Abstract
The first total synthesis of (8R) and (8S) methyl β-
The isolation and structural determination of methyl β-

Chemical structures of methyl 8-β-
We anticipated that synthesis of (8R) and (8S) methyl 8-β-

(a) EtMgBr/THF (76%); (b) HgO, HgBr2, drierite/CHCl3, 1-bromo-2,3,4,6- tetraacetylglucose (10) (19% for 11a, 16% for 11b); (c) NaOMe/Me0H (89% for 12a, 95% for 12b); (d) CuI (I), 35% aq. C2H5NH2, NHOH-HCl, 5-bromo-pent-4-yn-1-oic acid (14) (54% for 6a, 34% for 6b).
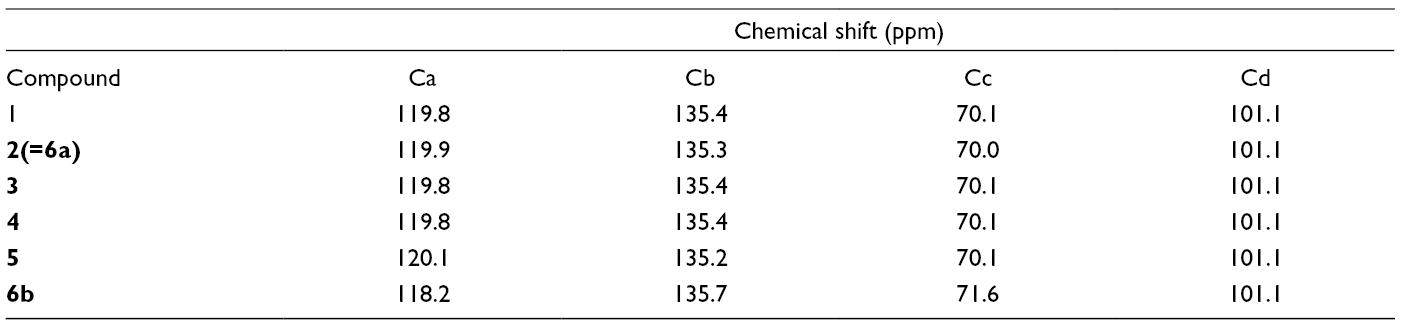
The partial 1H- and 13C-NMR spectral data for methyl β-
1 H-NMR Chemical Shift for the Partial Structure.

13C-NMR Chemical Shift for the Partial Structuure.
The first total syntheses of (8R) and (8S) methyl 8-β-
Experimental
General
Spectra were obtained with the following instruments: IR, Hitachi 285 spectrometer; optical rotation values, JASCO DIP-4 polarimeter; NMR, Brucker AM-500 FT-NMR spectrometer and JEOL JNM-EX 270 FT-NMR system; and FD- and EI-MS, JEOL JMS-O1SG-2 and JMS-DX-300 mass spectrometers, respectively.
Preparation of Methyl ( 8 R ) and ( 8 S ) Methyl 8-β- d-Glucopyranosyloxydec-9-Ene-6,4-Diyn-1-Oate (6a, 6b)
1-Trimethylsilylpent-4-En-1-Yn-3-Ol (9)
EtMgBr in THF was prepared from Mg (864 mg, 36 mmol) and EtBr (4.2 g, 2.9 mL, 38 mmol) in THF (40 mL) using a general method. Trimethylsilylacetylene (
IR υ max (film) cm−1: 3290, 2930, 2140, 1400, 1240, 1220, 1020, 980, 840, 760.
1H-NMR (270 MHz, CDCl3) ppm: 5.95 (1H, ddd, J = 16.8, 10.2, 5.6 Hz), 5.45 (1H, d, J = 16.8 Hz), 5.19 (1H, d, J = 10.2 Hz), 4.87 (1H, br.t, J = 5.6 Hz), 0.18 (9H, s).
13C-NMR (67.8 MHz, CDCl3) ppm: 137.0, 116.5, 104.7, 90.8, 63.4, 0.2.
EI-MS m/z (rel. int.): 99 (8), 91 (10), 83 (100), 73 (38), 55 (25).
