Abstract
The formation of amyloid fibrils is associated with many human illnesses, such as Alzheimer’s, Huntington’s, and Parkinson’s diseases, amyotrophic lateral sclerosis, spongiform encephalitis, type 2 diabetes, and primary and secondary systemic amyloidosis. Nutrition contributes to the prevention of these diseases. The aim of our work was to look for commercially available fruit juices that can inhibit the formation of amyloid fibrils. Of the fruit juices that we examined, that of pomegranate was found to be the most effective inhibitory agent using turbidity measurements and Congo red binding assay. According to our experiments, pomegranate juice reduced the amount of PMS-trypsin amyloid-like fibrils to 3.7% at 5-fold dilution compared with the sample without pomegranate. The inhibitory effect of the pomegranate juice was concentration dependent.
The formation of amyloid fibrils is associated with a great variety of human illnesses, such as Alzheimer’s (AD), Huntington’s, and Parkinson’s diseases, amyotrophic lateral sclerosis, spongiform encephalitis, type 2 diabetes, and primary and secondary systemic amyloidosis. 1,2 The insoluble amyloid is structurally dominated by a β-sheet structure. Research on the physicochemical properties and formation of amyloid is currently under way. 3,4 AD is a destructive neurodegenerative disease. Seventeen percent of 75- to 84-year-olds have AD. 5,6 The rate of type 2 diabetes was 8.8% among adults in 2017, but is expected to rise to 9.9% of the population between the ages of 20 and 79 by 2045. 7 Understanding the mechanism of amyloid fibril formation and elaborating a new approach to fibrillation inhibition is the key to the prevention of the aforementioned amyloid diseases. The results show that stabilizing the native state of the proteins prevents amyloidogenic conformational changes, which often appear in human amyloid diseases. 8 -12
Many plant compounds are known as effective anti-amyloid aggregation agents. Numerous epidemiological studies have shown the relationship between polyphenol-rich foods and human health. 13 The lower toxicity of natural compounds compared with many synthetic chemical molecules suggests that they can serve as a good starting point to discover protein misfolding inhibitors, which may be useful in the treatment of various incurable diseases. 14 Plants are not characterized by the presence of amyloid fibrils, suggesting that plants have a special mechanism against protein misfolding. 15 Currently there are more than 8 000 known plant polyphenols. 16 Polyphenols are structurally characterized by 2 or more hydroxyl groups attached to 1 or more benzene rings. 17 Many polyphenolic compounds have been shown to inhibit significantly the formation of amyloid fibrils in vitro, 18,19 because the aromatic polyphenolic rings of polyphenols and proteins may associate via π-π stacking interactions with each other. 20 They also block the self-assembly process of the protein. 21 Pomegranate (Punica granatum L.) fruit is a rich source of various polyphenols; 37 different ones have been identified in 3 pomegranate samples, 22 especially the ellagitannin, punicalagin, which is the most common polyphenol in it. Pomegranate peel also contains gallic acid, ellagic acid, pelargonidin, cyanidin, delphinidin, kaempferol, quercetin, and luteolin. Experimental results support the specific anti-amyloidogenic effect of the pomegranate extract. 23 -25 Punicalagin has been shown to have anti-amyloidogenic activity. 26 It has been found that pomegranate consumption is a potential nutrition strategy to slow the progression of AD. 27 It has been shown that long-term supplementation with pomegranate may weaken AD pathology by reducing inflammation and altering the β-amyloid precursor protein-dependent processes in a transgenic mice model of AD. 28 The pomegranate extract changed the level and ratio of Aβ42 and Aβ40 peptides, which had a positive effect on the reduction of AD pathogenesis. 24 Urolithins are the possible brain absorbable molecules that contribute to the anti-AD effects of pomegranate. Punicalagin is hydrolyzed to release ellagic acid and then biotransformed by gut microbiota to yield urolithins. Urolithins prevented β-amyloid fibrillation in vitro. 29
In this study, the anti-amyloidogenic effects of various fruit juices were investigated. We systematically screened some different commercially available fruit juices that can inhibit PMS-trypsin aggregation using turbidity measurements. Pomegranate juice proved to be the best inhibitory agent. To assess the effect of the pomegranate juice on the growth phase of amyloid fibril formation, turbidity measurements and Congo red (CR) binding assay were performed. For our experiments, trypsin chemically modified with phenylmethyl-sulfonyl fluoride was used as a model protein. The chemically modified enzyme has no catalytic activity, and so autolysis does not affect fibril formation. PMS-trypsin amyloid fibrils were prepared according to Kasi et al by incubation of the soluble protein in the presence of 60% ethanol at 24°C at pH 7.0. 30 The model system we used was not a physiological one, but it was suitable to detect the effectiveness of various inhibitory agents. Amyloid fibrils can be derived not only from amyloidogenic proteins, but from any protein under appropriate mild denaturing conditions. 31 Our results showed that pomegranate juice was able to inhibit effectively PMS-trypsin amyloid-like fibril formation in vitro.
