Abstract
In order to find out the influence of carboxylic acid functionalities in the N-lactam side chains of indenoisoquinolines on cytotoxic activities, several new compounds have been synthesized and structurally characterized by analytical and spectral methods. The incorporation of a carboxylic acid group into the lactam side chain of indenoisoquinolines results in differences in cytotoxicity. The results indicated that compound
Indenoisoquinolines are a class of non-camptothecin topoisomerase 1 poisons that display marked cytotoxic properties and , some of them, potent antitumor activities.
1,2
These highly fused compounds which contain a planar tetracyclic heteroring system equipped with multifarious functionalities as exemplified by the lead compound
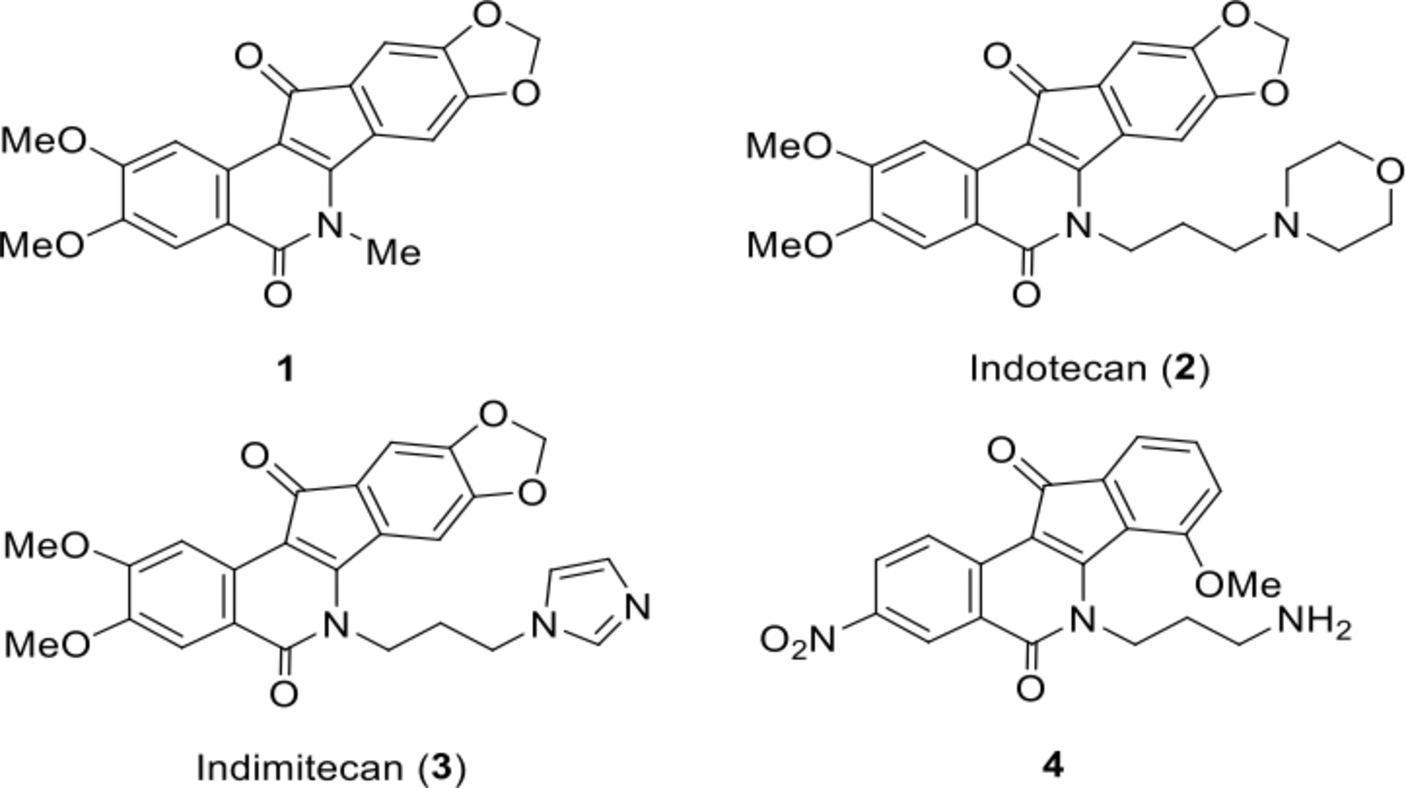
Chemical structure of several bioactive indenoisoquinolines.
