Abstract
Two new benzophenone derivatives (
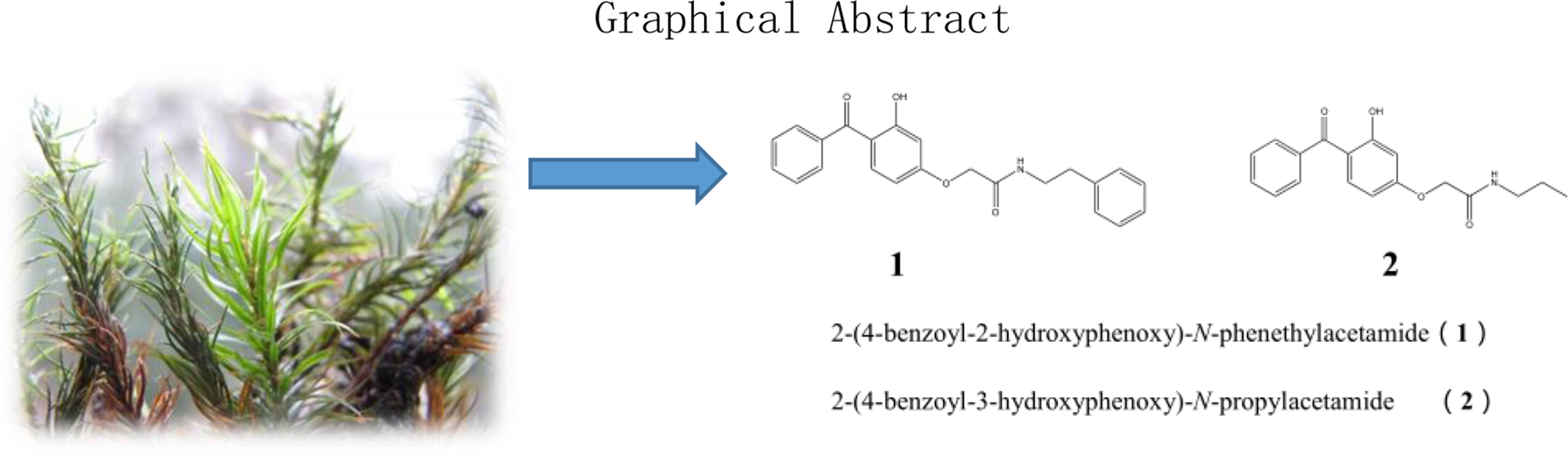
Mosses (Musci, Bryophyta) have relatively simple anatomical structures
1
and are currently represented by approximately 14 000 species that colonize diverse habitats.
2
The Polytrichaceae family are comprised of 17 genera and approximately 200 species,
3
of which the genera Polytrichum and Polytrichastrum are among the most familiar of all mosses, especially in boreal and temperate regions, due to their abundance, distinctiveness, and large size.
4
Previous phytochemical investigation of Polytrichaceae mosses has resulted in the isolation of benzonaphthoxanthenones,
5
-11
benzophenones,
12
cinnamoyl,
7
coumarin glucosides,
13
and flavonoids10,11with broad biological activities such as cytotoxicity,
5
-7,10,12
antioxidant,
9
anti-inflammatory,
14
tyrosine phosphatase 1B (PTP1B) inhibition,
8
and anti-neuroinflammatory activities.
11
During continuing chemical research of products from mosses, 2 new benzophenone derivatives (

The structures of compounds 1 and 2.
Results and Discussion
Compound
1H NMR (500 MHz) and 13C NMR (125 MHz) Spectroscopic Data for Compounds 1 and 2 in CDCl3 (δ in ppm, J in Hz).

The hydroxy resonance at δ
H 12.58 showed HMBC correlations to C-1 (δ
C 114.4), C-2 (δ
C 166.1), C-3 (δ
C 102.8), and C-4 (δ
C 163.3), indicating that HO-2 was ortho to the quaternary C-1 and meta to the oxygenated quaternary. The HMBC correlations of H-6 (δ
H 7.55) to C-1, C-7 (δ
C 200.3), and C-4; and of H-2' (and H-6') (δ
H 7.63) to C-7 connected a monosubstituted and a 1, 2, 4-trisubstituted aromatic rings to the keto carbonyl group to give a benzophenone core. Except for the other one monosubstituted aromatic ring, the remaining NMR signals were assigned to an amide side chain based on the COSY correlations (Figure 2) of N-H (δ
H 6.53)/H2-1"' (δ
H 3.62)/H2-2"' (δ
H 2.86) and HMBC correlations of H2-2" (δ
H 4.51) to C-1" (δ
C 167.1); of H2-1"' to C-1" and C-2"' (δ
C 35.7); and of H2-2"' to C-1"' (δ
C 40.3). The HMBC correlations of H-2" to C-4; of H2-1"' to C-3"' (δ
C 138.5); and of H2-2"' to C-3"' and C-4"' (and C-8"') (δ
C 128.9) suggested that the amide side chain was attached to C-4 via an oxygen atom, and a monosubstituted benzene ring was linked to C-2"' via a C-2"'/C-3"' single bond. Thus, the structure of compound