Amyloid growth is usually detected by measuring solution turbidity. 32,33 An increase in the absorption at 350 nm indicates a greater degree of aggregation. 34 The sample without fruit juice showed maximum absorption whereas in the presence of different juices it was significantly reduced. All the juices we used inhibited aggregation, but their levels of effectiveness were different. Among the commercially available fruit juices, that of pomegranate was found to be the most effective from this comparative study (Figure 1). At 5-fold dilution, pomegranate juice significantly inhibited the fibril formation by reducing the amount of fibrils to 3.7% compared with the sample without pomegranate.
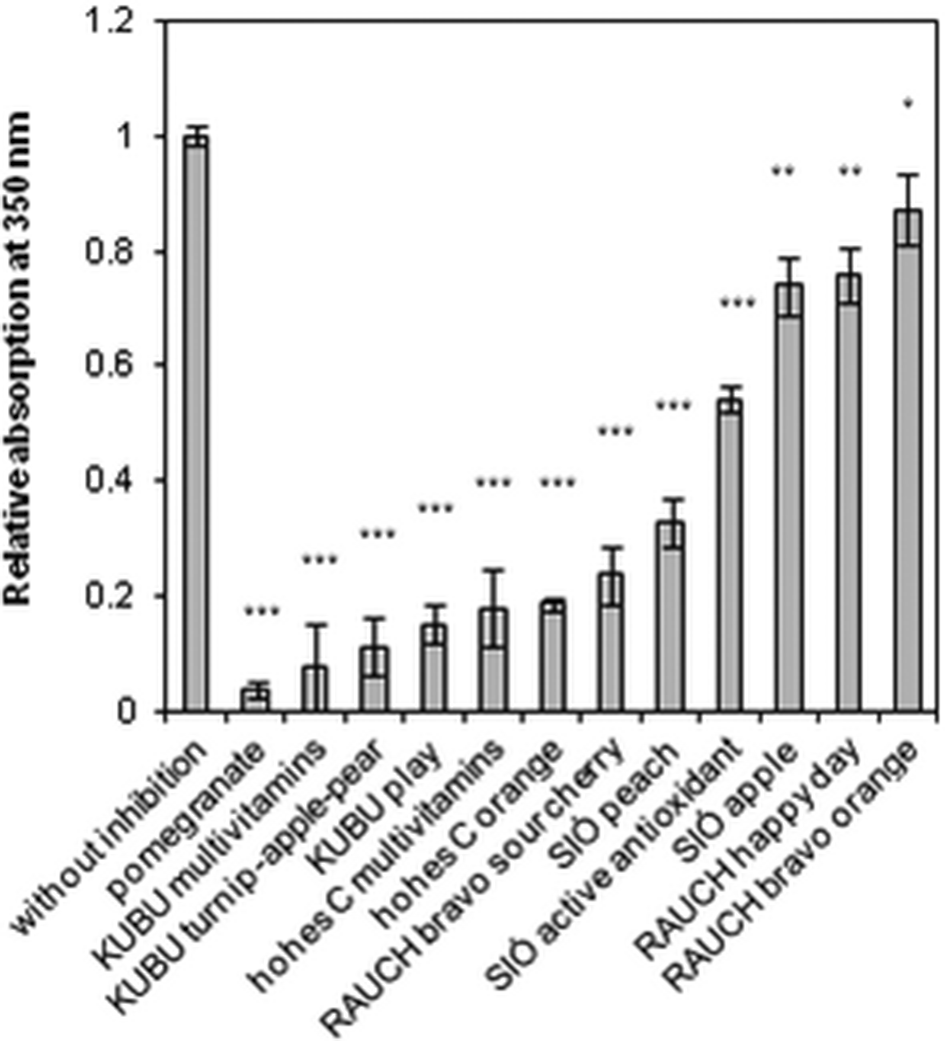
Turbidity measurements of different juices by recording the absorption at 350 nm. PMS-trypsin (0.13 mg/mL) was incubated in 60% ethanol in the absence or presence of different juices diluted 5 times at pH 7.0. Each bar represents the average of at least 3 independent measurements. All data are presented as mean ± standard error of the mean. Significance was defined as ***P < 0.001, **P < 0.01, and *P < 0.05.