Functionalized carboxylic acids are often part of biologically active agents. They might provide a point of attachment for the synthesis of prodrugs so that the pharmacokinetics could be modulated and optimized. Furthermore, the physiochemical properties of the carboxylate ion make it an attractive moiety for installation into drug candidates. For example, the ionization of a carboxylic acid, at physiological pH (7.4), improves its ability to hydrogen bond with neighboring water molecules, and thus may improve its overall water solubility. 10 The importance of the carboxylic acid functional group in drug design is illustrated by the fact that >450 marketed drugs are carboxylic acid containing molecules, including widely used nonsteroidal anti-inflammatory drugs, antibiotics, anticoagulants, and cholesterol-lowering statins. 10,11 A carboxylic acid group could possibly serve as a biologically active unit for incorporation with indenoisoquinoline skeleton in order to afford promising potent bioactive compounds. However, very little is known in the literature about the synthesis and bioactivities of indenoisoquinoline acids. 4,12,13 In that respect, this study presents the synthesis of a variety of novel indenoisoquinolines combining the indenoisoquinoline scaffold with the promising functionalized carboxylic acid moiety, linked together via the indenoisoquinoline lactam nitrogen, followed by a preliminary evaluation of the cytotoxic potential of these derivatives against two human cancer cell lines.
The synthesis of the indenoisoquinoline system has been performed by many methods in the literature. Several recently reported procedures are mostly based on (i) the oxidative cyclization of the cis-acid resulting from the condensation of homophthalic anhydrides and benzylidene Schiff bases, 14 (ii) the condensation of primary amines with the appropriately substituted indenoisochromenones, 14 (iii) the Suzuki-Miyaura cross-coupling followed by ring-closing metathesis, 15 and (iv) the cobalt-catalyzed dual annulation of o-halobenzaldimines with suitable alkynes. 16 The synthesis of the indenoisoquinolines was pursued in this work by applying the first method due to the ease in synthesis of intermediates and manipulation of starting materials to obtain indenoisoquinolines bearing the desired substituents.
In consideration of the chemical structure of indotecan (
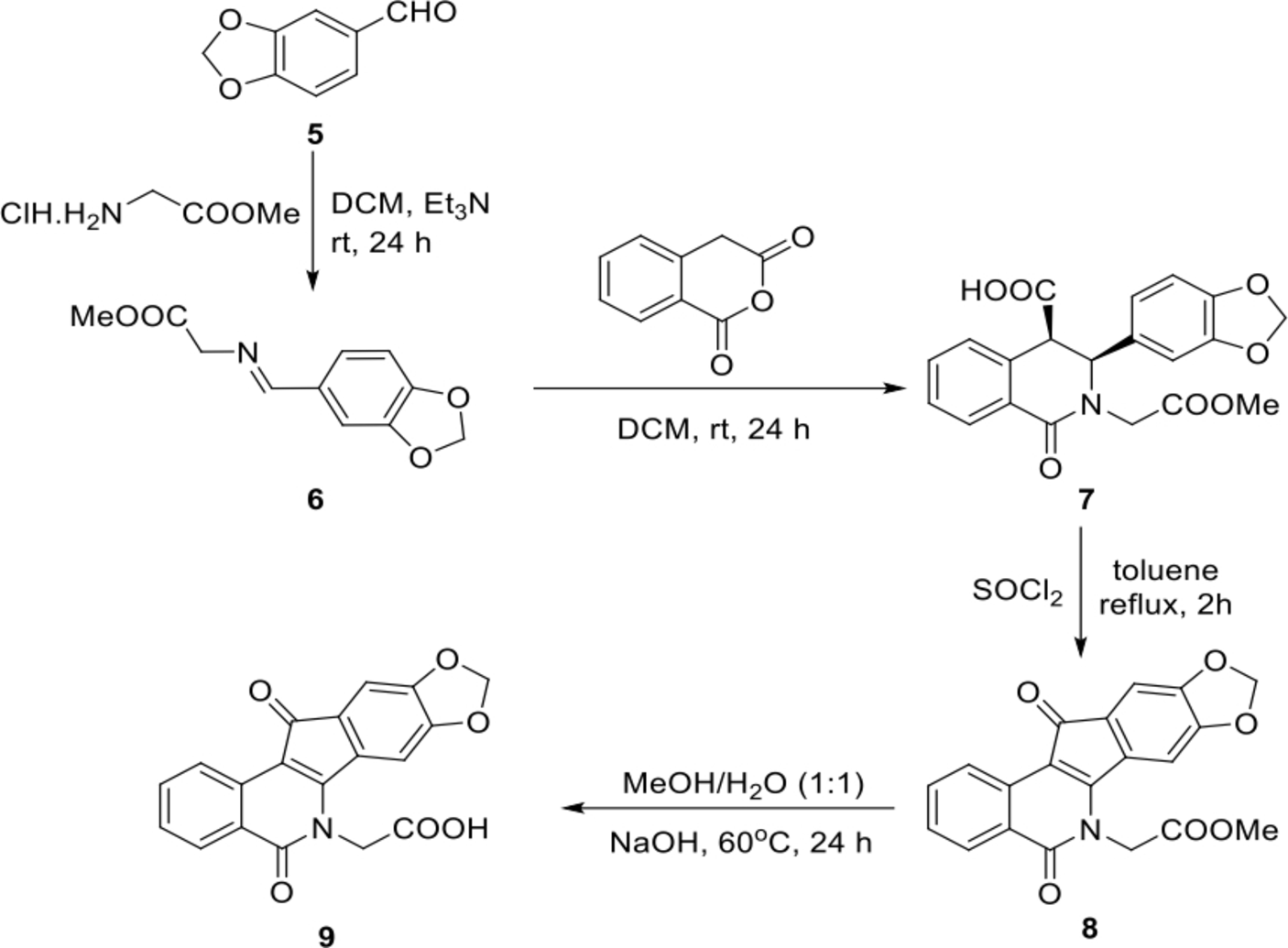
