Abstract
The synthesis of A-azepanobetulinic acid N-methylpiperazinylamide was performed through a series of transformations (oximation, Beckmann rearrangement, reduction) of previously synthesized betulonic acid N-methylpiperazinylamide. In vitro cytotoxic activity was detected for the obtained compound against a number of cancer cell lines, and its potential was revealed as an antibacterial agent.
Keywords
Pentacyclic triterpenoids are a class of natural compounds widely represented in plant sources. The best-known representatives are betulinic, oleanolic, ursolic, and glycyrrhetinic acids. As evident from literature data, triterpenoids are characteristic of a diverse biological activity, especially antitumor and antimicrobial. For example, betulinic acid is in stage II clinical trials as an antitumor agent against melanoma. 1 Oleanolic acid shows high activity against hepatocellular carcinoma. 2 Besides, oleanolic and ursolic acids are well-known agents against Mycobacterium tuberculosis. 3
Most terpene structures are characterized by common drawbacks, including low solubility, poor bioavailability, and instability to metabolism. 4 In addition, they have a weak pharmacological effect at low concentrations. 5 As a result, investigations into the chemical modification of the initial carbocyclic backbone have been undertaken in order to find biologically active compounds with an improved pharmacological profile and advanced mechanism of action against tumor cells. Various methods of triterpenoid molecule delivery to biological targets are being actively studied. 6
One of the important pharmacophore groups in the triterpenoid modifications is the piperazinyl fragment responsible for various biological activities: antibacterial, 7 anticancer, 8,9 antiviral, 10,11 antimalarial, 12 α-glucosidase inhibitory, 13 antileishmanial, 14 antifungal, 15 etc.
This paper reports on the synthesis and study of the cytotoxicity and antibacterial activity of a new triterpene conjugate with N-methylpiperazine. Based on previously obtained data, concerning the antitumor and antituberculosis activity of azepanotriterpene alcohols and their derivatives,
16,17
we carried out the transformation of the carbocyclic ring A of betulonic acid N-methylpiperazinylamide

Synthesis of betulonic acid N-methylpiperazinylamide derivatives 1
In addition, the NMR spectra definitely showed the modification of ring A. For example, the signal of C3 in the spectra of lactame
Compounds
Antibacterial Test Results for Compounds 1

Next, pDualrep2 system was used to analyze the mechanism of action of
At the second stage, compound
Cytotoxicity Test Results for Compounds 1
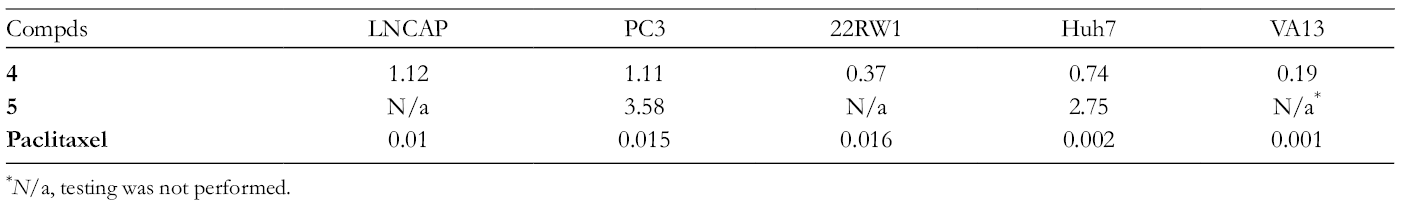
* N/a, testing was not performed.
We speculate that key factor in this case is the presence of the N-methylpiperazinylamide fragment in the structure of
Moreover, we evaluated the activity of compound
Finally, the synthesis of A-azepanobetulinic acid N-methylpiperazinylamide was performed through a series of betulonic acid N-methylpiperazinylamide transformations (oximation, Beckmann rearrangement, reduction). As a result, obtained compound demonstrated its high-value activity against various tumor cell lines, as well as antibacterial potential. High antitumor effect of N-methylpiperazinylamide derivative of betulinic acid suggests the possibility of its use as a precursor for a variety of prodrug-like conjugates.
Experimental
General
NMR spectra were recorded at the Center for Collective Use “Chemistry” of the UIC UFRS RAS. NMR spectra were recorded on a “Bruker AM-300” spectrometer at 300 (1H) and 75.5 MHz (13C) at 25°C. Chemical shifts were given in (δ in ppm) relative to the residual signals of CDCl3 (1H 7.24 ppm and 13C 76.90 ppm). Melting points were determined with a Boetius heating stage. Optical rotations were measured on Perkin-Elmer 241 polarimeter at room temperature (20°C to 22°C). Combustion analysis was performed with CHNSO-analyser (Model Euro EA 3000, Germany). TLC analysis were conducted on Sorbfil plates (Sorbpolimer, Russian Federation) using chloroform-ethylacetate, 40:1 (v/v). The substances were detected by a 10% solution of H2SO4 with subsequent heating at 100°C to 120°C for 2 to 3 minutes. Al2O3 (Reakhim, Russian Federation) was used for column chromatography. All the organic solvents were of analytical quality. Compounds
Synthesis of Compound 2
A mixture of compound
3-Oximino-17-(4-Methylpiperazin-1-yl)-Carbonylolup-20(29)-ene (2)
Yield: 0.42 g, 76%, as a white solid.
