Abstract
The leaf and twig essential oils of Magnolia hypolampra, growing wild in Na Hang Nature Reserve, Tuyen Quang province of Vietnam, were obtained by hydrodistillation and analyzed by gas chromatography-mass spectrometry. The oil yield calculated on a dry weight basis from leaves of M. hypolampra was very high (1.62%, v/w), while that from twigs was much lower (0.07%, v/w). The essential oils were dominated by monoterpenoids (74.3% and 84.8%) and sesquiterpenoids (24.4% and 13.3%) with β-pinene (36.5% and 41.3%), α-pinene (23.7% and 24.4%), and germacrene D (14.6% and 5.8%) as respective major components. Antibiotic activity of the essential oil samples was tested against Gram-positive bacteria Staphylococcus aureus, Gram-negative bacteria Escherichia coli, and yeast Candida albicans using an agar disk diffusion method. Both the leaf and twig oils showed strong inhibition against all 3 tested microorganism strains with inhibition zones from 18.5 to 30.5 mm and from 45.5 to 46 mm, respectively. Minimum inhibitory concentration of the essential oils was determined using microdilution broth susceptibility assay against 7 test microorganism strains including Bacillus subtilis, Lactobacillus fermentum, Salmonella enterica, Pseudomonas aeruginosa, and 3 abovementioned strains. Minimum inhibitory concentration values of the essential oil from the twigs were from 2.0 to 8.2 mg/mL, while those from the leaves were from 4.1 to 16.4 mg/mL.
Magnolia hypolampra (Dandy) Figlar (syn. Magnolia gioi (A.Chev.) Noot.; Michelia gioi (A.Chev.) Sima & W.H.Chen; Michelia hedyosperma Y.W.Law; Michelia hypolampra Dandy; and Talauma gioi A.Chev. (Vietnamese name is Giổi ăn hăt)) is a timber tree belonging to genus Magnolia L. of family Magnoliaceae. Species of this genus have been the subject of numerous phytochemical, pharmacological, and essential oil investigations over many decades due to their potential use and significant value in traditional health-care systems as well as in fragrance industry. 1 -3 The mature plant of M. hypolampra grows up to 21 m tall and to 60 cm diameter at breast height. 4,5 Buds, young petioles, brachyblasts, flower buds, and carpels appressed short sericeous but the other parts are glabrous. Twigs are black turning pale brown when old, sparsely scattered with lenticels. Leaf blade is obovate to elliptic-obovate, 6-13 × 5-5.5 cm, thinly leathery, both surfaces freshly green, glossy, and glabrous, secondary veins 8 to 10 on each side of midvein and prominent on both surfaces, reticulate veins slender, dense, and prominent on both surfaces, base broadly cuneate, apex with an obtuse tip. The timber of M. hypolampra is a termite-resistant construction material. 6 The subjects of the previous studies on M. hypolampra included the phenylpropanoid glycosides from the seeds, 7 the phylogenetic and biogeographic complexity. 8 The purpose of this work is to characterize the volatile components of leaves and twigs of M. hypolampra from Vietnam and their antimicrobial activity.
By hydrodistillation, essential oil yields of 1.62% (v/w) and 0.07% (v/w), calculated on a dry weight basis, were obtained from the leaves and twigs of M. hypolampra, respectively. Both essential oils were colorless liquids having lower densities than water.
The chemical compositions of the essential oils from leaves and twigs of M. hypolampra from Na Hang Nature Reserve in Vietnam are summarized in Table 1. A total of 40 and 41 compounds were identified in the essential oils, representing 99.2 % and 98.2% of the compositions, respectively. Monoterpenoids (74.3% and 84.8%) and sesquiterpenoids (24.4% and 13.3%) made up the bulk of the essential oil compositions, with α-pinene (23.7% and 24.4%), β-pinene (36.5% and 41.3%), and germacrene D (14.6% and 5.8%) as major components of the leaf oil and the twig oil, respectively. Chemical compositions of essential oils from leaves and twigs of M. hypolampra had a similar pattern in that β-pinene was the most abundant major constituent. In addition, another 29 compounds were also present as the constituents of both oils with varying amounts except 10 constituents present only in leaf oil and 11 constituents were found only in twig oil.
Essential Oils Compositions of the Leaves and Twigs of Magnolia hypolampra From Na Hang Nature Reserve.

RI, retention index.