Synthesis of indenoisoquinoline acetic acid 9.
Analogously, the indenoisoquinoline acetic acids containing methoxy group in D ring were synthesized by procedure as illustrated in Scheme 2. The treatment of homophthalic acid anhydride with imine

Preparation of indenoisoquinoline acids 15 and 16.
Furthermore, several indenoisoquinolines containing carboxylic acid functionalized groups were synthesized starting from indenobenzopyran

Synthetic route for the preparation of indenoisoquinolines 18a to 18e.
In the next part of this work, all the synthesized compounds were subjected to in vitro biological assessment against two human cancer cell lines (KB, epidermoid carcinoma; HepG2, hepatoma carcinoma) in order to evaluate their potential as cytotoxic agents, and the results are summarized in Table 1. Ellipticine was used as positive control. The presence of carboxylic acid groups in the lactam side chain of indenoisoquinoline scaffold gives different cytotoxic effects of these compounds. In the case of D-ring without substituents (compounds
Synthesis and Cytotoxic Evaluation of Indenoisoquinoline Derivatives.

aIsolated yield.
In conclusion, a series of novel indenoisoquinolines containing carboxylic acid functional group in lactam side chain were designed and synthesized. Their biological activities were evaluated in vitro against KB and HepG2 cell lines. The results indicated that the presence of functionalized carboxylic acid group in indenoisoquinoline moiety had different effects for anticancer activities. In summary, compound
Experimental
General
Solvents and chemicals were obtained from commercial sources and used without further purification. Flash column chromatography was performed using silica gel (60 Å, particle size 40-60 µm). Thin-layer chromatographic analysis was performed on precoated silica gel GF254-slides and visualized by either UV irradiation or Ce(SO4)2 staining. 1H NMR spectra were recorded on a Bruker Avance (500 MHz) with samples dissolved in CDCl3 and data are reported in ppm with the solvent signal as reference (δ = 7.26 ppm for 1H NMR). IR spectra were measured on a Spectrum Two Perkin Elmer.
Preparation of Acid Compounds 7 and 12
A solution of imine derivative
Preparation of Indenoisoquinoline 8, 13, 14
The cis-acid
Methyl 2-(5,12-dioxo-5H-[1,3]dioxolo[4′,5′:5,6]indeno[1,2-c]iso-quinolin-6(12H)-yl)acetate (8)
Red violet solid. MP: 207°C to 208°C. Yield 60%.
