Abstract
Primycin is a 36-membered marginolactone antibiotic that is biosynthesized through the modular type I polyketide synthase pathway produced by
Due to the accelerated spread of drug-resistant pathogenic bacteria,
1
diverse groups of bioactive natural products, especially antibiotics originating from rare Actinomycetes,
2
have become an essential source of drug research and strain development. Primycin as an active substance of Ebrimycin gel
3
is a marginolactone antibiotic complex, which was described for the first time in Nature by Vályi-Nagy et al.
4
As a topical gel formulation primycin was shown to be an effective treatment against skin infections, including acne or pyoderma,
5
and was successfully applied against small burns infected by Gram-positive bacteria.
6
Recent in vitro studies demonstrated that primycin has high antimicrobial activity against the most frequent Gram-positive pathogens including clinically important multiresistant strains
7
and fungi.
8,9
Although most of the marginolactone antibiotics act on the ribosome,
10,11
novel investigations revealed that primycin mainly acts by the disorganization of cell membranes.
12
-14
Primycin, similar to other guanidino marginolactone antibiotics,
15,16
is biosynthesized via modular type I polyketide synthase (PKS) multienzymes in which the assembly mechanism is closely related to fatty acid (FA) synthesis.
17,18
Through the synthesis of such structurally complex natural products the PKS assembly line generally utilizes malonyl- and methylmalonyl-CoA building blocks,
19
while during the synthesis of the
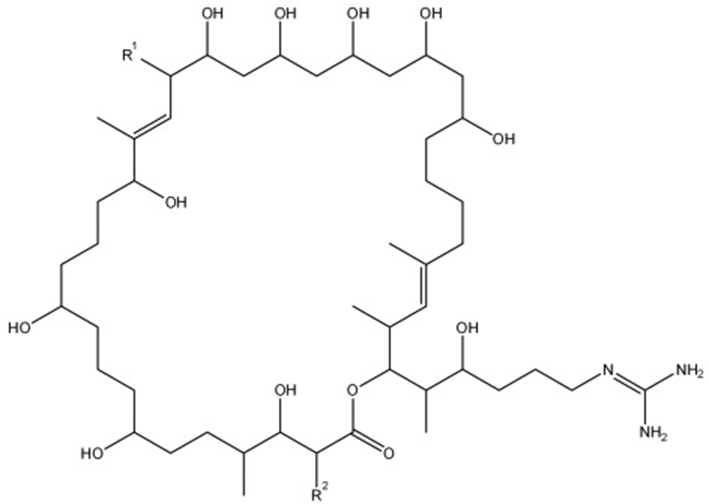
General structure of primycin molecule. R1 side chain represents -arabinosyl, -H, or -OH functional group while R2 side chain represents
Beside the directly added stearic acid, sunflower oil (1%-7% stearic acid and 4%-9% palmitic acid)
22
is also present in the primycin fermentation media, which may contribute to antibiotic synthesis. However, preliminary experiments have confirmed that sunflower oil has only slight effect on primycin production, when FA is present. Although plant oils are often added into industrial-scale fermentation media as antibiotic synthesis precursors,
23
only limited data are available about fermentations where directly added FAs are utilized to enhance antibiotic production. Lee et al.
24
found that the saturated LCFAs (palmitic acid) induced the highest synthesis of tylosin, a macrolide antibiotic produced by
In this present study various FA compositions of the primycin fermentation media were investigated in order to determine their effect on antibiotic production. As a first step of our FA experiments the original 3 g/L stearic acid-containing fermentation media 30 was compared with FA-free media and with 3 g/L palmitic acid-containing fermentation media in order to determine if a slight modification in FA composition—changing the 18 carbon length to 16 carbon—in the growth medium has any beneficial effect on primycin production. During fermentation primycin concentrations were measured from the fifth day till the seventh day by the high-performance liquid chromatography-diode array detector-mass spectrometry detection (HPLC-DAD-MSD) method (Figure 2).

Primycin concentrations in fermentation media (containing 3 g/L fatty acid) at different fermentation stages (days 5, 6, and 7).
During this period we could observe a constantly increasing primycin concentration at both FA-containing fermentation media. However, primycin concentration induced by palmitic acid exceeded that one induced by stearic acid each day. The highest primycin concentration (1583.63 mg/L) was measured at the seventh day of fermentation in the presence of palmitic acid, which represents a 14% increase compared to the stearic acid-induced yield (1395.09 mg/L). In contrast, the FA-free fermentation media induced significantly lower primycin production (470.91 mg/L), which means 34% of loss when compared to efficiency of stearic acid-containing media. On one hand, our results showed that the lack of stearic acid significantly decreases the primycin production. On the other hand, results indicated that substitution of stearic acid to the shorter, 16-carbon-containing palmitic acid evokes higher primycin concentration in the fermentation media.
To widen further the set of FAs on primycin production, 7 frequently used FAs (namely, stearic acid, palmitic acid, lauric acid, capric acid, enanthic acid, caproic acid, and butyric acid) were selected for screening. Among them palmitic acid induced the highest primycin concentration at the end of the 1-week fermentation period followed by the stearic acid-containing (original) fermentation media (Table 1). While stearic acid and palmitic acid caused the highest primycin concentration at the seventh day, in the presence of lauric acid the highest primycin concentration was measured at the fifth day, which was followed by a continuous concentration decrease till the end of the fermentation. During the fermentation period no significant differences were found between ratios of the individual primycin components (A1/C1) regarding the different FAs. Primycin A1 component varied between 80% and 90% while Primycin C1 component was between 10% and 20% in all cases.
Primycin Concentrations in Various Fatty Acid-Containing Fermentation Media at Different Fermentation Stages (5-7 Days).