R f 0.14.
MP: 192°С.
Anal. Calcd. for С35Н57N3O2 (Мr 551.839): С 75.72; Н 10.03; N 7.16. Found: С 76.18, Н 10.41, N 7.61.
1H NMR (300 MHz, CDCl3): δ 0.86 (3H, s, H-25), 0.92 (3H, s, H-26), 0.96 (3H, s, H-24), 1.02 (3H, s, H-27), 1.12 (3H, s, H-23), 1.22-2.12 (21Н, m, H-1, H-2, H5-H7, H-9, H11-H13, H-15, H-16, H-18, H-21, H-22), 1.67 (3Н, s, H-30), 2.06-2.18 (4Н, m, H-3′, H-5′), 2.29 (3Н, s, H-7′), 2.32-2.56 (3Н, m, H-13, H-16), 2.84-3.20 (7Н, m, H-19, H-3, H-2′, H-6′, ОН), 4.57 (1H, s, H-29), 4.73 (1H, s, H-29), 9.21 (1H, s, N-OH).
13C NMR (CDCl3, 75.5 MHz): δ 14.5 (C-27), 15.8 (C-25), 15.9 (C-26), 19.3 (C-11), 19.6 (C-30), 20.9 (C-6), 21.6 (C-24), 22.9 (C-23), 25.6 (C-12), 26.5 (C-2), 29.7 (C-15), 31.3 (C-21), 32.4 (C-16), 33.6 (C-22), 34.1 (C-7), 35.9 (C-4), 36.9 (C-13), 38.1 (C-10), 39.6 (C-1), 40.8 (C-8), 41.8 (C-14), 45.5 (C-7′), 45.9 (C-19), 47.3 (C-17), 50.2 (С-18), 52.6 (C-2′, C-6′), 54.3 (C-5′), 54.5 (С-3′), 55.0 (C-9), 55.2 (С-5), 109.1 (C-29), 151.2 (C-20), 167.3 (C-3), 173.4 (C-28).
Synthesis of Compound 3
A solution of compound
3-Oxo-3а-Homo-3а-Aza-17-(4-Methylpiperazin-1-yl)-Carbonylo-Lup-20(29)-ene (3)
Yield 0.36 g, 87%, as a yellow solid.
R f 0.10.
MP: 2270–229°С.
[α]D 20 +12.0° (c 1.00, CHCl3);
Anal. Calcd. for С35Н57N2O2 (Мr 551.86): С 76.18; Н 10.41; N 7.61. Found: С 76.27; Н 10.52; N 7.71.
1H NMR (300 MHz, CDCl3): δ 0.89 (3H, s, H-25), 0.92 (3H, s, H-26), 1.01 (3H, s, H-24), 1.21 (3H, s, H-27), 1.29 (3H, s, H-23), 1.22-2.08 (21Н, m, H-1, H-2, H5-H7, H-9, H11-H13, H-15, H-16, H-18, H-21, H-22), 1.69 (3Н, s, H-30), 2.06-2.18 (4Н, m, H-3′, H-5′), 2.29 (3Н, s, H-7′), 2.43-2.55 (3Н, m, H-13, H-16), 2.68-3.20 (5Н, m, H-19, H-2′, H-6′), 4.59 (1H, s, H-29), 4.71 (1H, s, H-29), 6.13-6.22 (1H, m, NH);
13C NMR (CDCl3, 75.5 MHz): δ 14.4 (C-27), 15.8 (C-25), 15.9 (C-26), 19.5 (C-11), 19.6 (C-30), 20.9 (C-6), 21.6 (C-24), 22.4 (C-23), 25.8 (C-12), 27.1 (C-2), 29.7 (C-15), 30.9 (C-21), 32.2 (C-16), 33.3 (C-22), 33.7 (C-7), 35.9 (C-4), 37.0 (C-13), 37.4 (C-10), 39.1 (C-1), 40.3 (C-8), 40.6 (C-14), 45.4 (C-7′), 45.9 (C-19), 47.3 (C-17), 51.1 (С-18), 52.3 (C-2′, C-6'), 53.7 (C-5'), 54.5 (С-3'), 55.0 (C-9), 56.9 (С-5), 109.7 (С-29), 150.4 (С-20), 173.8 (С-28), 176.7 (C-3).