IR (KBr) cm−1: 2913, 1743, 1667, 1610, 1580, 1549, 1497, 1477, 1431, 1375, 1304, 1271, 1229, 1072, 1033, 997, 979, 929, 830, 787.
1H NMR (CDCl3, 500 MHz) δ: 8.59 (1H, d, J = 8.0 Hz), 8.29 (1H, dd, J = 0.8 Hz, J = 8.3 Hz), 7.70 (1H, dt, J = 1.5 Hz, J = 7.5 Hz), 7.42 (1H, dt, J = 1.3 Hz, J = 7.3 Hz), 7.08 (1H, s), 6.77 (1H, s), 6.07 (2H, s), 5.26 (2H, s), 3.83 (3H, s).
13C NMR (CDCl3, 125 Hz) δ: 189.2, 168.0, 163.0, 154.7, 151.5, 149.3, 134.2, 132.5, 132.2, 130.2, 128.7, 126.8, 123.2, 122.6, 108.2, 105.7, 104.0, 102.7, 53.1, 45.5.
Methyl 2-(8-methoxy-5,11-dioxo-5H-indeno[1,2-c]isoquinolin-6(11H)-yl)acetate (13)
Orange solid. MP: 207°C to 208°C. Yield 40%.
IR (KBr) cm−1: 2922, 2851, 1740, 1696, 1658, 1611, 1576, 1550, 1500, 1467, 1419, 1368, 1322, 1274, 1221, 1082, 1036, 999, 979, 862, 784.
1H NMR (CDCl3, 500 MHz) δ: 8.69 (1H, d, J = 8.0 Hz), 8.35 to 8.3 (1H, m), 7.76 to 7.70 (1H, m), 7.56 to 7.52 (1H, m), 7.50 to 7.44 (1H, m), 6.83 (1H, d, J = 1.5 Hz), 6.71 (1H, dd, J = 2.0 Hz, J = 8.0 Hz), 5.30 (2H, s, H-1’), 3.85 (3H, s, H-18), 3.82 (3H, s).
13C NMR (CDCl3, 125 Hz) δ: 189.7, 168.2, 163.0, 158.1, 153.2, 139.2, 135.0, 134.1, 132.2, 128.8, 127.4, 123.7, 123.6, 119.0, 116.4, 114.6, 109.2, 56.1, 53.1, 45.6.
Methyl 2-(10-methoxy-5,11-dioxo-5H-indeno[1,2-c]isoquinolin-6(11H)-yl)acetate (14)
Orange solid. MP: 207°C to 208°C. Yield 41%.
IR (KBr) cm−1: 2953, 1741, 1693, 1652, 1613, 1579, 1554, 1499, 1474, 1443, 1414, 1275, 1185, 1157, 1070, 1047, 1014, 985, 873.
1H NMR (CDCl3, 500 MHz) δ: 8.76 (1H, d, J = 8.0 Hz), 8.35 (1H, dd, J = 0.8 Hz, J = 8.3 Hz), 7.74 (1H, dt, J = 1.3 Hz, J = 7.5 Hz), 7.48 (1H, dt, J = 0.8 Hz, J = 7.8 Hz), 7.37 (1H, m), 6.99 (1H, d, J = 9 Hz), 6.93 (1H, d, J = 7.5 Hz), 5.35 (2H, s), 4.00 (3H, s), 3.81 (3H, s).
13C NMR (CDCl3, 125 Hz) δ: 189.4, 168.2, 163.2, 157.5, 152.9, 139.6, 135.2, 134.2, 131.5, 128.7, 127.3, 123.7, 123.6, 119.1, 116.5, 114.5, 109.1, 56.2, 53.1, 45.8.
Preparation of Compounds 9, 15, 16
A respective solution of compound
2-(5,12-Dioxo-5,12-dihydro-6H-[1,3]dioxolo[4′,5′:5,6]indeno[1,2- c]isoquinolin-6-yl)acetic acid (9)
Violet solid. MP: 210°C to 211°C. Yield 92%.
IR (KBr) cm−1: 3427, 2960, 2928, 2861, 1727, 1649, 1462, 1377, 1273, 1123, 1072, 1036, 740, 708.
