Abstract
Reinvestigation of 2-hydroxy-3-alkylnaphthoquinones oxidative dimerization was carried out. On the example of dihydrolapachol it is shown that the oxidative dimerization of 2-hydroxy-3-alkylnaphthoquinones upon treatment with lead tetraacetate or cerium ammonium nitrate in aprotic media leads to the formation of diastereomeric 5aS*,6aS*,12aS*,12bS*- and 5aS*,6aR*,12aR*,12bS*-dihydrobinaphthofurantetraones (1:1). This result is significantly different from the previous one.
The cytotoxic pentacyclic naphthazarin-derived dimer, hybocarpone (
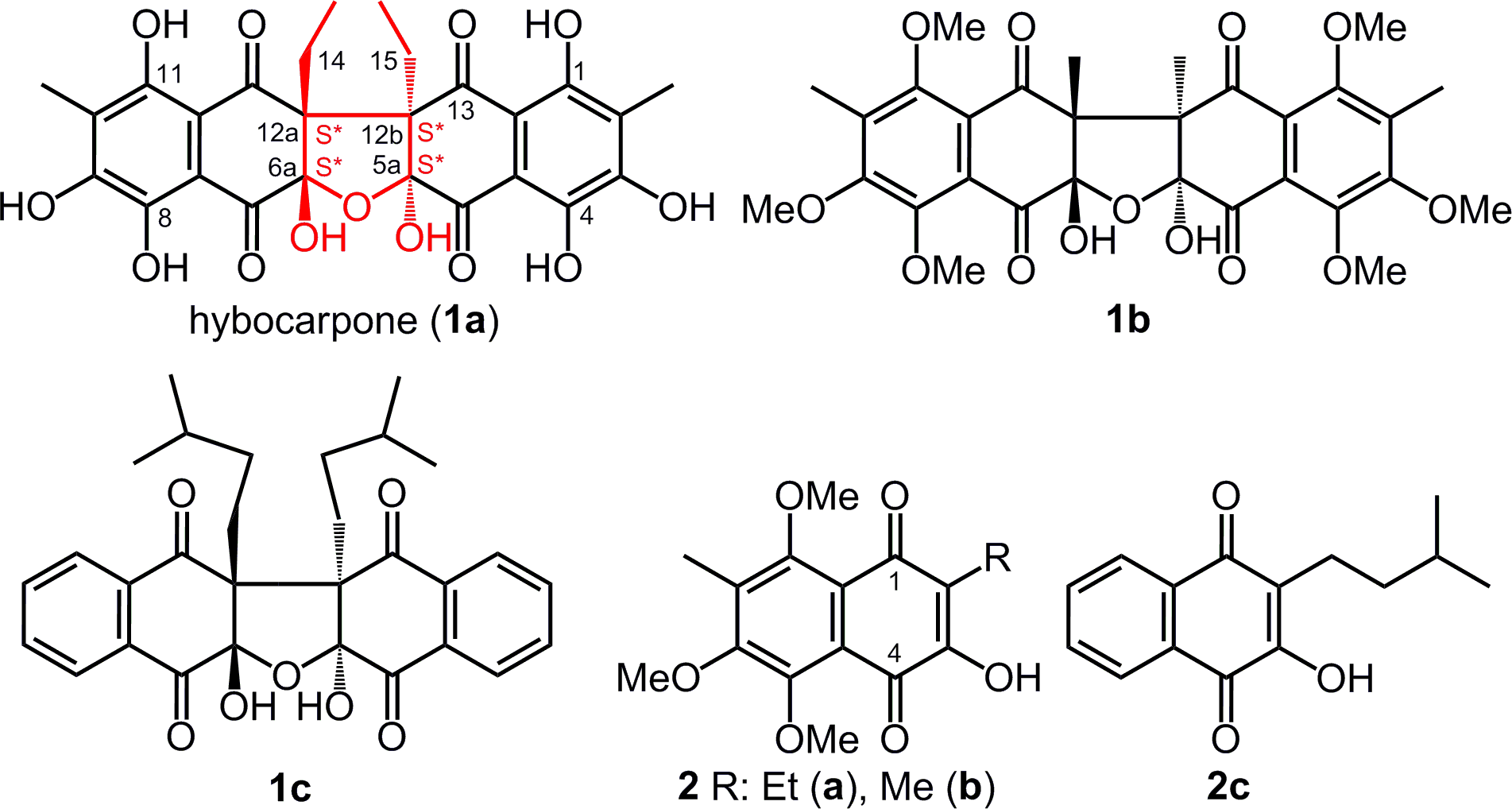
The use of the direct 2-hydroxynaphthazarin (2,5,8-trihydroxy-1,4-naphthoquinone) precursors

The products of the oxidative coupling of hydroxynaphthazarins 3a,b upon treatment with Pb(OAc)4 in benzene.