The inhibitory effect of the pomegranate juice on PMS-trypsin fibrillation was also determined at different concentrations. It was found that amyloid fibril formation of PMS-trypsin diminishes in the presence of various dilutions of pomegranate juice. Turbidity measurements showed its concentration-dependent inhibitory effect (Figure 2). Pomegranate juice diluted 250 times reduced the absorption at 350 nm to 63.4% compared with the sample that contained no inhibitory agent. It also inhibited the formation of fibrils at 500-fold dilution.

Turbidity at 350 nm of PMS-trypsin in 60% ethanol in the absence and presence of different concentrations of pomegranate juice. Protein concentration: 0.13 mg/mL. All data are presented as mean ± standard error of the mean from 3 independent measurements. Significance was defined as ***P < 0.001 and **P < 0.01.
CR staining is a qualitative method to identify amyloids. 35 To determine the anti-amyloidogenic activity of the pomegranate juice against PMS-trypsin fibrillation, the CR binding assay was used. The presence of amyloid-like fibrils causes an increase in the value of absorption and a spectral shift in the peak wavelength of CR. 36,37 Difference spectra indicate spectral changes of CR upon binding to PMS-trypsin amyloid-like fibrils in the absence or presence of 100-fold diluted pomegranate juice. In the presence of the pomegranate juice, the maximum of CR difference spectrum at 550 nm was reduced significantly. The CR binding assay also demonstrated the inhibitory effect of the pomegranate juice on fibril formation (Figure 3).
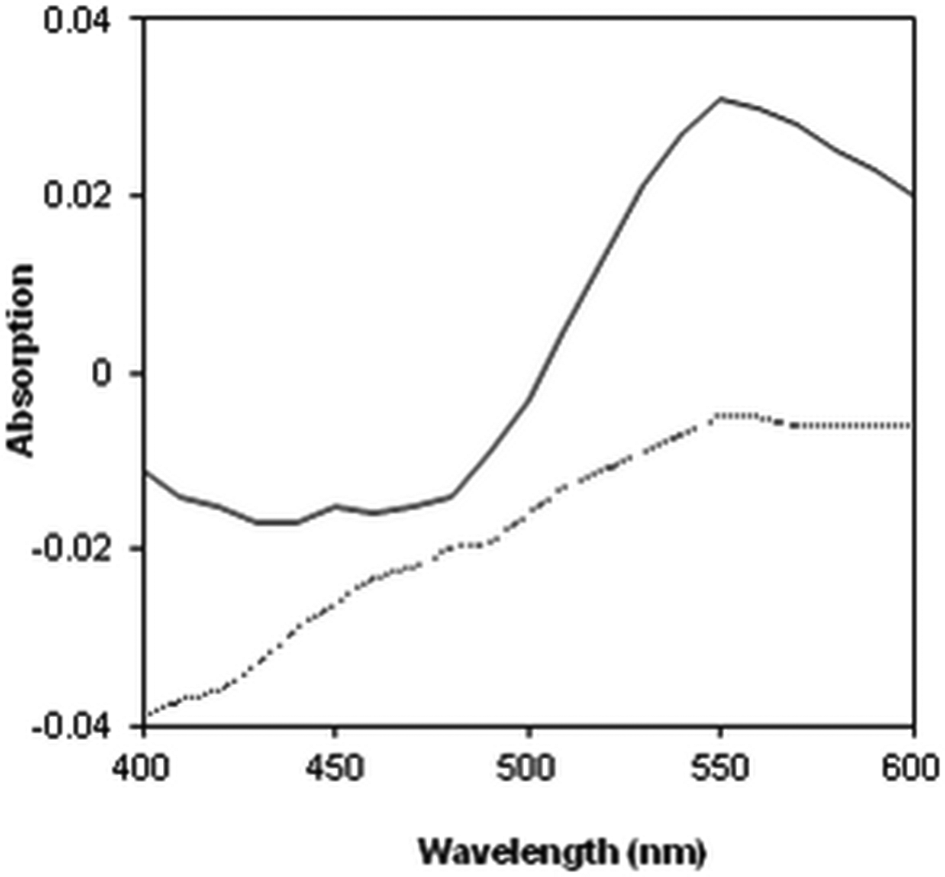
Congo red absorption difference spectra of PMS-trypsin in the absence (solid line) and presence of pomegranate juice diluted 100 times (dotted line).
According to our experimental findings, pomegranate juice significantly inhibits PMS-trypsin amyloid-like fibril formation, so is a very promising candidate for the effective prevention of amyloid-related diseases.
Experimental
General Experimental Procedures
