Abstract
Levopimaradiene and dehydroabietane are two direct precursors of ginkgolides and exist only in the root tissue of Ginkgo biloba. To study the biosynthesis of ginkgolide, we chose undifferentiated G. biloba root tissue stem cells, which contain all the information needed for plant growth and development. Only dehydroabietane and abietadiene were determined by gas chromatography mass spectrometry in G. biloba root stem cell cultures. The results are consistent with the fermentation products of levopimaradiene synthase present in Escherichia coli under ultrasonic and bright light conditions. In addition, the products were also changed with changes in light and ultrasonic conditions and with just changes in light, indicating that levopimaradiene is converted to either dehydroabietane or abietadiene in the original plant in response to changes in external regulation and the plant’s own needs.
Ginkgolides are only present in Ginkgo biloba L. and their medicinal values have become increasingly important to humans. 1 Although there have been extensive studies on the biosynthesis of terpenoids in the cytoplasm and plastids of angiosperms, there has been little investigation of the function of these enzymes in gymnosperms. Levopimaradiene synthase (GbLPS) promotes the cyclization of geranylgeranyldiphosphate to produce dehydroabietane, which is considered to be the first step in the biosynthesis of ginkgolides. 2 Dehydroabietane is transported from the plastid to the cytoplasm, where it is converted into ginkgolide through a series of complex reactions. 3
Studies that utilize metabolic localization show that although ginkgolides accumulate in leaves, the precursor compounds are only found in the roots. 4 The deep transcriptome sequencing was obtained of stably grown G. biloba root stem cells, which maintain embryogenic proliferation and differentiation capabilities, and G. biloba calli. The results showed that the stem cell marker genes WUS and WOX were significantly upregulated in G. biloba root stem cells compared with dedifferentiated calli (Figure 1).

Differential expression of stem cell marker genes in Ginkgo biloba root stem cells and dedifferentiated calli.
For the first time, G. biloba root stem cells have been used to produce direct precursor compounds of ginkgolides. 5 By gas chromatography mass spectrometry (GC-MS), G. biloba root stem cells were shown to produce dehydroabietane and abietadiene, but not levopimaradiene. Ginkgo biloba calli did not produce any ginkgolides-related compound (Figure 2). These results indicated that G. biloba root stem cells contain the factors necessary for the growth and development of G. biloba.
