Abstract
For enantiomer separation of a variety of chiral compounds, a number of chiral selectors have been developed and applied. Among these chiral selectors are chiral crown ethers, a class of synthetic polyether molecules that bind protonated chiral primary amines with high selectivity. This article focuses on enantiodiscrimination using (18-crown-6)-2,3,11,12-tetracarboxylic acid as a crown ether-type chiral solvating agent for nuclear magnetic resonance spectroscopic method in several chirotechnologies.
Keywords
For enantiodiscrimination in chirotechnology, the nuclear magnetic resonance (NMR) spectroscopic technique has been investigated and employed since 1970. 1,2 There are two approaches of analytical techniques for the determination of enantiomer composition by NMR spectroscopy: (1) the use of chiral derivatizing agents (CDAs) such as the Mosher’s reagent (α-methoxy-α-trifluoromethylphenylacetic acid; MTPA) and (2) the use of chiral solvating agents (CSAs) such as 1-(9-anthryl)-2,2,2-trifluoroethanol or lanthanide chiral shift reagents (CSRs) such as europium derivatives. 1,3 The first NMR technique as an indirect method is the analysis of covalent diastereomer mixtures prepared using CDAs like Mosher’s reagent (MTPA). For the determination of enantiomer composition, the corresponding diastereomer composition after derivatization of the analyte enantiomers with proper CDAs is measured by NMR spectroscopy. Prior to NMR analysis, therefore, permanent diastereomers derivatized using MTPA reagent with the analyte enantiomers should be prepared. Then chemical shift differences between the two diastereomers obtained from an enantiomeric sample are required for the measurement of enantiomer purity by 1H or 19F NMR spectroscopy. 3 However, this NMR technique has potential problems of kinetic resolution or racemization during derivatization reaction, although excess of the CDA may be used to overcome kinetic resolution. 2 And the CDA reagent must be enantiomerically pure. On the contrary, the second NMR technique using CSA or CSR is a direct convenient analytical method with the advantage that there is no need for derivatization process before NMR experiments. These reagents form transient diastereomeric complexes in situ with the analyte enantiomers in NMR solution. Under chiral environments in the presence of CSA or CSR with the analyte enantiomers in NMR solution, their enantiotopic groups become the corresponding diastereotopic moieties for spectral anisochrony (chemical shift difference), which provides information about the enantiomer composition. In comparison with CDAs, therefore, the second NMR technique is a very convenient analytical method without the possibility of kinetic resolution or racemization. 1 In particular, the NMR technique using CSAs is much more effective for enantiodiscrimination because high-field NMR spectrophotometers are detrimental to the application of lanthanide CSRs due to line broadening phenomenon. 2 This review focuses on enantiodiscrimination using chiral crown ethers as CSAs using NMR spectroscopy in chirotechnology.
Crown ethers are synthetic macrocyclic polyether molecules and they can bind protonated primary amines with high selectivity and affinity. Cram, a pioneer of the “host-guest chemistry,” first introduced several chiral crown ethers for the enantiomer separation of amino acids. 4 Among several investigated chiral crown ethers, l-tartaric acid-derived (+)-(18-crown-6)-2,3,11,12-tetracarboxylic acid (18-C-6-TA) prepared by Lehn has been usefully applied as a chiral selector for the separation of the enantiomers of chiral primary amines including amino acids (Figure 1) . 5,6
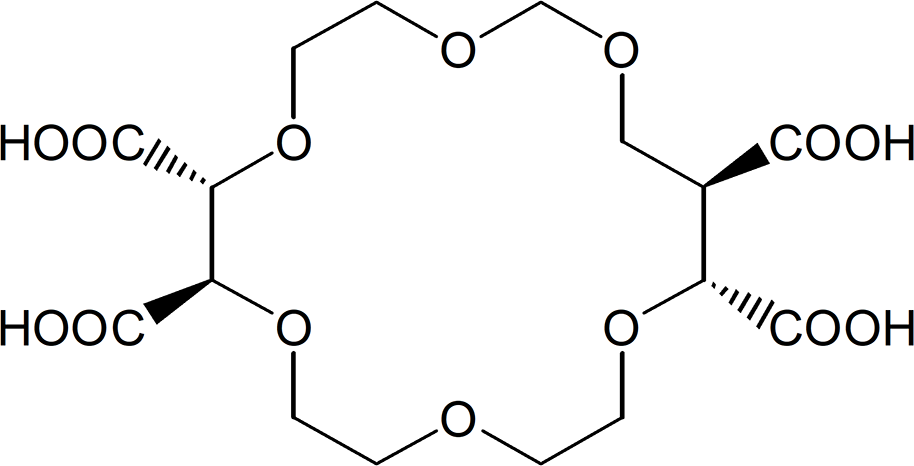
The structure of (+)−(18-crown-6)-2,3,11,12-tetracarboxylic acid of chiral crown ether for chiral solvating agent.
