Abstract
Synthesis of phenanthroindolizidine core was efficiently achieved through a pathway involving hetero Diels-Alder reaction of α-allenylchalcogenoketenes, generated in situ by thermal [3,3] sigmatropic rearrangement of alkynyl propargyl sulfides or selenides, with cyclic imines and the subsequent iodine-assisted photochemical cyclization.
Keywords
Tylophorine (
In the course of our research work on the synthesis of chalcogen-containing heteroaromatic compounds by using the reactivity of chalcogenocarbonyl functionalities, we have previously reported a conversion of alkynyl propargyl chalcogenides into quinolizidine and indolizidine rings via α-allenylchalcogenoketenes by using a sequential [3,3] sigmatropic rearrangement and the subsequent hetero Diels-Alder reaction with cyclic imines.
40
-45
These successful results urged us to the new synthesis of polysubstituted fused-indolizidine skeletons, i
e, phenanthroindolizine alkaloid cores. It is expected that these target compounds
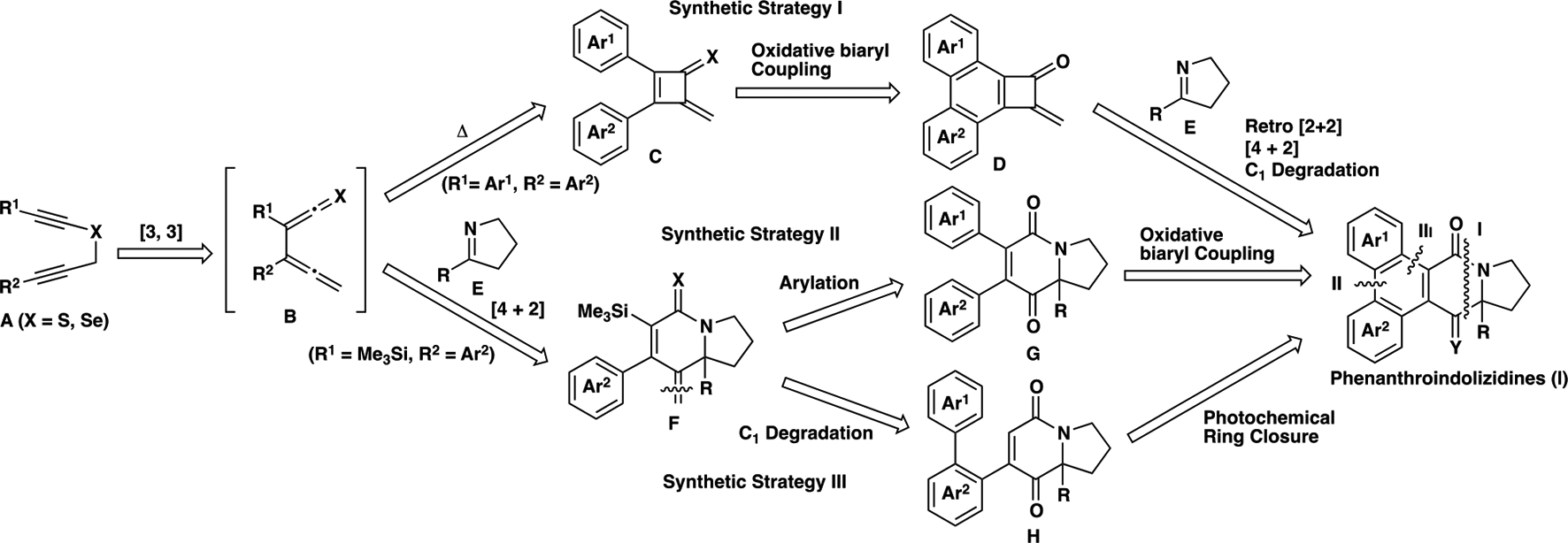
Synthetic strategies for phenanthrolindolizidine alkaloid skeleton I via hetero Diels-Alder reaction of in situ generated α-allenylchalcogenoketene intermediates B.
The synthetic strategy I involves the formation of 2,3-diaryl-4-methylenecyclobutene-1-chalcogenones
At first, 3,4-dimethoxybenzaldehyde (

Subsequently, alkynyl propargyl sulfides
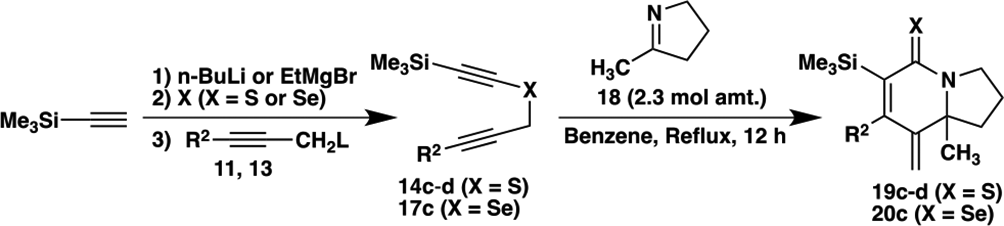
Preparation of Alkynyl Propargyl Sulfides 14a-

MDP, 3,4-(methylenedioxy)phenyl group.
aIsolated yields based on
On the other hand, alkynyl propargyl chalcogenides
Preparation of δ-Chalcogenolactams (19c-d, 20

aIsolated yields based on trimethylsilylacetylene.