1H NMR (CDCl3, 500 MHz) δ: 8.49 (1H, d, J = 8.0 Hz), 8.17 (1H, d, J = 7.5 Hz), 7.81 (1H, m), 7.50 (1H, m), 7.17 (2H, s), 6,19 (2H, s), 5.27 (2H, s).
13C NMR (CDCl3, 125 Hz) δ: 188.8, 169.3, 162.2, 155.9, 151.3, 149.1, 134.4, 131.9, 131.8, 129.3, 128.2, 126.8, 122.2, 121.8, 106.1, 105.3, 104.9, 103.0, 45.7.
2-(8-Methoxy-5,11-dioxo-5,11-dihydro-6H-indeno[1,2-c]iso quinolin-6-yl)acetic acid (15)
Orange solid. MP: 208°C. Yield 95%.
IR (KBr) cm−1: 3442, 2956, 2929, 2865, 1728, 1606, 1549, 1502, 1465, 1420, 1368, 1280, 1178, 1123, 1073, 1036, 980, 864, 784, 747, 708, 639, 575.
1H NMR (CDCl3, 500 MHz) δ: 8.58 (1H, d, J = 8.0 Hz), 8.22 (1H, d, J = 8.0 Hz), 7.84 (1H, m), 7.57–7.50 (2H, m), 7.05 (1H, d, J = 1.5 Hz), 6.92 (1H, dd, J = 1.5 Hz, J = 8.0 Hz), 5.27 (2H, s), 3.83 (3H, s).
13C NMR (CDCl3, 125 Hz) δ: 189.1, 169.3, 163.6, 162.1, 154.4, 138.8, 134.2, 131.8, 128.2, 127.4, 126.3, 124.6, 122.7, 122.6, 112.5, 112.2, 108.0, 56.0, 45.8.
2-(10-Methoxy-5,11-dioxo-5,11-dihydro-6H-indeno[1,2-c]iso quinolin-6-yl)acetic acid (16)
Orange solid. MP: 209°C. Yield 96%.
IR (KBr) cm−1: 3441, 2929, 2959, 2860, 1728, 1654, 1608, 1550, 1462, 1382, 1278, 1124, 1070, 1040, 985, 946, 872, 799, 757.
1H NMR (CDCl3, 500 MHz) δ: 8.59 (1H, d, J = 8.0 Hz), 8.20 (1H, d, J = 7.5 Hz), 7.83 (1H, t, J = 7.5 Hz), 7.55–7.48 (2H, m), 7.20 (1H, d, J = 8.5 Hz), 7.15 (1H, d, J = 7.5 Hz), 5.23 (2H, s), 3.90 (3H, s).
13C NMR (CDCl3, 125 Hz) δ: 188.4, 169.2, 162.2, 156.9, 153.8, 138.7, 135.8, 134.1, 131.9, 128.1, 127.2, 122.8, 122.6, 117.9, 117.4, 115.8, 106.9, 55.9, 46.0.
Preparation of Indenoisoquinoline Acids 18a to 18e
To a solution of indenobenzopyran
2-(5,11-Dioxo-5,11-dihydro-6H-indeno[1,2-c]isoquinolin-6-yl)acetic acid (18a)
Orange solid. MP: 286°C to 287°C. Yield 88%.
IR (KBr) cm−1: 3414, 3022, 2929, 2848, 2850, 1737, 1695, 1645, 1606, 1573, 1546, 1498, 1456, 1417, 1379, 1313, 1240, 1192, 1099, 1080, 962, 750, 694.
1H NMR (CDCl3, 500 MHz) δ: 8.65 (1H, d, J = 8.0 Hz), 8.29 (1H, d, J = 8.0 Hz), 7.72 (1H, dt, J = 1.5 Hz, J = 8.0 Hz), 7.58 (1H, d, J = 6.5 Hz), 7.44 (1H, dt, J = 1.0 Hz, J = 8.0 Hz), 7.32 to 7.38 (3H, m), 5.27 (2H, s).
13C NMR (CDCl3, 125 Hz) δ: 190.4, 169.3, 163.4, 155.8, 137.1, 134.6, 134.2, 133.4, 132.4, 131.1, 128.4, 127.4, 123.5, 123.3, 123.1, 121.8, 108.9, 45.8.