The chemical compositions of the essential oils obtained from the leaves, trunk, bark, fruit pulp, and fruit kernels of M. hypolampra growing in Vietnam were previously reported by Dũng and coworkers. 9 These authors reported 8 constituents in the leaf oil of M. hypolampra (syn. Talauma gioi) identified with the major components to be elemicin (46.3%), β-caryophyllene (16.9%), α-humulene (6.1%), and (E)-nerolidol (5.6%). They also presented 9 constituents in the trunk oil identified with camphor (23.8%) and β-caryophyllene (5.4%) as the major components. However, many constituents (mainly sesquiterpenes) of the leaf oil (20.6%) and the trunk oil (61.1%) could not be identified in that study. In this present work, we found no evidence for either elemicin, (E)-nerolidol in the leaf essential oil, or camphor in the twig essential oil. While the amounts of β-caryophyllene and α-humulene in leaf oil were, in comparison to the previous data, at lower levels (2.6% and 0.9%, respectively). Similarly, β-caryophyllene content in the twig oil was only 1.1%, which is much lower than the data previously reported. The findings for the leaf oil and twig oil of M. hypolampra in this work were obviously different from the oil in the previous report. The difference in growing location and the sampling time may play an important role in the difference in chemical composition of the essential oil of M. hypolampra. Magnolia hypolampra samples in the research of Dũng and coworker 9 were collected in Yen Bai province in November 1993, while the samples in the present study were collected in Tuyen Quang province in July 2017.
Several investigations on Magnolia essential oil compositions have been reported in the literature, and some examples of the essential oil compositions are listed in Table 2 for comparison. Magnolia calophylla leaf 10 and Magnolia virginiana leaf 10 essential oils have comparable compositions compared with M. hypolampra leaf and twig oils in this study, in that they contain β-pinene as the major constituent. As was the case with M. hypolampra leaf and twig in this present work, most of the Magnolia species examined have monoterpenoids dominating their essential oils. These species include Magnolia acuminata, 10 M. calophylla, 10 M. hypolampra fruit, 11 Magnolia sieboldii, 12 and M. virginiana. 10 Magnolia grandiflora and M. ovata are differently characterized with their essential oils dominated by either monoterpenoids 10,13 or sesquiterpenoids, 14,15 while Magnolia gloriensis (syn. Talauma gloriensis) had sesquiterpenoids dominating its essential oil. 16
Major Components of Some Magnolia Essential Oils.

aThis compound is not a natural product; the identification is doubtful.
Magnolia hypolampra essential oil extracts were used to screen the antimicrobial activity. The standard agar disk diffusion method was performed against 3 test microorganisms. The results of the test were obtained after 18 to 24 hours and the results are presented in Table 3.
Anti-Yeast and Antibacterial Activity of Leaf and Twig Essential Oils of Magnolia hypolampra.

Both of the investigated essential oils of M. hypolampra showed strong inhibition 17,18 against all 3 microorganism strains tested in this study with inhibition zones of more than 14.0 mm. The leaf essential oil was found to be strongly active against Escherichia coli, Staphylococcus aureus, and Candida albicans with inhibitory zone diameters of 18.5 (mm), 30.5, and 27.5 mm, respectively. The twig essential oil showed stronger activity against all 3 microorganisms tested (Table 3).
The essential oil samples that exhibited strong activity against the test strains of microorganisms were then subjected to microbroth dilution assays to determine the minimum inhibitory concentration (MIC) and median inhibitory concentration (IC50) values using 7 strains of microorganisms. The results of the assay were obtained after 16 to 14 hours and the results are presented in Table 4.
Microbial Minimum Inhibitory Concentrations and Median Inhibitory Concentrations of Leaf and Twig Essential Oils of Magnolia hypolampra.

IC50, median inhibitory concentration; MIC, minimum inhibitory concentration.
The essential oil from twigs of M. hypolampra showed stronger inhibitory effects on 7 test microorganisms than that from leaves. Minimum inhibitory concentration values of the twig oil were from 2.0 to 8.2 mg/mL, while those of the leaf oil were from 4.1 to 16.4 mg/mL. IC50 values of the twig and leaf oils ranged from 1.0 to 3.4 mg/mL and from 1.8 to 3.7 mg/mL, respectively. Salmonella enterica and E. coli were more sensitive to the essential oils than the other tested microorganisms (Table 4). Staphylococcus aureus is known to be a bacterium that causes pains, burns, sore throats, and pus infections on the skin and internal organs including infectious endocarditis; B. subtilis is nonpathogenic but it can contaminate food; Lactobacillus fermentum is a “friendly” bacterium in animals and is used for a wide variety of applications that include food and feed fermentation; S. enterica can cause 4 different clinical manifestations: gastroenteritis, bacteremia, enteric fever, and an asymptomatic carrier state; E. coli can cause some gastrointestinal diseases such as gastritis, colitis, enterocolitis, and bacillary dysentery; Pseudomonas aeruginosa is an opportunistic pathogen that can cause urinary tract infections, respiratory system infections, dermatitis, soft tissue infections, bacteremia, bone and joint infections, gastrointestinal infections, and a variety of systemic infections, particularly in patients with severe burns and in cancer and AIDS patients who are immunosuppressed; while C. albicans causes baby thrush in children and gynecological diseases. These assay results can be the basis to open new broader research of the antimicrobial activity of this plant species.