Model compound

Conversion of δ-thiolactam 19
In order to realize the synthesis via route

Preparation of alkynyl propargyl chalcogenides (14e, 17e) via Suzuki coupling of 25 and 26 [Procedures: (a) Br2, AcOH; (b) (i) n-BuLi (1,2 mol amt.), (ii) B(OCH3)3 (1.2 mol amt.), (iii) aq. HCl; (c) Pd(OAc)4 (10 mol%), Ph3P (20 mol%), Et3N (excess), DMF; (d) CBr4 (2.0 mol amt.), Ph3P (2.0 mol amt.), CH2Cl2; (e) (i) n-BuLi (2.0 mol amt.), THF, (ii) (CH2O) n (1.0 mol amt.); (f) Ph3P (1.1 mol amt.), CCl4 (excess); and (g) trimethylsilylacetylene (2.0 mol amt.), n-BuLi (2.1 mol amt.), elemental sulfur or selenium (2.0 mol amt.)].
When a benzene solution of sulfide
Preparation of δ-Chalcogenolactams (19e, 20e) from Alkynyl Propargyl Chalcogenides (14e, 17e) and 2-Methylpyrroline (18).
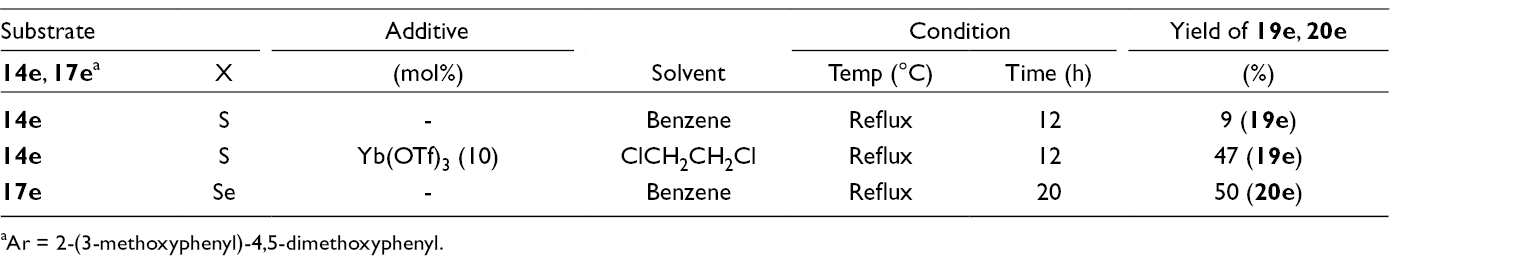
aAr = 2-(3-methoxyphenyl)-4,5-dimethoxyphenyl.

Conversion of δ-chalcogenolactams (19e, 20e) into 5,8-dioxoindolizidine 23e [Procedures: (a) (i) OsO4 (2 mol%), NaIO4 (4.0 mol amt.), aq. dioxane, (ii) H5IO6 (2.0 mol amt.), aq. dioxane; (b) anhydrous K2CO3 (2.0 mol amt.), CH3OH, reflux].
The final ring closure of 5,8-dioindolizidine
Synthesis of 9,14-dioxophenanthroindolizidine 31 by photocyclization of 5,8-dioxoindolizidine 23e

In conclusion, we found a new synthetic method of phenanthroindolizidine core via hetero Diels-Alder reaction of in situ generated α-allenylchalcogenoketenes with cyclic imines and the subsequent photochemical ring closure. Our hetero Diels-Alder methodology for the regioselective access to functionalized and fused indolizidine cores are highly flexible concerning the substitution patterns, and further applications of our new synthetic protocol to the synthesis of various phenanthroindolizidine derivatives having a variety of biological activities are expected in our laboratory.
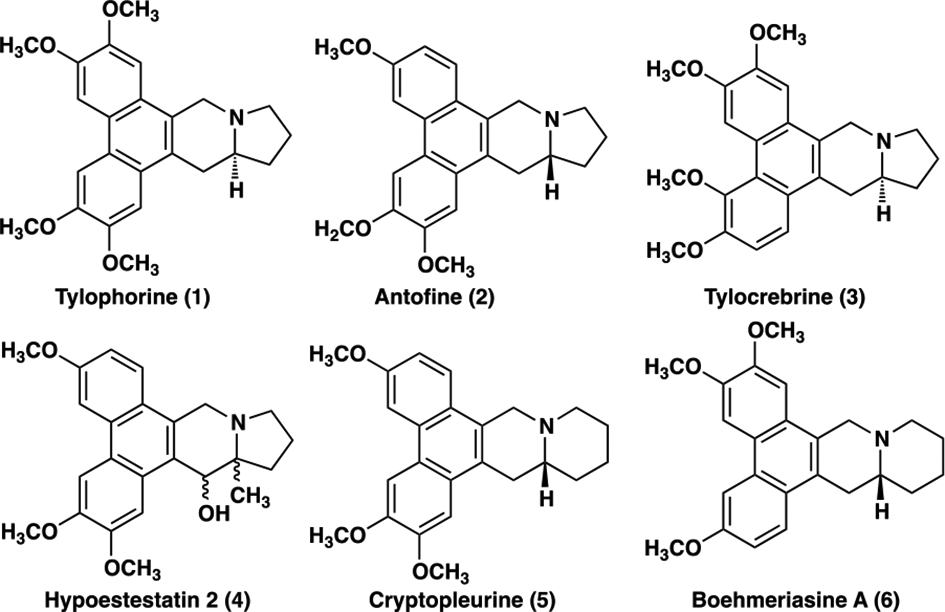
Phenanthroindorizidine and phenanthroquinolizidine alkaloids.