The ratio of the resulting compounds

S*S*- (6a,
It should be noted that in the previous reports
2
-4
the course of the reaction through the S*,S* hexaketone intermediate was only postulated, and the possibility of R*,S* diastereomer formation was not discussed. Based on this, the authors reported on the synthesis of only one 5aS*,6aS*,12aS*,12bS* diastereomeric product
Dihydrolapachol (
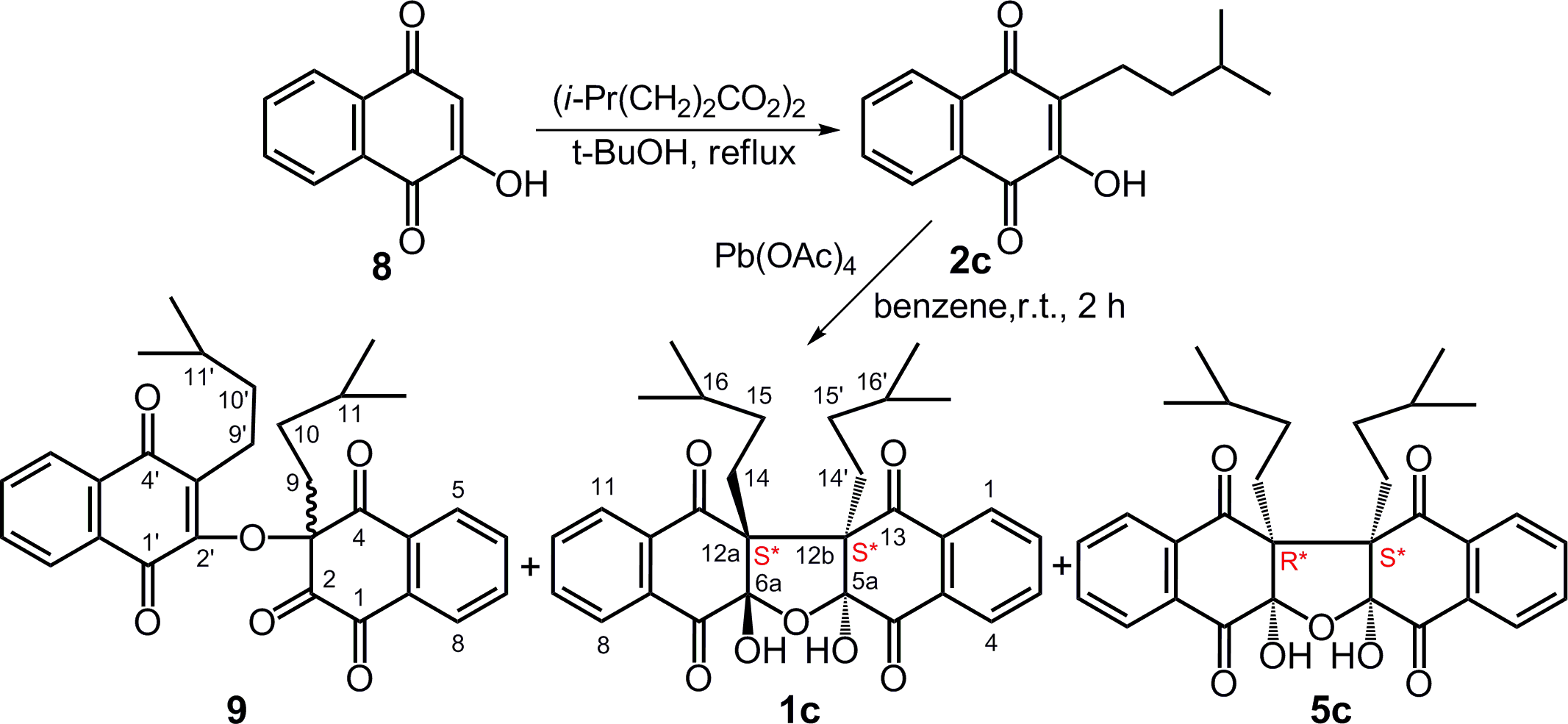
Oxidative coupling reaction of dihydrolapachol (
Two colorless products were determined to be pentacyclic compounds
1H and 13C Nuclear Magnetic Resonance Data of
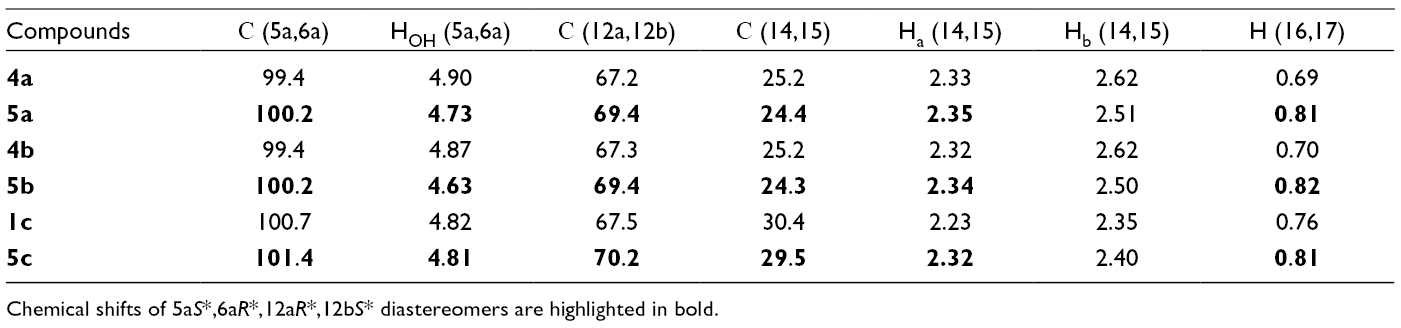
Chemical shifts of 5aS*,6aR*,12aR*,12bS* diastereomers are highlighted in bold.
In general, 2-hydroxy-3-alkylnaphthazarins 5 and 2-hydroxy-3-alkyl-1,4-naphthoquinones undergo oxidative dimerization upon treatment with lead tetraacetate or CAN in aprotic media to give diastereomeric 5aS*,6aS*,12aS*,12bS*- and 5aS*,6aR*,12aR*,12bS*-dihydrobinaphthofurantetraones in a ratio of circa 1:1. The ratio of arising compounds is determined by the equally possible formation of the corresponding S*,S* and R*,S* hexaketone intermediates. This conclusion is completely different from the results described in the article 3 where synthesis of only one 5aS*,6aS*,12aS*,12bS* diastereomeric product is noted.
Experimental
The melting points were determined with a Boetius apparatus and are uncorrected. For all purposes analytical grade solvents were used. 1H and 13C NMR spectra were recorded on Bruker Avance-300, Avance-500, Avance-700 spectrometers at 300 and 75 MHz, 500 and 125 MHz, and 700 and 176 MHz, respectively. The chemical shifts (δ) are represented as parts per million (ppm) relative to tetramethylsilane. High-resolution electrospray ionization mass spectra were measured on an Agilent 6510Q-TOF LC mass spectrometer. The course of reactions and the purity of the compounds obtained were assessed by thin-layer chromatography (TLC). Merck Kieselgel 60 F-254 plates were preliminarily treated with 0.05 M tartaric acid in MeOH and dried at ~50°C for 1 hour. Preparative TLC was performed on glass plates (20 × 20 cm) precoated with silica gel (Merck), 5 to 40 μm. The yields were not optimized.
