMR (CD3OD, 100 MHz): Table 2.
HR-ESIMS: m/z 322.1649 [M+H]+ (calcd. for C17H24NO5 +, 322.1649).
Aminobenzoate D (4)
1H (CD3OD, 400 MHz): Table 1.
13C NMR (CD3OD, 100 MHz): Table 2.
HR-ESIMS: m/z 306.1700 [M+H]+ (calcd. for C17H24NO4 +, 306.1700).
Naphthalenecarboxylate A (5)
1H (CD3OD, 400 MHz): Table 1.
13C NMR (CD3OD, 100 MHz): Table 2.
HR-ESIMS: m/z 217.0491 [M+H]+ (calcd. for C12H9O4 +, 217.0495).
Naphthalenecarboxylate B (6)
1H (CD3OD, 400 MHz): Table 1.
13C NMR (CD3OD, 100 MHz): Table 2.
HR-ESIMS: m/z 395.0973 [M+H]+ (calcd. for C18H19O10 +, 395.0973).
Glycosylatelactone A (7)
1H (CD3OD, 400 MHz): Table 1.
13C NMR (CD3OD, 100 MHz): Table 2.
HR-ESIMS: m/z 373.1499 [M+H]+ (calcd. for C17H25O9 +, 373.1493).
Supplemental Material
Supporting information - Supplemental material for Isolation and Identification of Secondary Metabolites From Streptomyces sp. SP301
Supplemental material, Supporting information, for Isolation and Identification of Secondary Metabolites From Streptomyces sp. SP301 by Mengyujie Liu, Peng Shi, Chunhua Lu, and Lihong Zhong in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the National Natural Science Foundation of China (81373304, 81530091, and 81673317), the Independent Innovation Foundation of Shandong University (IIFSDU, 2014JC027) and the National ADR Monitoring Construction Project of China (20171X001-04).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