Experimental
Instruments
The melting points were determined with a Barnstead International MEL-TEMP. 1H NMR spectra were recorded on a Bruker DRX-400P (400 MHz) spectrometer or a Bruker AVANCE III 500 (500 MHz) spectrometer, and the chemical shifts of the 1H NMR spectra are given in δ relative to internal tetramethylsilane (TMS). 13C NMR spectra were recorded on a Bruker DRX-400P (100 MHz) or a Bruker AVANCE III 500 (126 MHz) spectrometer. 77Se NMR spectra were recorded on a Bruker DRX-400P (76 MHz) spectrometer. Mass spectra were recorded on a JEOL JMS-700T mass spectrometer with electron-impact ionization at 20 or 70 eV using a direct inlet system. High-resolution mass spectra (HRMS) were also recorded on a JEOL JMS-700T spectrometer. IR spectra were recorded for thin film (neat) or KBr disks on a JASCO FT/IR-7300 spectrometer. Elemental analyses were performed using a Yanagimoto CHN corder MT-5.
A General Procedure for Preparation of Alkynyl Propargyl Chalcogenides (14, 17)
A THF solution of trimethylsilylacetylene was treated with n-butyllithium (1.1 mol amt.) at 0°C for 15 minutes, then with elemental sulfur (1.1 mol amt.) at 0°C for 15 minutes, and then with propargyl bromide (1.0 mol amt.) at room temperature for 1 hour. The reaction was quenched by the addition of water, and the reaction mixture was extracted with benzene. The organic layer was washed twice with water and was dried over anhydrous Na2SO4 powder. The organic solvent was removed in vacuo, and the residual crude products were subjected to column chromatography on silica gel to obtain alkynyl propargyl sulfide
Physical and Spectral Data for Alkynyl Propargyl Sulfides 14 and Selenides 17
Yellow oil.
IR (neat): 2898, 2166, 1501, 1487, 1250, 1226, 1039, 756 cm−1.
1H NMR (CDCl3) δ: 3.83 (2H, s), 5.97 (2H, s), 6.74 (1H, d, J = 1.6 Hz), 6.89 (1H, d, J = 8.0 Hz), 6.97 (1H, dd, J = 8.0, 1.6 Hz), 7.29-7.31 (3H, m), 7.42-7.44 (2H, m).
13C NMR (CDCl3) δ: 25.9 (t), 78.2 (s), 81.9 (s), 85.0 (s), 95.8 (s), 101.4 (t), 108.4 (d), 111.8 (d), 115.9 (s), 123.2 (s), 126.6 (d), 128.4 (d × 2), 131.7 (d), 147.4 (s), 148.1 (s).
HRMS Calcd for C18H12O2S: m/z 292.0558. Found: m/z 292.0558.
Reddish oil.
IR (neat): 2905, 2155, 1595, 1506, 1448, 1327, 1238, 1135, 1033, 812, 616 cm−1.
1H NMR (CDCl3) δ: 3.81 (2H, s), 3.83 (3H, s), 3.88 (3H, s), 5.95 (2H, s), 6.72 (1H, d, J = 8.4 Hz), 6.78 (1H, d, J = 8.4 Hz), 6.88 (1H, d, J = 1.6 Hz), 6.95-6.98 (2H, m), 7.06 (1H, dd, J = 8.4, 1.6 Hz).
13C NMR (CDCl3) δ: 25.9 (t), 55.8 (q), 55.9 (q), 76.3 (s), 82.0 (s), 85.0 (s), 95.8 (s), 101.3 (t), 108.4 (d), 110.9 (d), 111.8 (d), 114.7 (d), 115.3 (s), 116.9 (s), 125.5 (d), 126.5 (d), 147.4 (s), 148.0 (s), 148.5 (s), 149.8 (s).
HRMS Calcd for C20H16O4S: m/z 352.0769. Found: m/z 352.0770.
Yellow oil.
IR (neat): 2960, 2095, 1491, 1250, 833 cm−1.
1H NMR (CDCl3) δ: 0.18 (9H, s), 3.77 (2H, s), 7.30-7.31 (3H, m), 7.43-7.46 (2H, m).
13C NMR (CDCl3) δ: 0.20 (q), 25.5 (t), 83.2 (s), 85.0 (s), 93.0 (s), 103.5 (s), 122.5 (s), 128.1 (d), 128.4 (d), 131.7 (d).
MS (m/z): 244 (M+; bp), 230 (M+-CH3; 96%).
Calcd for C14H16SSi: C, 68.79; H, 6.60%. Found: C, 68.54; H, 6.48%.
Yellow oil.
IR (neat): 2960, 2092, 1514, 1248 cm−1.
1H NMR (CDCl3) δ: 0.17 (9H, s), 3.77 (2H, s), 3.87 (6H, s), 6.79 (1H, d, J = 8.3 Hz), 6.94 (1H, s), 7.05 (1H, dd, J = 8.3, 1.8 Hz).
13C NMR (CDCl3) δ: −0.20 (q), 25.6 (t), 55.7 (q × 2), 81.6 (s), 85.0 (s), 91.3 (s), 103.4 (s), 110.8 (d), 114.4 (d), 114.7 (s), 125.0 (d), 148.4 (s), 149.5 (s).
MS (m/z): 304 (M+; 3%), 175 (M+-TMSCCS; bp).
Calcd for C16H20O2SSi: C, 63.11; H, 6.62%. Found: C, 62.94; H, 6.51%.
Pale yellow oil.
IR (neat): 2960, 2088, 1491, 1250, 860, 844, 758 cm−1.
1H NMR (CDCl3) δ: 0.18 (9H, s), 3.78 (2H, s), 7.25-7.31 (3H, m), 7.42-7.44 (2H, m).
13C NMR (CDCl3) δ: −0.10 (q), 15.4 (t), 84.4 (s), 85.3 (s), 110.5 (s), 122.7 (s), 128.2 (d), 128.4 (d), 131.8 (d).