(S)-2-(5,11-dioxo-5,11-dihydro-6H-indeno[1,2-c]isoquinolin-6-yl)-3-methylbutanoic acid (18b)
Red solid. MP: 168°C to 169°C. Yield 86%.
IR (KBr) cm−1: 3390, 2958, 2926, 2866, 2358, 1728, 1701, 1645, 1606, 1546, 1498, 1456, 1379, 1317, 1271, 1195, 1078, 1080, 993, 760, 700.
1H NMR (CDCl3, 500 MHz) δ: 8.45 (1H, d, J = 8.0 Hz), 8.00 (1H, d, J = 8.0 Hz), 7.57 (1H, d, J = 7.0 Hz), 7.50 (1H, t, J = 8.0 Hz), 7.17 to 7.44 (4H, m), 4.64 (1H, d, J = 9.5 Hz), 2.84 (1H, m), 1.13 (3H, d, J = 5.5 Hz), 0.51 (3H, d, J = 6.5 Hz).
(S)-2-(5,11-dioxo-5,11-dihydro-6H-indeno[1,2-c]isoquinolin-6-yl)-3-phenylpropanoic acid (18c)
Red solid. MP: 124°C to 125°C. Yield 85%.
IR (KBr) cm−1: 3385, 2956, 2924, 2854, 2358, 1726, 1697, 1647, 1604, 1570, 1544, 1498, 1450, 1411, 1379, 1197, 1024, 754, 700.
1H NMR (CDCl3, 500 MHz) δ: 8.51 (1H, d, J = 8.0 Hz), 8.16 (1H, d, J = 8.0 Hz), 7.62 (1H, J = 7.0 Hz), 7.31 to 7.36 (2H, m), 7.05 to 7.12 (3 H, m), 6.83 to 6.94 (5H, m), 5.33 (1H, m), 3.56 (1H, d, J = 7.0 Hz), 3.24 (1H, d, J = 1.5 Hz).
(S)-2-(5,11-Dioxo-5,11-dihydro-6H-indeno[1,2-c]isoquinolin-6-yl)-3-(4-hydroxyphenyl)propanoic acid (18d)
Red solid. MP: 183°C to 184°C. Yield 81%.
IR (KBr) cm−1: 3373, 2918, 2850, 1691, 1654, 1600, 1546, 1514, 1498, 1458, 1411, 1379, 1246, 1201, 1022, 813, 758, 694.
1H NMR (CDCl3, 500 MHz) δ: 8.53 (1H, d, J = 8.0 Hz), 8.16 (1H, d, J = 7.5 Hz), 7.60 (1H, t, J = 7.5, J = 8.0 Hz), 7.47 (1H, t, J = 7.5 Hz), 7.33 (1H, d, J = 7.0 Hz), 7.18 (1H, d, J = 7.0 Hz), 7.08 (1H, t, J = 7.5 Hz), 7.00 (1H, t, J = 7.5 Hz), 6.63 (2H, d, J = 8.0 Hz), 6.30 (2H, J = 8.0 Hz), 5.30 (1H, m), 5.30 (1H, m), 3.60 (1H, d, J = 10.0 Hz), 3.48 (1H, d, J = 11.0 Hz).
(S)-3-(3,4-Dihydroxyphenyl)-2-(5,11-dioxo-5,11-dihydro-6H-indeno [1,2-c]isoquinolin-6-yl)propanoic acid (18e)
Red solid. MP: 188°C to 189°C. Yield 80%.
1H NMR (CDCl3, 500 MHz) δ: 8.62 (1H, d, J = 8.0 Hz), 8.29 (1H, d, J = 8.0 Hz), 7.56 (1H, t, J = 8.0 Hz), 7.50 to 7.52 (2H, m), 7.42 (1H, d, J = 7.0 Hz), 7.28 to 7.41 (2H, m), 6.43 (1H, d, J = 2.0 Hz), 6.37 (1H, d, J = 8.0 Hz), 6.30 (1H, dd, J = 2.0, J = 8.0 Hz) 5.69 (1H, dd, J = 5.0, J = 10.0 Hz), 3.57 (2H, m).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are indebted to the Vietnamese National Foundation for Science and Technology Development (NAFOSTED, code: 104.01-2017.27) for financial support.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