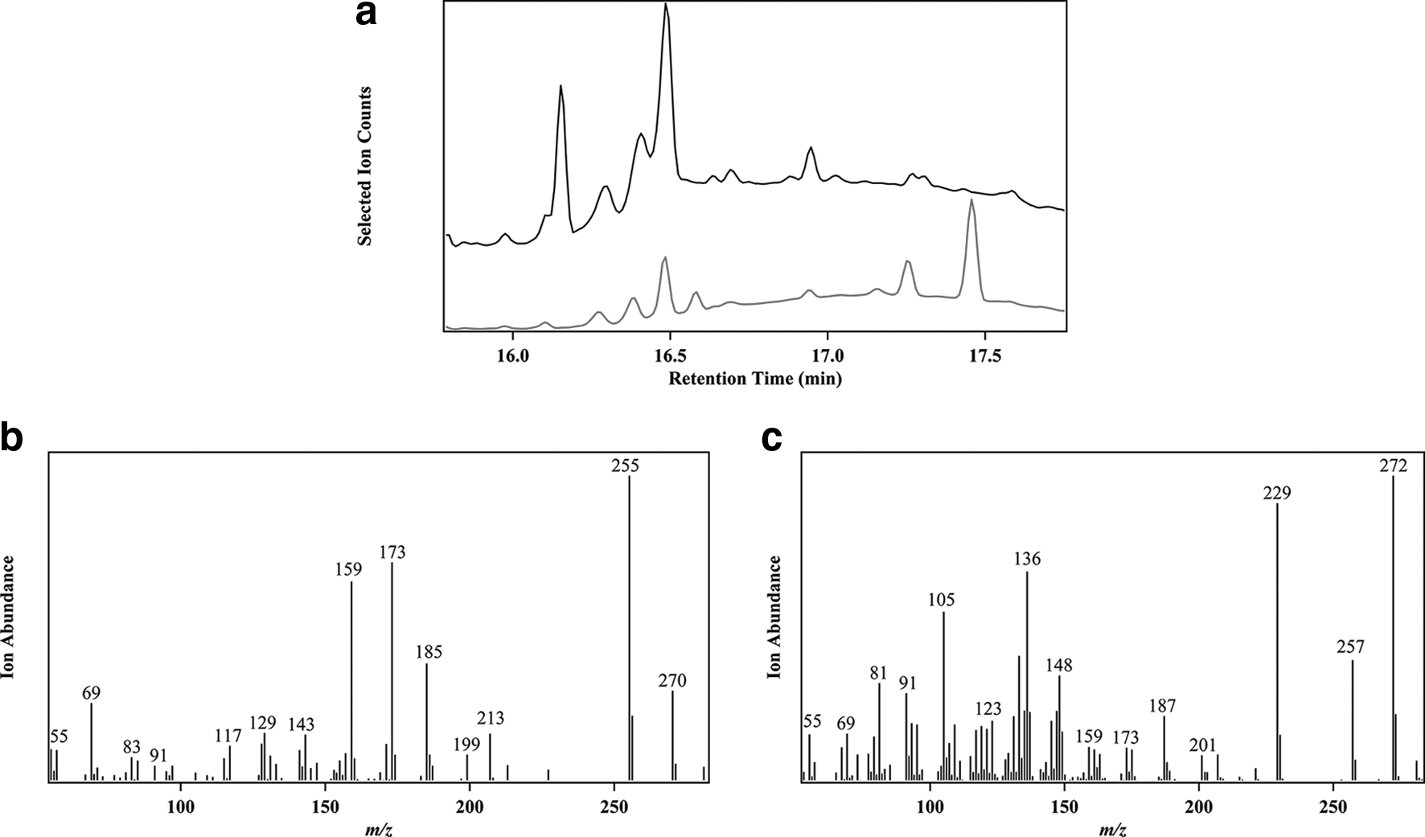
(a): Gas chromatography mass spectrometry profile of Ginkgo biloba root stem cells (a) and calli (b) suspension media. (b) Selected mass ion chromatogram of peak 1 (dehydroabietane) (m/z 270). (c) Selected mass ion chromatogram of peak 2 (abietadiene) (m/z 272). Escherichia coli engineered expressed compounds dehydroabietane and abietadiene mass spectra were identical to those in the NIST database.
Levopimaradiene has been reported to undergo a further oxidation process to produce dehydroabietane. 5 Therefore, we decided to clone and perform a functional characterization of GbLPS in Escherichia coli to study further the biosynthetic pathway of ginkgolide biosynthesis in G. biloba root stem cells.
The results demonstrated that the fermentation products of GbLPS contained dehydroabietane and abietadiene, but no levopimaradiene. This result is consistent with the detection of these compounds in G. biloba root cell suspension medium. Thus, we could propose that levopimaradiene is not very stable, and electron migration and oxidation reactions very likely occur under ultrasound and light conditions. Based on the above experiments, two results were obtained: three compounds were produced in the experimental group for the fermentation product under ultrasound and dark conditions, levopimaradiene, dehydroabietane, and abietadiene, but only two compounds, levopimaradiene and dehydroabietane, were produced in the experimental group for fermentation products under dark conditions without ultrasound.
Levopimaradiene generates dehydroabietane through an oxidation reaction, and the intermediate compound can produce abietadiene through electron migration. Light, ultrasound, and thermal energy are all different forms of energy. The conjugated unsaturated double bond in levopimaradiene is prone to electronic transitions and oxidation reactions at high temperatures and upon exposure to light and oxygen (Figure 3). We speculate that due to the external environmental pressure, levopimaradiene present in G. biloba root tissue could generate dehydroabietane through further spontaneous oxidation reactions, followed by the catalytic action of terpene synthases and subsequent generation of ginkgolide. Figure 3 gives the electron migration and oxidation reactions of levopimaradiene.