77Se NMR (CDCl3) δ: 260.0.
MS (m/z): 292 (M+; bp, 80Se), 277 (M+-CH3; 74%, 80Se), 195 (C6H5CCCH2Se; 60%).
Calcd for C14H16SeSi: C, 57.72, H, 5.54%. Found: C, 57.80, H, 5.59%
A General Procedure for the Synthesis of 2,3-Disubstituted 4-Methylene-2-cyclobutene-1-thiones 15
A hexane solution of alkynyl propargyl sulfide
Physical and Spectral Data for 4-Methylene-2-cyclobutene-1-thiones 15
Red oil.
IR (neat): 2899, 1610, 1478, 1244, 1099, 1037, 756 cm−1.
1H NMR (CDCl3) δ: 5.06 (1H, s), 5.40 (1H, s), 6.08 (2H, s), 6.74 (1H, d, J = 8.0 Hz), 6.89 (1H, d, J = 1.6 Hz), 6.95 (1H, d, J = 8.0 Hz), 7.32 (1H, s), 7.38-7.46 (3H, m), 7.49 (1H, d, J = 8.0 Hz), 7.90 (1H, d, J = 8.0 Hz).
13C NMR (CDCl3) δ: 94.6 (t), 102.0 (t), 107.8 (d), 109.2 (d), 124.6 (d), 124.8 (s), 128.1 (d), 128.6 (d), 129.7 (s), 129.8 (d), 148.4 (s), 151.2 (s), 153.9 (s), 157.7 (s), 171.4 (s), 225.8 (s).
HRMS Calcd for C18H12O2S: m/z 292.0558. Found: m/z 292.0561.
Red powder.
MP: 160.5°C-161.7°C
IR (KBr) 2929, 1590, 1510, 1445, 1368, 1267, 1031, 851 cm−1.
1H NMR (CDCl3) δ: 3.86 (3H, s), 3.93 (3H, s), 4.99 (1H, d, J = 1.6 Hz), 5.34 (1H, d, J = 1.6 Hz), 6.09 (2H, s), 6.92 (1H, d, J = 8.4 Hz), 6.96 (1H, d, J = 8.4 Hz), 7.37 (1H, d, J = 1.6 Hz), 7.51 (1H, dd, J = 8.4, 1.6 Hz), 7.57-7.60 (2H, m).
13C NMR (CDCl3) δ: 54.9 (q × 2), 92.3 (t), 101.0 (t), 106.7 (d), 108.1 (d), 110.0 (d × 2), 120.5 (d), 121.5 (s), 123.3 (d), 124.0 (s), 147.3 (s), 147.7 (s), 149.4 (s), 149.9 (s), 153.0 (s), 156.2 (s), 169.0 (s), 225.2 (s).
HRMS Calcd for C20H16O4S: m/z 352.0769. Found: m/z 352.0781.
A General Procedure for the Synthesis of 2,3-Disubstituted 4-Methylene-2-cyclobuten-1-ones 16
A dichloromethane solution of 4-methylene-2-cyclobutene-1-thione
Physical and Spectral Data for 4-Methylene-2-cyclobuten-1-ones 16
Yellow oil.
IR (neat): 2908, 1748, 1548, 1484, 1441, 1351, 1243, 1031, 699 cm−1.
1H NMR (CDCl3) δ: 5.01 (1H, s), 5.26 (1H, s), 6.07 (2H, s), 6.94 (1H, d, J = 8.0 Hz), 7.26 (1H, d, J = 1.6 Hz), 7.36-7.42 (4H, m), 7.80 (2H, dd, J = 8.0, 1.6 Hz).
13C NMR (CDCl3) δ: 95.6 (t), 101.9 (t), 107.8 (d), 109.0 (d), 123.7 (d), 125.0 (s), 127.6 (d), 128.8 (d), 129.4 (s), 129.8 (d), 148.2 (s), 150.5 (s), 154.4 (s), 156.9 (s), 171.6 (s), 188.3 (s).
HRMS Calcd for C18H12O2S: m/z 276.0786. Found: m/z 276.0780.
Yellow oil.
IR (neat): 2910, 1757, 1595, 1512, 1445, 1359, 1256, 1032 cm−1.
1H NMR (CDCl3) δ: 3.86 (3H, s), 3.92 (3H, s), 4.94 (1H, d, J = 1.6 Hz), 5.20 (1H, d, J = 1.6 Hz), 6.08 (2H, s), 6.88 (1H, d, J = 8.4 Hz), 6.95 (1H, d, J = 8.4 Hz), 7.30 (1H, s), 7.37-7.41 (2H, m), 7.48 (1H, dd, J = 8.4, 1.6 Hz).
13C NMR (CDCl3) δ: 55.9 (q), 94.4 (t), 101.9 (t), 107.8 (d), 108.8 (d), 110.2 (d), 111.1 (d), 121.2 (d), 122.2 (d), 123.4 (d), 125.2 (s), 148.2 (s), 148.9 (s), 150.2 (s), 150.5 (s), 154.1 (s), 156.9 (s), 169.8 (s), 188.6 (s).
HRMS Calcd for C20H16O5: m/z 336.0998. Found: m/z 336.0994.
A Typical Procedure for the Synthesis of δ-Chalcogenolactams (19, 20)
A benzene solution of alkynyl propargyl sulfide
Physical and Spectral Data for δ-Chalcogenolactams (19, 20)
Yellow needles.
MP: 155.0°C-156.5°C
IR (KBr): 2971, 2359, 1623, 1246 cm−1.
