Abstract
Two new acetophenone derivatives named knepachycarpanone A (
Knema is a genus of evergreen trees in the Myristicaceae family, comprising around 60 species distributed in tropical and subtropical regions of South East Asia.
1
Knema pachycarpa de Wilde is a native tree in Vietnam found in several central provinces.
2
Knema plants have been used in the traditional medicine for treatment of pimples, sores, and skin diseases.
3,4
Previous chemical studies of Knema species have reported the isolation of anacardic acids, cardanols, resorcinols, acetophenones, lignans, stilbene, and flavonoids.
5
-12
Knema plant exhibited antibacterial, anti-inflammatory, antioxidant, cytotoxic, and acetylcholinesterase inhibitory activities.
5
-14
Several lignans have been reported from the ethyl acetate extract of K. pachycarpa fruits
15
but the chemical composition of the stems of this plant has not yet been investigated. Herein, we describe the isolation and structure elucidation of 2 new acetophenone derivatives named knepachycarpanone A (
Compound
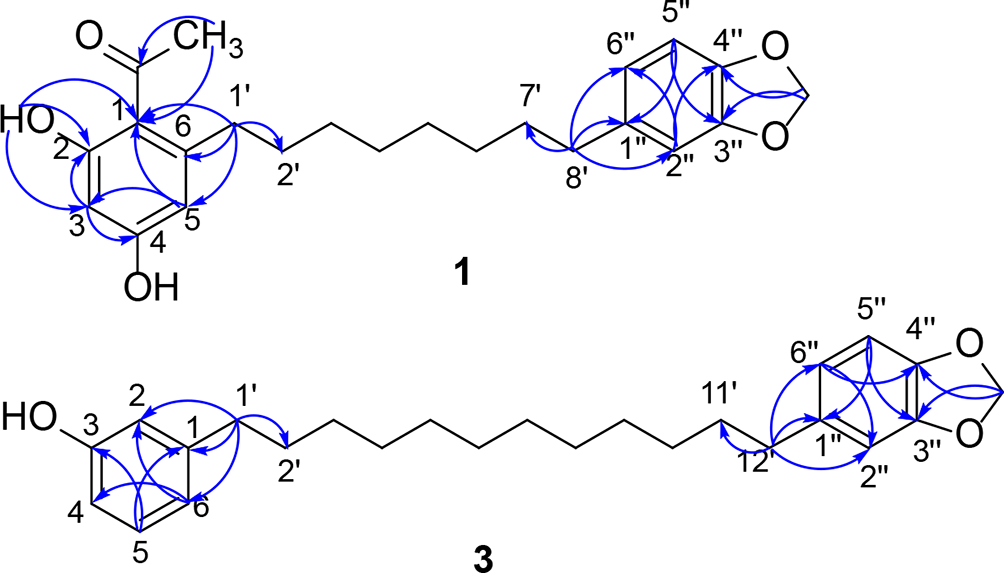
Key heteronuclear multiple bond correlations of 1 and 3.

Structures of new compounds 1 to
Compound
Compound
Acetophenone and cardanol derivatives have been isolated from Knema species such as the roots of Knema globularia and the stems of Knema glomerata and evaluated for cytotoxicity against several human cancer lines.
7,9
Therefore, in our study the cytotoxicity of these isolated compounds
Cytotoxicity of 1 to