aValues are reported as means ± standard deviation of 3 independent experiments (
These results are contrary to those earlier findings where lauric acid possessed the strongest antimicrobial activity against Gram-positive bacteria
31,32
by separating the inner and outer membranes of the cell resulting in cytoplasmic disorganization.
33
As it was expected, capric acid and enanthic acid did not induce antibiotic production, but complete inhibition of cell growth could be observed, due to the fact that middle-chain FAs have potential antimicrobial activity against various Gram-positive and Gram-negative bacteria.
34,35
It is generally assumed that undissociated forms of FAs can easily penetrate the phospholipid bilayer of the cell membrane and be transferred into the cytosol where these weak carboxylic acids start to dissociate into their ions. To maintain the optimal pH (nearly neutral) the cell is forced to remove excess protons, which results in exhausting of energy resources by utilization of adenosine triphosphate.
36
However, this phenomenon cannot explain the behavior of capric acid and enanthic acid, because their p
FAs with higher primycin-producing ability were selected and proceed for further investigation. Using the same methodology and conditions as for the initial screening of the primycin fermentation media, different FA concentrations (3, 4.5, and 6 g/L) were tested. As shown in Figure 3, the highest primycin concentration could be observed in the presence of palmitic acid for all tested concentrations. When comparing the three tested FA concentrations, 4.5 g/L treatment proved to be the most efficient regardless the type of the FA. In the case of palmitic acid this resulted in a maximum primycin concentration of 2177.19 mg/L measured on the sixth day, while stearic acid induced a much lower, 1388.87 mg/L primycin concentration.

Primycin concentrations in fermentation media with 3 g/L (a), 4.5 g/L (b), and 6 g/L (c) concentrations of various fatty acids at different fermentation stages (3-7 days).
Regarding fermentation time it can be stated that in most cases the antibiotic production started slowly around the second day of fermentation and increased rapidly to reach the maximum around the sixth day. A tendency of slightly decreasing antibiotic production could be observed during the following days, except in the case of 4.5 and 6 g/L lauric acid treatments where the primycin concentration started to decrease on the sixth day after reaching the maximum value (1076.70 mg/L) on the fifth day.
Based on our observations primycin synthesis is highly dependent on the FA composition of the fermentation media and can vary according to the applied FA concentration. In primycin fermentation media the easily accessible water-soluble starch is the primary carbon source for cell growth. It is well known that in aqueous media the solubility of FAs decreases according to the chain length
37
; therefore, slightly soluble or insoluble LCFAs are hardly utilized during normal growth. With depletion of starch the microbes are forced to turn to hardly accessible carbon sources like LCFAs during their secondary metabolism processes including antibiotic synthesis. To improve primycin production through optimization of FA composition we found palmitic acid a better alternative than the original substrate stearic acid. Namely, the 4.5 g/L palmitic acid treatment proved to be the most efficient resulting in 1.7-fold higher primycin content when compared to the original 3 g/L stearic acid composition. Although palmitic acid induced better primycin production than stearic acid in all conditions (including numerous preliminary researches), the difference may vary between 15% and 75% depending on the applied inoculum (cell line, strain age, number of passages, and so on). Palmitic acid plays an important role in the primycin producer bacteria’s cell cycle, which is underlined by the fact that the composition of major cellular FAs found in a close relative
Experimental
Bacterial Strain
The primycin-producing
Culture Conditions
Culture conditions were carried out according to
HPLC-DAD-MSD Analysis
For HPLC-DAD-MSD analysis 0.5 mL sample was taken at different fermentation stages directly from the fermentation media and diluted with 4.5 mL BEW buffer (Butanol-Ethanol-Water 1:1:2). The mixture was vortexed thoroughly for 1 minute, then centrifuged at 4 000 rpm for 10 minutes (Hettich Universal 32). The supernatant was filtered through a 17 mm HPLC syringe filter (PTFE, pore size 0.45 µm, APG Europe) before HPLC-DAD-MSD analysis. As a reference solution was used primycin-sulfate standard in BEW buffer with 0.05, 0.10, and 0.20 mg/mL concentrations. The measurements were conducted with a Poroshell 120 SB C-18 (2.7 µm, 4.6 × 100 mm, Agilent) column. Mobile phase consisted of 60% 10 mM NH4OAc buffer (pH = 5.0; HCOOH) and 40% acetonitrile at a flow rate of 0.5 mL/min. DAD detector measured at 210 nm, mass spectrometer was scanning in the positive ionization mode from
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for theresearch, authorship, and/or publication of this article: This work was supported by PannonPharma Pharmaceutical Ltd.