1H NMR (CDCl3) δ: −0.26 (9H, s), 1.48 (3H, s), 2.09-2.30 (4H, m), 3.81 (1H, br. dt, J = 14.1, 9.3 Hz), 4.07 (1H, br. dt, J = 14.1, 2.2 Hz), 4.88 (1H, s), 5.26 (1H, s), 7.14-7.17 (1H, m), 7.28–7.38 (4H, m).
13C NMR (CDCl3) δ: 2.29 (q), 21.4 (t), 26.2 (q), 38.2 (t), 52.3 (t), 65.3 (s), 118.8 (dd), 127.7 (d), 127.9 (d), 128.5 (d), 130.0 (d), 139.7 (s), 140.9 (s), 147.7 (s), 151.5 (s), 190.4 (s).
MS (m/z): 327 (M+−1; 4%), 296 (M+-S; bp), 73 ((CH3)3Si; 30%).
Calcd for C19H25NSSi: C, 69.67; H, 7.69; N, 4.28%. Found: C, 69.45; H, 7.56; N, 4.33%.
Yellow needles.
MP: 131.5°C-132.6°C
IR (KBr): 3097, 2971, 1602, 1454, 1266, 1246 cm−1.
1H NMR (CDCl3) δ = −0.21 (9H, s), 1.47 (3H, s), 2.12-2.27 (4H, m), 3.76 (3H, s), 3.83 (3H, s), 3.80-–4.10 (2H, m), 4.96 (1H, s), 5.26 (1H, s), 6.76-6.83 (3H, m).
13C NMR (CDCl3) δ: 2.11 (q), 2.13 (q), 21.1 (t), 25.7 (q), 37.8 (t), 37.9 (t), 51.8 (t), 51.9 (t), 65.0 (s), 109.9 (d), 110.7 (d), 112.7 (d), 114.4 (d), 118.3 (s), 139.7 (s), 140.1 (s), 146.9 (s), 151.1 (s), 190.1 (s).
MS (m/z): 387 (M+; 2%), 327 (M+-CH3; bp), 73 ((CH3)3Si; 3%).
Calcd for C21H29NO2SSi: C, 65.07; H, 7.54; N, 3.61%. Found: C, 64.92; H, 7.44; N, 3.57%.
Red needles.
MP: 142.9°C-133.1°C
IR (KBr): 3074, 2970, 1635, 1519, 1488, 1241, 1176, 861 cm−1.
1H NMR (CDCl3) δ: 0.08 (9H, s), 1.48 (3H, s), 2.15-2.36 (4H, m), 3.72-3.80 (1H, m), 4.06-4.12 (1H, m), 5.00 (1H, s), 5.34 (1H, s), 7.16-7.18 (1H, m), 7.32-7.39 (4H, m).
13C NMR (CDCl3) δ: 2.68 (q), 21.4 (t), 25.2 (q), 38.1 (t), 55.9 (t), 65.8 (s), 119.2 (d), 127.7 (d), 128.1 (d), 128.6 (d), 130.1 (d), 131.0 (d), 139.7 (s), 144.0 (s), 145.2 (s), 151.5 (s), 192.2 (s).
77Se NMR (CDCl3) δ: 671.5 (s).
MS (m/z): 375 (M+; 18%), 360 (M+-CH3; bp), 83 (C5H9N; 95%).
Calcd for C19H25NSeSi: C, 60.94; H, 6.73; N, 3.74%. Found: C, 61.21; H, 6.48; N, 3.51%.
Conversion of δ-Thiolactam 19c Into δ-Lactam 22c
An aqueous dioxane solution (dioxane:H2O = 4:1) of δ-thiolactam
Physical and Spectral Data for δ-Lactam 22c
Yellow needles.
MP: 118.6°C-119.4°C
IR (neat): 2974, 1713, 1441, 1243 cm−1.
1H NMR (CDCl3) δ: −0.03 (9H, s), 1.35 (3H, s), 1.95-2.20 (4H, m), 3.60-3.70 (1H, m), 3.74-3.81 (1H, m), 7.05-7.20 (2H, m), 7.35-7.40 (3H, m).
MS (m/z): 314 (M++1; 54%), 298 (M+-CH3; bp).
Calcd for C18H23NO2Si: C, 68.97; H, 7.40; N, 4.47%. Found: C, 68.72; H, 7.34; N, 4.56%.
Desilylation of δ-Lactam 22c
A methanol solution of δ-lactam
Physical and Spectral Data for 5,8-Dioxoindolizidine 23c
Yellow oil.
IR (neat): 2997, 1694, 1655, 1597, 1433, 1112, 704 cm−1.
1H NMR (CDCl3) δ: 1.25 (3H, s), 1.60-1.80 (2H, m), 1.90-2.05 (2H, m), 3.35-3.50 (1H, m), 3.55-3.75 (1H, m), 7.06 (1H, s), 7.10-7.30 (5H, m).
Preparation of Biphenyl Derivative 27
A THF solution of 3-bromoanisole was treated with n-butyllithium (1.2 mol amt.) at −78°C for 30 minutes, and the reaction mixture was treated with B(OCH3)3 (1.2 mol amt.) at −78°C for 1 hour and then at room temperature for 2 hours. The reaction was quenched by the addition of aqueous 1 M HCl solution, and the reaction mixture was extracted with diethyl ether. The organic layer was washed with water and was dried over anhydrous Na2SO4 powder. The organic solvent was removed in vacuo, and the residual crude products were washed with hexane to obtain boronic acid
Physical and Spectral Data for Biphenyl Aldehyde 27
Yellow prisms.