Absorption of our samples was measured at 350 nm on a Cecil CE-5501 UV-vis spectrophotometer in a cuvette with a 1 cm path length. The CR absorption spectra were taken with a Hitachi U 2000 spectrophotometer.
Materials
Bovine pancreas trypsin (EC 3.4.21.4) was purchased from Sigma-Aldrich Ltd. (Budapest, Hungary). The bio pomegranate juice (100%) was the product of Jacoby Ltd. (Auggen, Germany), KUBU multivitamins (51%), KUBU turnip-apple-pear (57%), and KUBU play (15%) juices of Maspex Olympos Ltd. (Nyárlőrinc, Hungary), hohes C multivitamis (100%), hohes C orange (100%), SIÓ peach (25%), SIÓ active antioxidant (40%), and SIÓ apple (100%) juices of Sió-Eckes Ltd. (Siófok, Hungary), RAUCH bravo sour cherry (12%), RAUCH happy day (100%), and RAUCH bravo orange (12%) juices of Rauch Hungaria Ltd. (Budapest, Hungary). All other reagents and buffer components used were of analytical grade.
Turbidity Measurements
The turbidity measurements were performed to detect the effects of different fruit juices on the aggregation of the PMS-trypsin. The absorption was read at 350 nm using a 1 cm quartz cuvette after 1 day incubation in 10 mM phosphate buffer at pH 7.0 in the presence and absence of different fruit juices as an indication of the degree of aggregation. These measurements were performed at 0.13 mg/mL PMS-trypsin concentration in 60% ethanol.
Congo Red Binding Assay
The CR binding assay was used to detect the presence of amyloid fibrils. 38 CR (disodium-3,3′[[1,1-biphenyl]-4,4′-diylbis(azo)]bis(4-amino-naphthalin-1-sulfonate)) absorption spectra were recorded from 400 to 600 nm using a 1 cm path length quartz cuvette. One-day-aged PMS-trypsin samples (200 µL) were mixed with 800 µL CR solution. CR binding experiments were performed in 5 mM phosphate buffer (pH 7.0) containing 150 mM NaCl. The samples were incubated for 15 minutes before the measurements. CR difference spectra were obtained by the subtraction of the spectra of CR alone and PMS-trypsin alone from the spectra of CR and PMS-trypsin. 39
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by project EFOP-3.6.1–16-2016-00008.