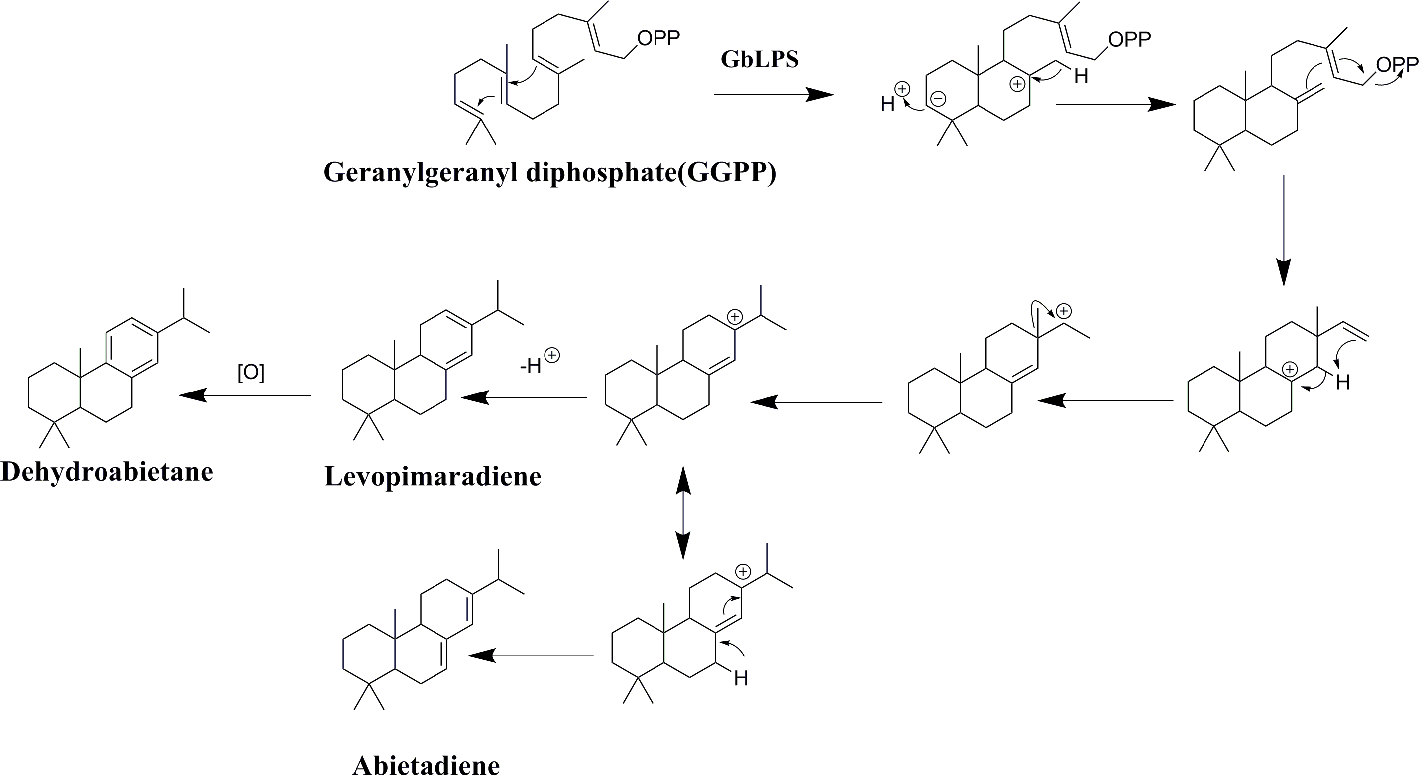
Speculated electron migration and oxidation reactions of levopimaradiene.
Experimental
Induction of Ginkgo biloba Root Stem Cells
The washed root tissue of G. biloba (with a 1-year growth period) was first sterilized with 75% ethanol for 1 minute, then with 0.1% HgCl2 for 6 minutes, rinsed with distilled water until no sterilizing agent was left, and placed in 150 mg/L citric acid solution. A penicillin-streptomycin solution was added to the surface of the culture medium (MS +1.0 mg/L 2,4-D + 1.0 mg/L NAA; agar, 9 g/L; sucrose, 30 g/L; pH = 5.8), and 0.8 mL penicillin-streptomycin solution per 50 mL culture medium. G. biloba root tissue was cut into approximately 1 cm segments using a medical scalpel. The pith was removed longitudinally, and the cambium layer placed upright on the surface of the culture medium. The culture was incubated in the dark at 25°C. 6
Ginkgo biloba Root Stem Cells and Calli Culture Detection
Gas chromatography mass spectrometry experiments were performed as in reference. 7
GbLPS Gene Cloning and Recombinant Expression
Full-length coding sequences for GbLPS (GenBank: AY574248.2) were PCR amplified using gene-specific primers, GbLPS F (5′-CATATGATGGCTGGGGTGCTCTTTGC-3′) and GbLPS R (5′-CTCGAGCTACGCCACAGGATCGAAAAG-3′). GbLPS recombinant expression was conducted as in reference. 2,7
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Sciences Foundation of China (No. 81 573 568 and 81673571).