MP: 97.5°C-97.9°C
IR (KBr): 2940, 1669, 1506, 1272, 1154, 1042, 991, 757 cm−1.
1H NMR (CDCl3) δ: 3.85 (3H, s), 3.95 (3H, s), 3.97 (3H, s), 6.86 (1H, s), 6.91-6.98 (3H, m), 7.30-7.38 (1H, m), 7.53 (1H, m), 9.84 (1H, s).
13C NMR (CDCl3) δ: 55.0 (q), 55.8 (q), 55.9 (q), 108.2 (d), 112.2 (d), 113.1 (d), 115.7 (d), 122.5 (d), 126.7 (d), 129.0 (d), 138.7 (s), 141.0 (s), 148.5 (s), 153.1 (s), 159.2 (s), 190.8 (d).
MS (m/z): 272 (M+; bp), 241 (M+-OCH3; 20%).
Calcd for C16H16O4: C, 70.57; H, 5.92%. Found: C, 70.67; H, 5.85%.
Conversion of Biphenyl Aldehyde 27 Into 1,1-Dibromoalkene 28
A dichloromethane solution of biphenyl aldehyde
Physical and Spectral Data for 1,1-Dibromoalkene 28
Yellow needles.
MP: 78.0°C-78.7°C
IR (KBr): 3018, 2936, 1606, 1566, 1488, 1464, 1254, 1137, 1051, 1030, 872, 753 cm−1.
1H NMR (CDCl3) δ: 3.85 (3H, s), 3.91 (3H, s), 3.94 (3H, s), 6.86 (1H, s), 6.86 (1H, s), 6.90 (1H, br. d, J = 7.8 Hz), 6.92 (1H, br. d, J = 7.8 Hz), 7.21 (1H, s), 7.28 (1H, s), 7.33 (1H, t, J = 7.8 Hz).
13C NMR (CDCl3) δ: 55.2 (q), 55.9 (q), 56.0 (q), 89.1 (s), 111.8 (d), 112.4 (d), 113.1 (d), 115.0 (d), 121.9 (d), 125.7 (s), 129.2 (d), 134.2 (s), 137.0 (d), 141.3 (s), 147.7 (s), 148.9 (s), 159.2 (s).
MS (m/z): 430 (M+; 2%, 81Br), 428 (M+; 3%, 81Br+79Br), 426 (M+; 2%, 79Br), 320 (M+-C6H4OCH3; 3%), 268 (M+-Br2; 39%).
Calcd for C17H16Br2O3: C, 47.69; H, 3.77%. Found: C, 47.61; H, 3.72%.
Conversion of 1,1-Dibromoalkene 28 Into Propargyl Alcohol 29
A THF solution of 1,1-dibromoalkene
Physical and Spectral Data for Propargyl Alcohol 29
Yellow oil.
IR (neat): 3504, 2920, 2225, 1516, 1496, 1255, 1219, 1152, 1028, 999, 755 cm−1.
1H NMR (CDCl3) δ: 1.70 (1H, br. s), 3.86 (3H, s), 3.90 (6H, s), 4.35 (2H, d, J = 5.8 Hz), 6.87 (1H, s), 7.02 (1H, s), 7.10-7.16 (2H, m), 7.16 (1H, s), 7.32 (1H, t, J = 7.9 Hz).
13C NMR (CDCl3) δ: 51.5 (t), 55.2 (q), 55.8 (q), 55.9 (q), 85.2 (s), 88.5 (s), 112.2 (d), 112.5 (s), 112.7 (d), 114.8 (d), 115.3 (d), 121.5 (d), 128.9 (d), 137.0 (s), 141.6 (s), 147.7 (s), 149.3 (s), 159.0 (s).
MS (m/z): 298 (M+; 37%), 281 (M+-OH; 6%), 267 (M+-OCH3; 7%).
Calcd for C18H18O4: C, 72.47; H, 6.08%. Found: C, 72.31; H, 6.20%
Conversion of Propargyl Alcohol 29 Into Propargyl Chloride 30
A CCl4 solution (excess) of propargyl alcohol
Physical and Spectral Data for Propargyl Chloride 30
Yellow needles.
MP: 80.5°C-81.2°C
IR (KBr): 2937, 1602, 1516, 778 cm−1.
1H NMR (CDCl3) δ: 3.78 (3H, s), 3.89 (6H, s), 4.25 (2H, d, J = 0.7 Hz), 6.68 (1H, s), 6.89 (1H, d, J = 8.3 Hz), 7.02 (1H, s), 7.07-7.13 (1H, m), 7.32 (1H, t, J = 8.3 Hz).
13C NMR (CDCl3) δ = 31.3 (t), 55.1 (q), 55.8 (q), 55.9 (q), 84.8 (s), 86.2 (s), 111.8 (s), 112.2 (d), 113.1 (d), 114.4 (d), 115.4 (d), 121.4 (d), 128.9 (d), 137.6 (s), 141.3 (s), 147.7 (s), 149.6 (s), 159.0 (s). MS (m/z): 318 (M+; 10%, 37Cl), 316 (M+; 23%, 35Cl), 281 (M+-Cl; 11%), 175 (C6H3(OCH3)2CCCH2; bp), 51 (CH2Cl; 3%, 37Cl), 49 (CH2Cl; 11%, 35Cl).
Calcd for C18H17ClO3: C, 68.25; H, 5.41%. Found: C, 68.10; H, 5.42%.
A Typical Procedure for Preparation of Alkynyl Propargyl Chalcogenides (14e, 17e)
A THF solution of trimethylsilylacetylene (930 mg, 2.0 mol amt.) was treated with n-butyllithium (7.0 mL, 2.1 mol amt.) at 0°C for 30 minutes, then with elemental selenium (748 mg, 2.0 mol amt.) at 0°C for 15 minutes, and then with propargyl chloride
Physical and Spectral Data for 14e and 17e
Yellow oil.