Antimicrobial activities of some Magnolia species have been reported. The essential oil from leaf of M. gloriensis (syn. Talauma gloriensis) that had myrcene (31.7%) and germacrene D (43.5%) as the major components was devoid of antibacterial activity (Bacillus cereus, S. aureus, E. coli, and P. aeruginosa). 16 Volatile oil of twigs, leaves and flowers of M. foveolata (syn. Michelia foveolata) exhibited a significant antibacterial activity against S. enterica, Staphylococcus epidermidis, S. aureus, and B. cereus. 19 Magnolol, honokiol, and 3,5′-diallyl-2′-hydroxy-4-methoxybiphenyl of M. grandiflora exhibited significant activity against Gram-positive and acid-fast bacteria and fungi. 20 The oil from M. grandiflora leaves had a MIC of 500 µg/mL against S. aureus and 125 µg /mL against Streptococcus pyogenes. 21 The essential oil of Magnolia liliflora inhibited growth of test fungi strains with the MIC and minimum fungicidal concentration of the essential oil found in the range of 125 to 500 and 125 to 1000 µg/mL, respectively. 22 The antimicrobial activity of essential oil of M. ovata (syn. Talauma ovata) changed during year. The oil from leaves collected in October was the most active one which inhibited the growth of 19 out of 22 tested microorganisms, whereas the oil from trunk bark collected in January had the highest activity which inhibited the growth of 15 out of 22 tested microorganisms. 23 α-Pinene and β-pinene, the two major components in M. hypolampra leaf and twig essential oils of this study, were previously reported to be able to inhibit significantly the growth and cell viability of potential infectious endocarditis causing Gram-positive bacteria including S. aureus. 24 In another study, only positive enantiomers of α-pinene and β-pinene exhibited antimicrobial activity against the fungi and bacteria tested. 25 Some studies reported on the positive effects of α-pinene such as inhaling α-pinene caused significant anxiolytic-like activity in mice, 26 anti-tumor on human hepatoma cell lines in vitro and in vivo, 27 and olfactory stimulation by α-pinene induced physiological relaxation. 28 Another study reported that β-pinene showed antidepressant-like and sedative-like activities in mice. 29 The anxiolytic-like, anti-tumor, and physiological relaxation effect of α-pinene and β-pinene as well as antimicrobial activity and fragrance of M. hypolampra leaf and twig essential oils suggests that they have potential for use in health-care and fragrance fields.
Experimental
Plant Material
Leaves and twigs of M. hypolampra growing wild in Na Hang Nature Reserve in Thanh Tuong commune, Na Hang district, Tuyen Quang province, North of Vietnam were collected in July 2017. The plant was identified by Dr Tien Hiep Nguyen. Voucher specimen (TQ1702) was deposited at the Herbarium of Institute of Ecology and Biological Resources (HN), Vietnam Academy of Science and Technology. In total, 0.63 kg and 0.93 kg samples of the fresh leaf and twig materials, respectively, were shredded and hydrodistilled for 3 hours using a Clevenger type apparatus. After that, the essential oils were separated and dried with anhydrous MgSO4. The obtained oils were stored at −5°C until analysis.
Gas Chromatography-Mass Spectrometry
Analysis of the essential oils was carried out by gas chromatography-mass spectrometry (GC-MS) using an Agilent GC7890A system with Mass Selective Detector (Agilent 5975C). A HP-5MS fused silica capillary column (60 m × 0.25 mm i.d. × 0.25 µm film thickness) was used. Helium was the carrier gas with a flow rate of 1.0 mL/min. The inlet temperature was 250°C and the oven temperature program was as follows: 60°C to 240°C at 4 °C/min with an interphase temperature of 270°C. The split ratio was 1:100, the detector temperature was 270°C, and the injection volume was 1 µL. The MS interface temperature was 270°C, MS mode, E.I. detector voltage 1200 V, and mass range 35 to 450 Da at 1.0 scan/s. Identification of components was achieved based on their retention indices and by comparison of their mass spectral fragmentation patterns with those stored on the MS library (HPCH1607, NIST08, and Wiley09). Component relative contents were calculated based on total ion current without standardization. Data processing software was MassFinder4.0.