C-Alkylation of Lawsone (8) With 4-Methylpentanoyl Peroxide
Alkanoyl peroxide was added dropwise to a boiling solution of lawsone (1.74 g, 10 mmol) in t-BuOH (30 mL), monitoring the reaction course by TLC. When the conversion reached 50% to 60%, by TLC the reaction was stopped by cooling, the solvent was removed in vacuo, and the residue was separated by column chromatography on silica gel in a hexane-acetone, 3:1 to give compound
2-Hydroxy-3-Isopentylnaphthalene-1,4-Dione (2c)
MP: 88–90°C (MP: 92°C 10 ).
Rf : 0.65 (hexane-acetone, 3: 1).
1H NMR (300 MHz, CDCl3): 0.95 (6H, d, J = 6.6 Hz, 2Me), 1.35-1.46 (2H, m), 1.59-1.67 (1H, m), 2.58 (2H, dd, J 1 = 8.2 Hz, J 2 = 8.0 Hz), 7.34 (1H, br. s, OH), 7.69 (1H, dt, J 1 = 7.5 Hz, J 2 = 1.4 Hz, Harom), 7.77 (1H, dt, J 1 = 7.5 Hz, J 2 = 1.5 Hz, Harom), 8.06 (1H, dd, J 1 = 7.5 Hz, J 2 = 1.5 Hz, Harom), 8.09 (1H, dd, J 1 = 7.3 Hz, J 2 = 1.4 Hz, Harom).
13C NMR (75 MHz, CDCl3): 2 × 21.2, 22.1, 28.2, 37.1, 73.9, 125.7, 2 × 126.4, 132.7, 132.9, 134.6, 152.8, 181.3, 184.3.
HRMS: m/z [M + H+] calcd for C15H17O3: 245.1172; found: 245.1128.
Oxidative Coupling of 2c
Commercially available Pb(OAc)4 (135 mg, 0.3 mmol) was added with vigorous stirring to a solution of
3-Isopentyl-3-(3-Isopentyl-1,4-Dioxo-1,4-Dihydronaphthalen-2-Yloxy)naphthalene-1,2,4(3H)-Trione (9)
MP: 76-79°C (MP: 77-81°C 9 ).
Rf : 0.50 (hexane-acetone, 3:1).
1H NMR (500 MHz, CDCl3): 0.82 (3H, d, J = 6.5 Hz, Me-11a), 0.84 (3H, d, J = 6.5 Hz, Me-11b), 1.02 (6H, d, J = 6.6 Hz, 2 × Me-11′), 1.33 (1H, m, H-11), 1.42 (1H, m, H-11′), 1.46 (1H, m, Ha-9), 1.56 (2H, m, H-10), 1.74 (1H, m, Hb-9), 2.03 (2H, m, H-10′), 2.80 (2H, t, J = 7.1 Hz, H-9′), 7.50 (1H, dt, J 1 = 7.5 Hz, J 2 = 1.3 Hz, H arom-7′), 7.64 (1H, dt, J 1 = 7.4 Hz, J 2 = 1.3 Hz, H arom-6′), 7.65 (1H, d, J = 7.2 Hz, H arom-8′), 7.92 (1H, dt, J 1 = 7.2 Hz, J 2 = 1.6 Hz, Harom-6), 7.95 (1H, dt, J 1 = 7.4 Hz, J 2 = 2.0 Hz, Harom-7), 8.02 (1H, m, Harom-5′), 8.22 (1H, dd, J 1 = 7.0 Hz, J 2 = 2.0 Hz, Harom-5), 8.41 (1H, dd, J 1 = 7.0 Hz, J 2 = 2.0 Hz, Harom-8).