IR (neat): 2961, 2093, 1602, 1516, 1252, 1029, 880, 845 cm-1.
1H NMR (CDCl3) δ: 0.15 (9H, s), 3.68 (2H, s), 3.84 (3H, s), 3.89 (6H, s), 6.84-6.89 (3H, m), 7.02 (1H, s), 7.11 (1H, s), 7.29-7.34 (1H, m).
13C NMR (CDCl3) δ = −0.22 (q), 25.8 (t), 55.1 (q), 55.7 (q), 55.8 (q), 84.2 (s), 84.8 (s), 93.2 (s), 103.2 (s), 111.8 (s), 112.2 (d), 113.1 (d), 114.4 (d), 115.4 (d), 121.4 (d), 128.9 (d), 137.6 (s), 141.4 (s), 147.7 (s), 149.6 (s), 159.0 (s).
MS (m/z): 410 (M+; 12%), 395 (M+-CH3; 7%), 281 (M+-C5H3SSi; 87%), 73 ((CH3)3Si; bp).
Calcd for C23H26O3SSi: C, 67.28; H, 6.38%. Found: C, 67.11; H, 6.21%.
Orange oil.
IR (neat): 2958, 2087, 1712, 1602, 1516, 1250, 860 cm−1.
1H NMR (CDCl3) δ: 0.14 (9H, s), 3.70 (2H, s), 3.87 (3H, s), 3.91 (6H, s), 6.85-6.87 (1H, m), 6.90 (1H, d, J = 7.9 Hz), 7.01 (1H, s), 7.10-7.11 (1H, m), 7.17 (1H, d, J = 7.9 Hz), 7.34 (1H, t, J = 7.9 Hz).
13C NMR (CDCl3) δ: −0.12 (q), 15.3 (t), 55.2 (q), 55.8 (q), 55.9 (q), 85.1 (s), 85.3 (s), 85.8 (s), 110.2 (s), 112.2 (d), 112.7 (d), 112.9 (d), 114.5 (d), 115.4 (d), 121.5 (d), 129.0 (d), 137.4 (s), 141.5 (s), 147.7 (s), 149.3 (s), 159.1 (s).
MS (m/z): 458 (M+; 8%), 281 (M+-C5H9SeSi; bp), 250 (M+-C6H12OSeSi; 70%), 73 ((CH3)3Si; 19%).
Calcd for C23H26O3SeSi: C, 60.38; H, 5.73%. Found: C, 60.23; H, 5.82%.
A Typical Procedure for the Synthesis of δ-Chalcogenolactams (19e, 20e)
A benzene solution of alkynyl propargyl selenide
Synthesis of δ-Thiolactam 19e by Thermal Reaction of 14e in the Presence of Yb(OTf)3
A dichloromethane solution of alkynyl propargyl sulfide
Physical and Spectral Data for 19e and 20e
Yellow needles.
IR (KBr): 2958, 1711, 1602, 1516, 1253 cm−1.
1H NMR (CDCl3) δ: 0.21 (9H, s), 0.78 (3H, s), 1.24-1.27 (2H, m), 1.95-2.04 (4H, m), 3.75 (3H, s), 3.88 (3H,s), 3.92 (3H, s), 4.67 (1H, s), 4.86 (1H, s), 6.74-6.79 (4H, m), 7.19-7.32 (2H, m).
13C NMR (CDCl3) δ: 3.33 (q), 20.7 (t), 26.7 (q), 38.5 (t), 52.5 (t), 55.1 (q), 55.9 (q × 2), 65.2 (s), 111.7 (d), 113.4 (d), 115.2 (d), 115.5 (d), 118.9 (d), 121.6 (d), 129.0 (d), 129.9 (s), 133.5 (s), 140.2 (s), 142.7 (s), 147.6 (s), 148.4 (s), 148.8 (s), 149.0 (s), 159.0 (s), 189.4 (s).
MS (m/z): 493 (M+; 4%), 478 (M+-CH3; bp), 462 (M+-OCH3; 10%), 31 (CH3O; 11%).
Calcd for C28H35NO3SSi: C, 68.11; H, 7.15; N, 2.84%. Found: C, 67.97; H, 7.18; N, 2.81%.
Red needles.
MP: 79.9°C-80.6°C
IR (KBr): 2958, 1601, 1521, 1437, 1251, 1173, 842 cm−1.
1H NMR (CDCl3) δ: 0.26 (9H, s), 0.86 (3H, s), 2.02-2.04 (4H, m), 3.70-3.74 (1H, m), 3.76 (3H, s), 3.92 (3H, s), 3.94 (3H, s), 4.00-4.06 (1H, m), 4.77 (1H, s), 4.92 (1H, s), 6.77 (1H, d, J = 7.8 Hz), 6.81 (1H, s), 6.87-6.89 (3H, m), 7.20 (1H, t, J = 7.8 Hz).
13C NMR (CDCl3) δ: 3.85 (q), 20.7 (t), 26.0 (q), 38.5 (t), 55.1 (q), 55.9 (q × 2), 56.3 (t), 65.8 (s), 111.7 (d), 113.5 (d), 115.2 (d), 115.4 (d), 119.5 (s), 121.6 (d), 129.0 (d), 129.9 (s), 133.4 (s), 142.7 (s), 143.0 (s), 146.6 (s), 147.6 (s), 148.3 (s), 149.1 (s), 159.0 (s), 190.8 (s).
