Abstract
Three new compounds, phomanones A-C (
Mariana Trench, located in the western Pacific Ocean, is known as the deepest trench in the world, extending to a depth of approximately 10 920 m, corresponding to about 110 MPa of hydrostatic pressure. 1 -3 Due to the unique geographical location and environment, it is regarded to be a unique ecosystem, which is the habitat for deep-sea microorganisms with the ability to produce structurally diversified secondary metabolites. 4
Within our ongoing work of searching for new metabolites from deep-sea derived fungi,
5
-7
a Phoma sp. strain (HDN16-618) was obtained from a seawater sample collected from the Mariana Trench at a depth of 4000 m. Further chemical studies of this strain led to the isolation of 4 polyketides (

Structures of compounds 1 to
The Phoma sp. was incubated for 60 days in a rice medium in Erlenmeyer flasks under static conditions. The whole (about 40 L) culture was then extracted with MeOH and the organic extract (28.0 g) fractionated by repeated column chromatography (CC) using silica gel and High Performance Liquid Chromatography (HPLC) octadecyl silane (ODS) columns leading to the isolation of compounds
Phomanone A (
1H (500 MHz) and 13C (125 MHz) NMR Data for Compound 1 in DMSO-d 6 .

1H (500 MHz) and 13C (125 MHz) NMR Data for Compounds 2 to


Key 1H-1H COSY and HMBC correlation of compounds 1 to
To determine the absolute configuration, the solution conformers and electronic circular dichroism (ECD) spectra of (4R)