13C NMR (125 MHz, CDCl3): 2 × 22.2, 22.4, 2 × 22.5, 28.0, 28.8, 31.6, 35.6, 37.0, 93.2, 126.2, 126.6, 128.5, 129.0, 130.7, 131.7, 132.9, 133.0, 133.7, 134.0, 134.4, 134.8, 136.1, 150.9, 180.8, 182.2, 184.3, 184.4, 189.1.
HRMS: m/z [M + H+] calculated for C30H31O6: 487.2115; found: 487.1092.
(5aS*,6aS*,12aS*,12bS*)-12a,12b-Diisopentyl-5a,6a-Dihydroxy-Dinaphtho[2,3-B:2′,3′-d]furan-5,7,12,13(5aH,6aH,12aH,12bH)-Tetraone (1c)
Rf : 0.40 (hexane-acetone, 3:1).
1H NMR (700 MHz, CDCl3): 0.73 (2H, m, Ha-15,15′), 0.76 (12H, d, J = 6.7 Hz, 4Me-16,16′), 0.93 (2H, m, Hb-15,15′) 1.45 (2H, m, H-16,16′), 2.23 (2H, dt, J 1 = 13.2 Hz, J 2 = 4.2 Hz, Ha-14,14′), 2.35 (2H, dt, J 1 = 13.2, J 2 = 4.1 Hz, Hb-14,14′), 4.82 (2H, br. s, OH-5a,6a), 7.74 (2H, dt, J 1 = 7.5 Hz, J 2 = 1.2 Hz, Harom-2,10), 7.83 (2H, dt, J = 7.6 Hz, 0.8 Hz, Harom-3,9), 8.02 (2H, dd, J 1 = 7.6 Hz, J 2 = 0.8 Hz, Harom-1,11), 8.19 (2H, dd, J 1 = 7.9 Hz, J 2 = 0.8 Hz, Harom-4,8).
13C NMR (176 MHz, CDCl3): 22.1, 22.3, 28.8, 30.4, 34.1, 67.6, 100.6, 127.1, 127.6, 129.7, 133.6, 135.3, 137.5, 191.1, 193.9.
HRMS: m/z [M + H+] calculated for C30H33O7: 505.2221; found 505.2202.
(5aS*,6aR*,12aR*,12bS*)-12a,12b-Diisopentyl-5a,6a-Dihydroxy-Dinaphtho[2,3-B:2′,3'-d]furan-5,7,12,13-(5aH,6aH,12aH,12bH)-Tetraone (5c)
Rf : 0.35 (hexane-acetone, 3: 1).
1H NMR (500 MHz, CDCl3): 0.81 (12H, d, J = 6.7 Hz, Me-16,16’), 0.86 (2H, m, Ha-15,15’), 1.03 (2H, m, Hb-15,15’), 1.50 (2H, m, H-16,16’), 2.32 (2H, dt, J 1 = 12.5 Hz, J 2 = 4.9 Hz, Ha-14,14’), 2.39 (2H, dt, J 1 = 12.5 Hz, J 2 = 4.2 Hz, Hb-14,14’), 4.81 (2H, br. s, OH-5a,6a), 7.69 (2H, dt, J 1 = 7.5 Hz, J 2 = 1.4 Hz, Harom-2,10), 7.73 (2H, dt, J 1 = 7.4 Hz, J 2 = 1.5 Hz, Harom-3,9), 7.85 (2H, dd, J 1 = 7.6 Hz, J 2 = 1.4 Hz, Harom-1,11), 7.95 (2H, dd, J 1 = 7.4 Hz, J 2 = 1.3 Hz, Harom-4,8).
13C NMR (125 MHz, CDCl3): 2 × 22.3, 29.0, 29.5, 34.3, 70.2, 101.4, 2 × 127.3, 130.9, 134.1, 135.4, 136.4, 191.1, 195.3.
HRMS: m/z [M + H+] calculated for C30H33O7: 505.2221; found 505.2202.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