(3R) and (3S) 3-Tetra-O-Acetyl-β-d -Glucopyranosyloxy-1-Trimethylsilylpent-4-En-1-Yne (11a, 11b)
A CHCl3 (50 mL) solution containing HgO (II) (yellow) (5.6 g), HgBr2 (468 mg, 1.3 mmol), and drierite (14 g) was placed in a 500 mL three-necked, round-bottom flask equipped with a stirring bar, septum cap, and N2 balloon. Compound
(3R) 3-Tetra-O-Acetyl-β-d -Glucopyranosyloxy-1-Trimethylsilylpent-4-En-1-Yne (11a)
IR υ max (KBr) cm−1: 2900, 2200, 1750, 1370, 1260, 1230, 1090, 1060, 980, 840, 760.
1H-NMR (500 MHz, CDCl3) ppm: 5.86 (1H, ddd, J = 16.9, 10.4, 6.4 Hz), 5.42 (1H, dt, J = 16.9, 1.3 Hz), 5.29 (1H, dt, J = 10.4, 1.3 Hz), 5.20 (1H, t, J = 9.5 Hz), 5.09 (1H, t, J = 9.5 Hz), 5.04 (1H, dd, J = 9.5, 8.0 Hz), 4.95 (1H, d, J = 6.4 Hz), 4.70 (1H, d, J = 8.0 Hz), 4.24 (1H, dd, J = 12.2, 4.6 Hz), 4.12 (1H, dd, J = 12.2, 2.1 Hz), 3.66 (1H, ddd, J = 9.5, 4.6, 2.1 Hz), 2.09 (3H, s), 2.05 (3H, s), 2.02 (3H, s), 2.01 (3H, s), 0.19 (9H, s).
13C-NMR (125.8 MHz, CDCl3) ppm: 170.7, 170.3, 169.4, 169.3, 134.2, 118.8, 100.6, 97.5, 93.2, 73.0, 71.8, 71.1, 70.5, 68.4, 62.0, 20.7, 20.6, -0.2.
EI-MS m/z (rel. int.): 469 (0.1), 259 (3), 245 (6), 169 (9), 139 (20), 109 (12), 97 (16), 59 (16), 43 (100).
FDHRMS m/z [M]+ calculated for C22H33O10Si: 485.1843, found 485.1867.
(3S) 3-Tetra-O-Acetyl-β-d -Glucopyranosyloxy-1-Trimethylsilyl-Pent-4-En-1-Yne (11b)
IR υ max (KBr) cm−1: 2900, 2100, 1750, 1560, 1220, 1040.
1H-NMR (500 MHz, CDCl3) ppm: 5.85 (1H, ddd, J = 17.0, 10.1, 5.1 Hz), 5.49 (1H, dd, J = 17.0, 1.2 Hz), 5.26 (2H, m), 5.11 (1H, t, J = 9.9 Hz), 5.02 (2H, m), 4.89 (1H, d, J = 8.0 Hz), 4.29 (1H, dd, J = 12.3, 4.6 Hz), 4.13 (1H, br.d, J = 12.3 Hz), 3.73 (1H, m), 2.09 (3H, s), 2.04 (3H, s), 2.03 (3H, s), 2.02 (3H, s), 0.21 (9H, s).
13C-NMR (125.8 MHz, CDCl3) ppm: 170.6, 170.3, 169.3, 133.6, 118.0, 100.4, 97.4, 93.4, 72.6, 71.8, 70.9, 68.6, 68.4, 61.8, 20.7, 20.6, 20.5, −0.3.
EI-MS m/z (rel. int.): 469 (0.1), 259 (6), 245 (15), 169 (26), 139 (43), 97 (22), 59 (24), 43 (100).
FDHRMS m/z [M]+ calculated for C22H33O10Si: 485.1843, found 485.1824.
(3R) 3-β-d -Glucopyranosyloxypent-4-En-1-Yne (12a)
NaOMe (4 mmol) in MeOH was added to a stirred mixture of
IR υ max (film) cm-−1: 3300, 2900, 2100, 1400, 1250, 1060.
1H-NMR (500 MHz, CD3OD) ppm: 5.89 (1H, ddd, J = 17.1, 10.1, 7.3 Hz), 5.46 (1H, br.d, J = 17.1 Hz), 5.36 (1H, br.d, J = 10.1 Hz), 5.07 (1H, br.dt, J = 7.3, 2.1 Hz), 4.36 (1H, d, J = 7.8 Hz), 3.76 (1H, dd, J = 11.9, 2.1 Hz), 3.66 (1H, dd, J = 11.9, 5.9 Hz), 3.34-3.19 (4H, m), 2.97 (1H, d, J = 2.1 Hz).