Microbial Strains
The antimicrobial activity of the essential oils was evaluated using 1 strain of Gram-positive test bacteria S. aureus (ATCC 13709), 1 strain of Gram-negative test bacteria E. coli (ATCC 25922), and 1 strain of yeast C. albicans (ATCC 10231). Minimum inhibitory concentration and median inhibitory concentration (IC50) values were determined using 3 strains of Gram-positive test bacteria including S. aureus (ATCC 13709), B. subtilis (ATCC 6633), and L. fermentum (VTCC N4), 3 strains of Gram-negative test bacteria including S. enterica (VTCC), E. coli (ATCC 25922), and P. aeruginosa (ATCC 15442), and 1 strain of yeast C. albicans (ATCC 10231). The ATCC strains were obtained from American Type Culture Collection. The VTCC strains were obtained from Vietnam Type Culture Collection—Vietnam National University, Hanoi.
Screening of Antimicrobial Activity
The agar disk diffusion method was performed to test the antimicrobial activity of essential oil. 30 -32 Testing media included Mueller-Hinton Agar used for bacteria and Sabouraud Agar used for fungi. Microorganisms were stored at −80°C and activated by culture medium prior to testing to reach concentration of 1.0 × 106 CFU/mL. A 100 µL inoculum solution was taken and spread evenly over the surface of the agar. Two holes were made on agar plates (about 6 mm in diameter each hole) using an aseptic technique. A total of 50 µL essential oil was put into each hole using a pipette. The petri dishes were kept at room temperature for 2 to 4 hours and then incubated at 37°C for 18 to 24 hours. The presence or absence of growth around each antimicrobial disk on each plate culture was observed. The diameters of inhibition growth zones values were measured using a ruler with millimeter markings. The zone of inhibition is the point at which no growth is visible to the unaided eye. An inhibition zone of 14 mm or greater (including diameter of the hole) was considered as high antibacterial activity. 17,18 Minimum inhibitory concentration and median inhibitory concentration (IC50) values were measured by the microdilution broth susceptibility assay. 33,34 Stock solutions of the oil were prepared in dimethylsulfoxide. Dilution series were prepared from 16 384 to 2 μg/mL (214, 213, 212, 211, 210, 29, 27, 25, 23, and 21 µg/mL) in sterile distilled water in micro-test tubes from where they were transferred to 96-well microtiter plates. Bacteria grown in double-strength Mueller-Hinton broth or double-strength tryptic soy broth, and fungi grown in double-strength Sabouraud dextrose broth were standardized to 5 × 105 and 1 × 103 CFU/mL, respectively. The last row, containing only the serial dilutions of sample without microorganisms, was used as a negative control. Sterile distilled water and medium served as a positive control. After incubation at 37°C for 24 hours, the MIC values were determined at well with the lowest concentration of agents completely inhibiting the growth of microorganisms. The IC50 values were determined by the percentage of microorganisms inhibited growth based on the turbidity measurement data of EPOCH2C spectrophotometer (BioTeK Instruments, Inc Highland Park Winooski, United States) and Rawdata computer software (Belgium) according to the following equations:
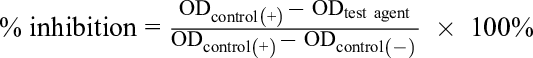

where OD is the optical density, control (+) is the only cells in medium without antimicrobial agent, test agent corresponds to a known concentration of antimicrobial agent, control (−) is the culture medium without cells, HighConc/LowConc is the concentration of test agent at high concentration/low concentration, and HighInh%/LowInh% is the % inhibition at high concentration/% inhibition at low concentration).
Reference materials: Ampicillin for Gram-positive bacterial strains with MIC values in the range of 0.004 to 1.2 µg/mL, Cefotaxime for Gram-negative bacterial strains with MIC values in the range of 0.07 to 19.23 µg/mL, and Nystatine for fungal strains with MIC value of about 2.8 to 5.0 µg/mL.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was financially supported by Vietnam Academy of Science and Technology under Grant number VAST.ĐTCB.02/17-19 (UQĐTCB.02/17-19).