In conclusion, chemical investigation of the stems of K. pachycarpa resulted in the isolation and identification 2 new acetophenone derivatives, knepachycarpanone A (
General Experimental Procedures
The UV spectra were recorded on a Shimadzu UV-240 spectrophotometer. IR spectra were obtained on a Bruker TENSOR 37 FT-IR spectrometer using KBr pellets. The HR-ESI-MS were obtained using an Agilent 6530 Accurate mass quadrupole time of flight liquid chromatography mass spectrometry system. One-dimensional and 2D NMR spectra were recorded on a Bruker Avance 500 MHz spectrometer, chemical shifts (δ) are expressed in ppm with reference to the tetramethyl silane signals. Column chromatography (CC) was performed with silica gel (230-400 mesh, Merck). Fractions were monitored by thin-layer chromatography (TLC), spots were visualized by UV light (254 and 365 nm) or by heating silica gel plates sprayed with Ce-Mo stain. All solvents used in CC were redistilled.
Plant Materials
The stems of K. pachycarpa de Wilde (Myristicaceae) were collected in A-Luoi, Hue city, Viet Nam, in October, 2015, and identified by Dr. Nguyen The Cuong, Institute of Ecology and Biological Resources, Vietnam Academy of Science and Technology. A voucher specimen (VN-1527) was deposited at the Herbarium of Institute of Marine Biochemistry and Institute of Ecology and Biological Resources, Vietnam Academy of Science and Technology.
Extraction and Isolation
The air-dried and powdered stems of K. pachycarpa (1.2 kg) were extracted with MeOH (10 L × 4 times) at room temperature for 1 day each time. The MeOH extracts were concentrated in vacuo, suspended in H2O (500 mL), and partitioned successively with n-hexane and ethyl acetate (1 L × 3). The EtOAc extract (160 g) was subjected to silica gel CC and eluted with solvent gradient of n-hexane/EtOAc to afford 8 fractions (E1-E8). Fraction E1 (2.5 g) was chromatographed on silica gel column (n-hexane/acetone, 98:2) to give 4 subfractions E1.1-E1.4. Subfraction E1.4 (0.65 g) was separated by silica gel CC and eluted with n-hexane/acetone, 95:5 to yield 4 smaller fractions E1.4.1-E1.4.4. Fraction E1.4.3 (0.5 g) was purified by Sephadex® LH-20 CC, eluted with MeOH-CH2Cl2 (9:1) to yield
Knepachycarpanone a (1)
White amorphous powder.
MP: 66-67°C.
IR νmax (KBr): 3363, 2925, 2854, 1725, 1587, 1503, 1488, 1244, 1039 cm–1.
UV (CHCl3) λ max (log ε) 281 (1.04), 322 (0.37).
1H NMR (CDCl3, 500 MHz) and 13C NMR (CDCl3, 125 MHz): Table 2.
1H and 13C NMR of Compounds 1, 2, and 3 (CDCl3).

Recorded at a) 125 MHz, b) 500 MHz
HR-ESI-MS (positive-ion mode) m /z 385.2008 [M + H]+, calcd for C23H29O5, 385.2015.
Knepachycarpanone B (2)
White amorphous powder.
MP: 67-68°C.
IR νmax (KBr): 3170, 2922, 2849, 1731, 1589, 1504, 1488, 1235, 1032 cm–1.
UV (CHCl3) λ max (log ε) 281 (1.49), 322 (0.48).
1H NMR (CDCl3, 500 MHz) and 13C NMR (CDCl3, 125 MHz): Table 2.
HR-ESI-MS (positive-ion mode): m/z 413.2302 [M+H]+, calcd for C25H33O5, 413.2328.
Knepachycarpanol C (3)
White amorphous powder.
MP: 56-57°C.
IR νmax (KBr) 3381, 2923, 2852, 1588, 1503, 1488, 1442, 1243, 1155, 1039 cm–1.
UV (CHCl3) λ max (log ε) 280 (0.49).
1H NMR (CDCl3, 500 MHz) and 13C NMR (CDCl3, 125 MHz): Table 2.
HR-ESI-MS (negative-ion mode): m/z 417.2210 [M+Cl]-, calcd for C25H34O3Cl, 417.2196.
Cytotoxicity Assay
The cancer cell lines (Hela, MCF-7, and Hep3B) were maintained in RPMI 1640 or DMEM medium, supplemented with 10% FBS, l-glutamine (2 mM), penicillin G (100 UI/mL), and streptomycin (100 µg/mL). Stock solutions of compounds were prepared in DMSO, and cytotoxicity assays were carried out in 96-well microtiter plates against Hela, MCF-7, and Hep3B cancer cell lines (2 × 105 cells/mL) using MTT method. 16 After 72 hours of incubation at 37o C in air/CO2 (95:5) with or without test compounds, cell growth was estimated by colorimetric measurement of formazan. Optical density was determined at 540 nm with a Titertek Multiskan photometer. The IC50 value was defined as the concentration of a sample necessary to inhibit the cell growth to 50% of the control. Camptothecin was used as a reference compound.
Supplemental Material
Supplementary - Supplemental material for New Acetophenone and Cardanol Derivatives From Knema pachycarpa
Supplemental material, Supplementary, for New Acetophenone and Cardanol Derivatives From Knema pachycarpa by Tran Huu Giap, Ha Thi Thoa, Vu Thi Kim Oanh, Nguyen Thi Minh Hang, Nguyen Hai Dang, Dinh Ngoc Thuc, Nguyen Van Hung, and Le Nguyen Thanh in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Vietnam National Foundation for Science and Technology Development (NAFOSTED) under grant number 104.01-2017.47.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