MS (m/z): 541 (M+; 14%), 526 (M+-CH3; bp), 462 (M+-Se; 9%), 388 (M+-Se-(CH3)3Si; 7%).
Calcd for C28H35NO3SeSi: C, 62.21; H, 6.53; N, 2.59%. Found: C, 62.02; H, 6.63; N, 2.52%
A Typical Procedure for the Conversion of δ-Chalcogenolactams (19e, 20e) Into δ-Lactam 22e
An aqueous dioxane solution (dioxane:H2O = 4:1) of δ-selenolactam
Physical and Spectral Data for δ-Lactam 22e
Yellow needles.
MP: 74.6°C-75.6°C
IR (KBr): 2977, 1689, 1629, 1604, 1512, 1420, 1251, 1049 cm−1.
1H NMR (CDCl3) δ: 0.17 (3H, s), 0.78 (9H, s), 1.90-1.92 (4H, m), 3.55-3.60 (1H, m), 3.67-3.73 (1H, m), 3.77 (3H, s), 3.89 (3H, s), 3.92 (3H, s), 6.64 (1H, s), 6.79 (1H, s), 6.79 (1H, d, J = 8.4 Hz), 6.86 (1H, d, J = 7.2 Hz), 6.87 (1H, s), 7.23 (1H, t, J = 8.4, 7.2 Hz).
13C NMR (CDCl3) δ: 0.58 (q), 20.3 (dd), 25.8 (q), 34.6 (t), 55.0 (q), 55.8 (q), 55.9 (q), 68.3 (s), 112.1 (d), 113.0 (d), 114.5 (d), 115.2 (d), 121.6 (d), 126.0 (s), 129.1 (d), 133.8 (s), 142.3 (s), 147.7 (s), 149.3 (s), 151.4 (s), 152.6 (s), 159.0 (s), 163.9 (s), 198.3 (s).
MS (m/z): 478 (M+−1; 50%), 464 (M+-CH3; bp), 448 (M+-OCH3; 6%), 406 (M+-(CH3)3Si; 36%), 73 ((CH3)3Si; 10%).
Calcd for C27H33NO5Si: C, 67.61; H, 6.93; N, 2.92%. Found: C, 67.25; H, 7.05; N, 3.01%.
Desilylation of δ-Lactam 22e
A methanol solution of δ-lactam
Physical and Spectral Data for 5,8-Dioxoindolizidine 23e
Yellow needles.
MP: 75.3°C-76.7°C
IR (KBr): 2976, 1704, 1651, 1516, 1455, 1254, 1161, 1047 cm−1.
1H NMR (CDCl3) δ: 0.97 (3H, s), 1.92-1.97 (4H, m), 3.56-3.62 (1H, m), 3.74-3.78 (1H, m), 3.79 (3H, s), 3.90 (3H, s), 3.91 (3H, s), 6.75 (1H, s), 6.78-6.79 (1H, m), 6.83 (2H, d, J = 7.5 Hz), 6.86 (1H, s), 6.88 (1H, s), 7.23 (1H, t, J = 7.5 Hz).
13C NMR (CDCl3) δ: 20.1 (t), 25.6 (q), 34.2 (t), 44.9 (t), 55.2 (q), 55.9 (q), 56.0 (q), 62.6 (s), 112.1 (d), 113.1 (d), 113.4 (d), 115.5 (d), 121.9 (d), 123.7 (s), 129.3 (d), 134.7 (s), 142.4 (s), 145.6 (s), 148.2 (s), 149.6 (s), 159.2 (s), 160.3 (s), 197.6 (s).
MS (m/z): 406 (M+−1; bp), 391 (M+-CH3-1; 35%).
Calcd for C24H25NO5: C, 70.74; H, 6.18; N, 3.44%. Found: C, 70.59; H, 6.02; N, 6.21%.
Synthesis of 9,14-Dioxophenanthroindolizidine 31 by Iodine-Assisted Photochemical Cyclization of 5,8-Dioxoindolizidine 23e
A dichloromethane or a methanol solution of 5,8-dioxoindolizidine
Physical and Spectral Data for 9,14-Dioxophenanthroindolizidine 31
Pale yellow needles.
MP: 218.7°C-219.4°C
IR (KBr): 3119, 2980, 1646, 1614, 1520, 1425, 1260, 1112, 1051 cm−1.
1H NMR (CDCl3) δ: 1.47 (3H, s), 2.12-2.18 (3H, m), 2.47-2.50 (1H, m), 3.88-3.92 (2H, m), 4.05 (3H, s), 4.09 (3H, s), 4.14 (3H, s), 7.03 (1H, dd, J = 9.4, 2.6 Hz), 7.86 (1H, d, J = 2.6 Hz), 7.89 (1H, s), 8.81 (1H, s), 9.41 (1H, d, J = 9.4 Hz).
13C NMR (CDCl3) δ: 21.3 (t), 26.2 (q), 34.6 (dd), 46.6 (t), 55.4 (q), 55.7 (q), 55.8 (q), 68.9 (s), 102.8 (d), 103.8 (d), 106.9 (d), 116.0 (d), 122.1 (s), 122.4 (s), 123.2 (s), 126.0 (s), 131.1 (s), 131.9 (d), 134.9 (s), 150.2 (s), 160.2 (s), 161.9 (s), 201.6 (s).
MS (m/z): 406 (M++1; bp), 391 (M+-CH3 + 1; 35%).
Calcd for C24H23NO5: C, 71.10; H, 5.72; N, 3.45%. Found: C, 71.21; H, 5.85; N, 3.49%.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed no financial support for the research, authorship, and/or publication of this article.