Key COSY and HMBC correlations of compounds 1 and 2.
Compound
The cytotoxicity of the 2 new compounds against HCT-116, A-549, HepG2 and HeLa human cancer cell lines were assessed, but no obvious cytotoxic activity was observed (IC50 >100 µM).
Experimental
General Experimental Procedures
Optical rotations were measured on a JASCO P-1020 polarimeter in MeOH at room temperature. IR spectra were recorded on a Bruker Tensor 27 spectrometer using KBr pellets, 1D and 2D NMR spectra on a Bruker AVIII 500 MHz spectrometer (Bruker, Karlsruhe, Germany) in CDCl3 with tetramethylsilane (TMS) as an internal standard, and high-resolution electrospray mass spectrometry (HRESIMS) on an Agilent 6529B Q-TOF instrument (Agilent Technologies, Santa Clara, CA, USA). Preparative HPLC was performed on a Prep-HTGF ODS column (21.2 mm × 250 mm i.d., 7 µm, Agilent Technologies, Santa Clara, CA, USA) with a flow rate of 20.0 mL/min. Silica gel (200, 300 mesh, Qingdao Haiyang Chemical Co., Ltd, Qingdao China), Sephadex LH-20 gel (Amersham Biosciences), MCI (Mitsubishi, Japan) and RP-C18 silica (40, 63 mm, FuJi, Japan) were used for column chromatography.
Plant Material
The endohydric moss, Polytrichastrum formosum (Hedw.) G. L. Sm, was collected from Cape Mount, Dujun city, Guizhou Province (latitude 26°35′42.8″N, longitude 107°38′32.1″E, altitude 1250 m), People’s Republic of China in November, 2019. A specimen (HBSD2019110401) was deposited in the College of Life Sciences, Hebei Normal University.
Extraction and Isolation
The air-dried whole plant (1.3 kg) of P. formosum was powdered and extracted with 95% EtOH by infusion (5 × 5 L, 3 days each). The concentrated exact (39 g) was suspended in H2O and partitioned with light petroleum, CH2Cl2, EtOAc, and n-BuOH, successively. The CH2Cl2 extract (7.5 g) was subjected to MCI gel column chromatography eluting with MeOH/H2O mixtures (30%, 60%-90%), and the 90% MeOH/H2O fraction (2.6 g) was chromatographed on a silica gel column eluting with light petroleum and acetone mixtures (10:1−5:1−3:1−3:2−1:1) to give 5 fractions, Fr.1−Fr.5. Fraction Fr.3 (650 mg) was separated on an ODS gel column (eluted with MeOH/H2O, 60%‐100% in a step gradient manner) to afford 5 fractions, Fr.3.1−Fr.3.5. Fraction Fr.3.2 (0.92 g) was separated by RP-C18 preparative HPLC with 60% ACN/H2O and the chromatographic peak at t
R = 19 minutes was collected and purified by Sephadex LH-20 CC (eluted with MeOH) to afford compound
2-(4-Benzoyl-2-Hydroxyphenoxy)-N-Phenethylacetamide (1)
White amorphous powder;
2-(4-Benzoyl-3-Hydroxyphenoxy)-N-Propylacetamide (2)
Colorless crystals (CDCl3);
Cytotoxicity
Cytotoxicity was measured using the sulforhodamine B method with human colon cancer (HCT-116), human lung carcinoma (A-549), liver hepatocellular carcinoma (HepG2), and human cervical carcinoma (HeLa)cell lines. The half-maximal inhibitory concentration value was analyzed using GraphPad Prism 5 software. Adriamycin was used as a positive control. 17
Conclusion and Discussion
Two new benzophenones, 2-(4-benzoyl-2-hydroxyphenoxy)-N-phenethylacetamide (
Supplemental Material
Supplementary material - Supplemental material for Two New Benzophenones From the Endohydric Moss Polytrichastrum formosum
Supplemental material, Supplementary material, for Two New Benzophenones From the Endohydric Moss Polytrichastrum formosum by Xu-Hong Duan, Xue-Wen Zhang, Meng Qin, Pei He, Lin Pei, Jian-Cheng Zhao and Yu-Ling Chen in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support forthe research, authorship, and/or publication of this article: This project was supported by the National Natural Sciences Foundation of China [grant number 31370237 and 31370184].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