Experimental electronic circular dichroism spectrum of 1 (black curve), and calculated electronic circular dichroism spectrum of truncated models 1a (blue curve) and 1b (red curve).
Phomanone B (
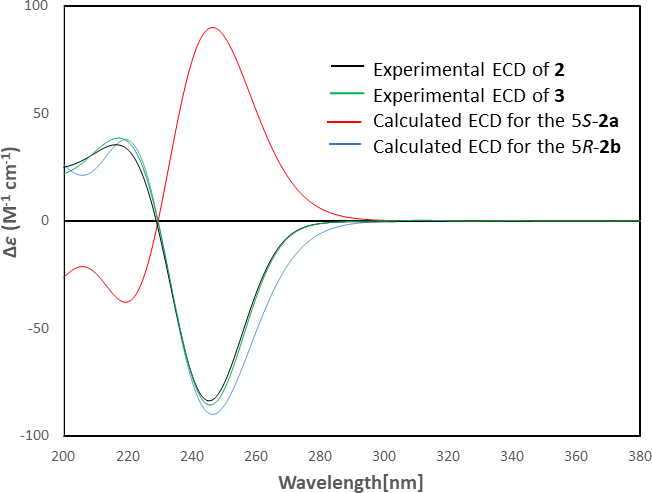
Experimental electronic circular dichroism spectra of 2 (black curve) and 3 (green curve), and calculated electronic circular dichroism spectrum of truncated models 2a (red curve) and 2b (blue curve).
Phomanone C (
All new compounds (
In conclusion, 3 new compounds phomanones A-C (
Experimental
General
Specific rotations were obtained on a JASCO P-1020 digital polarimeter. UV spectra were recorded on a HITACHI 5430, CD spectra on a JASCO J-715 spectropolarimeter, and NMR spectra on an Agilent 500 MHz DD2 spectrometer (Agilent, Beijing, China) using tetramethylsilane as an internal standard; chemical shifts were recorded as δ values. HRESIMS were obtained using a Thermo Scientific LTQ Orbitrap XL mass spectrometer. Semipreparative HPLC was performed using an ODS column (HPLC [YMC-Pack ODS-A, 10 × 250 mm, 5 µm, 3 mL/min]).
Medium-pressure preparative liquid chromatography was conducted on a Bona-Agela CHEETAH HP100 (Beijing Agela Technologies Co., Ltd.). For CC, silica gel (100-200 mesh, 200-300 mesh, Qingdao Marine Chemical Inc.) and Sephadex LH-20 (Amersham Biosciences) columns were used.
Fungal Material
The strain HDN16-618 was isolated from a seawater sample (142°25.823′E, 11°21.136′N) collected at a depth of 4000 m from the Mariana Trench. The strain was identified by Internal Transcribed Spacers sequence and the sequence data have been submitted to GenBank (accession number: HQ914816). A voucher specimen is deposited in our laboratory at −20°C and its working stocks were prepared on potato dextrose agar slants stored at 4°C.
Fermentation, Extraction, and Isolation
Strain HDN16-618 was cultured on a PDA plate at 28°C for 5 days. The fresh mycelia and spores were inoculated into 1000 mL Erlenmeyer flasks (×10) each containing 80 g of rice and 120 mL of sea water to perform large-scale fermentation. The cultures were incubated statically at 28°C for 60 days.
The fermented broth was then extracted first with MeOH 3 times, and then with EtOAc 3 times. The organic solvent was evaporated under reduced pressure to afford an organic extract (32.0 g), which was partitioned with 90% MeOH in H2O and then extracted with light petroleum (PE) 4 times. The MeOH layer was concentrated to provide a defatted extract (28.0 g), which was chromatographed over silica gel using gradient elution with PE-DCM-MeOH (1:1:0→ 0:5:1) to obtain 8 fractions (Fr.1-Fr.8). Fr.4 (2.1 g) was subjected to CC on ODS H2O-MeOH (20%→100%) to obtain 8 fractions (Fr.4-1-Fr.4-8). Fr.4-5, followed by repeated CC over silica gel using a gradient of DCM-MeOH (50:0 → 5:1), yielded subfraction Fr.4-5-6. Final purification by preparative HPLC (PHPLC, H2O-MeOH, 35%) yielded
Compound 1
Colorless oil.
IR (MeOH) ν max: 3397, 2917, 1695, 1639, 1429, 1384, 1205, 1176, 1003, 676, cm−1;
1H and 13C NMR: Table 1.
HRESIMS: m/z 173.0628 [M+H]+ (calcd for C8H12O2S, 173.0631).
Compound 2
Colorless oil.
IR (MeOH) ν max: 2956, 2357, 1728, 1632, 1211, 841, 776 cm−1.
1H and 13C NMR: Table 1.
HRESIMS: m/z 201.0756 [M+H]+ (calcd for C9H13O5, 201.0757).
Compound 3
Colorless crystals.
IR (MeOH) ν max: 3096, 2933, 1715, 1621, 1470, 1380, 1090, 868, 784, 638 cm−1;
1H and 13C NMR: Table 1.
HRESIMS: m/z 215.0911 [M+H]+ (calcd for C10H15O5, 215.0914).
Computation Section
Conformational searches were run by employing the “systematic” procedure implemented in Spartan’14 using the MMFF. All MMFF minima were reoptimized with DFT calculations at the B3LYP/6-31+G(d) level using the Gaussian09 program. The geometry was optimized starting from various initial conformations, with vibrational frequency calculations confirming the presence of minima.
13,14
Time-dependent DFT calculations were performed on the 8 lowest-energy conformations for (4R)
Footnotes
Acknowledgment
The authors acknowledge the support of the Research Vessel KEXUE of the National Major Science and Technology Infrastructure from the Chinese Academy of Sciences.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Key R&D Program of China (2018YFC0310800), the Fundamental Research Funds for the Central Universities (201941001), Marine S&T Fund of Shandong Province for Pilot National Laboratory for Marine Science and Technology(Qingdao) (No. 2018SDKJ0401-2), Global Change and Air-Sea Interaction Project (Grant GASI-IPOVAI-01-03), NSFC-Shandong Joint Fund for Marine Science Research Centers (U1606403), and Taishan Scholar Youth Expert Program in Shandong Province (No. tsqn201812021).