13C-NMR (125.8 MHz, CD3OD) ppm: 135.9, 119.5, 100.8, 82.1, 78.1, 76.2, 74.9, 71.6, 69.6, 62.8.
EI-MS m/z (rel. int.): 97 (14), 81 (41), 73 (43), 65 (81), 60 (49), 55 (60), 39 (100).
FDHRMS m/z [M+H]+ calculated for C11H17O6: 245.1025, found 245.1015.
(3S) 3-β-d -Glucopyranosyloxypent-4-En-1-Yne (12b)
The deprotection of
IR υ max (film) cm–1: 3300, 2900, 2100, 1400-1220, 1080.
1H-NMR (500 MHz, CD3OD) ppm: 5.96 (1H, ddd, J = 17.0, 10.2, 5.4 Hz), 5.53 (1H, dt, J = 17.0, 1.4 Hz), 5.24 (1H, dt, J = 10.2, 1.4 Hz), 5.20 (1H, m), 4.65 (1H, d, J = 7.9 Hz), 3.87 (1H, dd, J = 11.8, 1.3 Hz), 3.66 (1H, dd, J = 11.8, 1.6 Hz), 3.03 (1H, d, J = 2.2 Hz).
13C-NMR (125.8 MHz, CD3OD) ppm: 135.2, 117.9, 100.9, 80.9, 78.1, 77.6, 74.8, 71.6, 66.5, 62.7.
EI-MS m/z (rel. int.): 97 (18), 85 (19), 73 (72), 65 (100), 57 (51), 39 (46).
FDHRMS m/z [M+H]+ calculated for C11H17O6: 245.1025, found 245.1036.
5-Bromopent-4-Yn-1-Oic Acid (14):
Compound
( 8 R ) Methyl 8-β- d-Glucopyranosyloxydec-9-Ene-6,4-Diyn-1-Oate (6a):
CuI (0.5 mg) and NHOH-HCl (5 mg) were placed in a three-necked, round-bottom flask equipped with a stirring bar, septum cap, and N2 balloon. The mixture was then suspended in 35% aqueous EtNH2 (0.5 mL). Compound
[α]D 25: −55.8 (c 1.18 MeOH).
1H-NMR (500 MHz, CD3OD) ppm: 5.86 (1H, ddd, J = 17.2, 10.2, 7.1 Hz), 5.44 (1H, dd, J = 17.2, 0.9 Hz), 5.32 (1H, dd, J = 10.2, 0.9 Hz), 5.11 (1H, br.d, J = 5.1 Hz), 4.33 (1H, d, J = 7.8 Hz), 3.86 (1H, dd, J = 11.9, 2.1 Hz), 3.69 (3H, s), 3.66 (1H, m), 3.31 (1H, m), 3.22 (1H, m), 2.57 (4H, m).
13C-NMR (125.8 MHz, CD3OD) ppm: 173.7, 135.3, 120.0, 101.0, 81.0, 78.1, 78.0, 74.9, 74.3, 71.2, 71.5, 70.0, 65.6, 62.7, 52.4, 33.5, 15.7.
(8S) Methyl 8-β - d-Glucopyranosyloxydec-9-Ene-6,4-Diyn-1-Oate (6b):
The coupling reaction of
[α]D 25: −61.2 (c 0.50 MeOH).
1H-NMR (500 MHz, CD3OD) ppm: 5.93 (1H, m), 5.48 (1H, d, J = 17.0 Hz), 5.26 (2H, m), 5.20 (1H, m), 4.58 (1H, d, J = 7.8 Hz), 3.87 (1H, br.d, J = 12.2 Hz), 3.69 (3H, s), 3.66 (2H, m), 3.20 (2H, m), 2.57 (4H, m).
13C-NMR (125.8 MHz, CD3OD) ppm: 173.6, 135.7, 118.2, 101.0, 80.8, 78.2, 78.0, 74.9, 73.4, 73.0, 71.6, 65.5, 62.7, 52.4, 33.4, 15.7.
Footnotes
Acknowledgments
The authors acknowledge the assistance of Dr Eri Fukushi and Mr Yusuke Takata (Research Faculty of Agriculture, Hokkaido University) in obtaining the spectroscopic data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