Synthesis of Compound 4
LiAlH4 (0.456 g; 12 mM) was added to a solution of compound
3-Deoxy-3а-Homo-3а-Aza-17-(4-Methylpiperazin-1-yl)-Carbonylolup-20(29)-ene (4)
Yield 0.46 g, 87%, as a white solid.
Rf 0.08.
MP: 153°С.
[α]D 20 +25° (c 1.00, CHCl3).
Anal. Calcd. for С35Н59N3O (Мr 537.88): C 78.16; Н 11.06; N 7.81. Found: С 78.24; Н 11.14; N 7.92.
1H NMR (300 MHz, CDCl3): δ 0.89 (3H, s, H-25), 0.92 (3H, s, H-26), 0.98 (3H, s, H-24), 1.02 (3H, s, H-27), 1.12 (3H, s, H-23), 1.17-2.08 (22Н, m, H-1, H-2, H5-H7, H-9, H11-H13, H-15, H-16, H-18, H-21, H-22), 1.69 (3Н, s, H-30), 2.29-2.45 (7Н, m, H-13, H-16, H-3′, H-5′), 2.32 (3Н, s, H-7′), 2.51-3.02 (6H, m, H-3, H-19, H-2′, H-6′), 3.03-3.21 (1H, m, NH), 4.54 (1H, s, H-29), 4.68 (1H, s, H-29).
13C NMR (CDCl3, 75.5 MHz): δ 14.5 (C-27), 15.7 (C-25), 16.4 (C-26), 19.1 (C-11), 19.6 (C-30), 20.9 (C-6), 21.3 (C-24), 22.7 (C-23), 25.8 (C-12), 26.9 (C-2), 29.0 (C-15), 31.3 (C-21), 33.5 (C-16), 33.9 (C-22), 34.1 (C-7), 35.9 (C-3), 36.9 (C-13), 38.3 (C-10), 39.4 (C-1), 40.7 (C-8), 41.8 (C-14), 45.5 (C-7′), 45.9 (C-19), 47.3 (C-17), 50.2 (С-18), 52.4 (C-2′, C-6′), 54.3 (C-5′), 54.5 (С-3′), 55.0 (C-9), 56.9 (С-5), 63.2 (С-3), 109.8 (С-29), 150.2 (С-20), 174.1 (С-28).
Minimum Inhibitory Concentration
MIC in LB medium for BW25113 E. coli and BW25113 E. coli ΔtolC (JW5503 from Keio collection) was determined using broth microdilution assay. 22 The cell concentration was adjusted to approximately 5 × 105 cells/mL. The tested compounds were serially diluted twice in a 96-well 2 mL microplate (200 µL per well). Microplates were covered and incubated at 37°C with shaking. The OD600 of each well was measured, and the lowest concentration of the tested compound that resulted in no growth after 16–20 hours was assigned to the MIC value.
Testing the In Vivo Activity of 4
The reporter strain dtolC-pDualrep2 was used as previously described.
19
Briefly, 2 µL of compound
Cytotoxicity Measurements
The measurements were carried out using the standard MTT method. 23 3000 cells per well for PC3, Huh7, and 22Rv1 cell lines or 4000 cells per well for VA13 and LNCAP were plated out in 135 µL of media in 96-well plate. DMEM-F12 media (Gibco) was used for VA13 and Huh7, and RPMI (Gibco) was used for PC3, 22Rv1 and LNCAP cell lines. Media was supplemented with 10% heat-inactivated FBS, 1 × Penstrep, 1 × Glutamax (Gibco). Cells were incubated in the 5% CO2 incubator for first 16 hours without treating. Then 15 µL of media-dimethyl sulfoxide (DMSO) solutions of tested substances to the cells (final DMSO concentrations in the media were 1% or less) and treated cells 72 hours (triplicate each). The MTT reagent then was added to cells up to the final concentration of 0.5 g/L (10× stock solution in phosphate buffered saline was used) and incubated for 2 hours at 37°C in the incubator, under an atmosphere of 5% CO2. The MTT solution was then discarded and 140 µL of DMSO was added. The plates were swayed on a shaker (80 rpm) to solubilize the formazan. The absorbance was measured using a microplate reader (VICTOR X5 Plate Reader) at a wavelength of 565 nm (in order to measure the formazan concentration). The results were used to construct a dose-response graph and to estimate CC50 value by Prism software (GraphPad Software, Inc.).
Footnotes
Acknowledgments
Compounds
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Biological screening of antibacterial activity was supported by grant from the Russian Foundation for Basic Research (project No. 18-34-20055). Cytotoxicity testing of compound