The Kuhn group reported the chiral capillary electrophoresis (CE) method using the crown ether, (+)-18-C-6-TA, as a chiral selector for the enantiomer resolution of several α-amino acids. 7,8 Since then, this chiral crown ether has been employed as an effective chiral selector for the enantiomer separation of various chiral amine-containing pharmaceutical compounds in CE. 9,10 In our group, we employed 18-C-6-TA to perform the enantiomer resolution of several aromatic amino acids and their esters in chiral CE. 11,12 This report is the first to conduct enantiomer separation using (+)- or (−)−18-C-6-TA to ensure the exactly opposite migration order of the analytes by crosschecking the two migration times in CE. In addition, when the enantiomers of 8 β-blockers of amino alcohols were simultaneously resolved by CE, the additional use of (+)- or (−)-18-C-6-TA to dimethyl-β-cyclodextrin (DM-β-CD) as dual chiral additives rather than the use of only DM-β-CD as a single chiral selector was applied for better results of enantioseparation. 13 The use of DM-β-CD and (+)-18-C-6-TA as dual chiral selectors resulted in better simultaneous enantiomer separation of 8 β-blockers by CE. Furthermore, the capillary electrochromatography (CEC) technique, an analytical method that combines the advantages of both CE and high-performance liquid chromatography (HPLC), was applied for enantioseparation of α-amino acids and their derivatives using the chiral stationary phase (CSP) derived from 18-C-6-TA. 14,15 On the other hand, we reported the development of (+)-18-C-6-TA-derived CSP for chiral HPLC and its application for resolution of the enantiomers of several α-amino acids and primary amino compounds. 16-23 This chiral crown ether-type 18-C-6-TA-derived CSP in HPLC has been usefully employed for the enantioseparation of various chiral primary amines including amino acid-type compounds. Currently, the chiral crown ether covalently bonded-type CSPs are commercially available as ChiroSil RCA(+) and ChiroSil SCA(−) derived from (+)- and (−)-18-C-6-TA (RS Tech Corp., Daejeon, Korea), respectively. Either (+)- or (−)-18-C-6-TA-derived CSP can be used to invert the elution order as desired for the exact determination of the enantiomeric purity. 21
Because the chiral crown ether of (+)- or (−)-18-C-6-TA as a chiral selector was successfully applied not only in CE or CEC but also in HPLC for enantiomer analysis, we expected that 18-C-6-TA could be useful as a CSA for enantiodiscrimination of these analytes in solution NMR. Therefore, when (+)-18-C-6-TA as a CSA was used in NMR experiments, all the investigated α-amino acids showed high chemical shift nonequivalences (ΔΔδ = 0.10-0.29) of their α-protons, as shown in Table 1. 24-27
1H NMR Data for the Enantiomer Discriminations of Several Amino Acids.

NMR, nuclear magnetic resonance;
aChemical shift nonequivalences ([ΔΔδ(d-l)], ppm) were obtained by subtracting l-isomer values from d-isomer values of α-protons after complexation. Chemical shifts were determined from the spectra of racemic analytes (2 mM) in the presence of 1 equivalent of 18-C-6-TA.
b,cTwo and 4 equivalents of 18-C-6-TA were used, respectively.
It was shown that 18-C-6-TA successfully enantiodiscriminated all these analytes by NMR spectroscopy. All α-proton chemical shifts of d- and l-isomers in the presence of (+)-18-C-6-TA were moved in the downfield shift direction compared to the free state. Also, all α-proton chemical shift nonequivalences [ΔΔδ(d-l)] observed from the investigated d-isomers in the presence of (+)-18-C-6-TA were greater than those of the l-isomers upon chiral complexation with (+)-18-C-6-TA. This indicates that the d-isomers interact more strongly with the CSA (+)-18-C-6-TA than the l-isomers. 24 As the stoichiometric ratio of 18-C-6-TA to analytes was increased, the chemical shift nonequivalences of α-protons of analytes gradually increased. 24-26 In the case of lysine and threonine in Table 1, 2 or 4 equivalents of 18-C-6-TA were used, because their α-proton peaks in the presence of equimolecular amounts of 18-C-6-TA overlapped with other NMR peaks. The magnitude of chemical shift nonequivalences observed in NMR was not directly related to the enantioselectivities observed from HPLC, because the chemical shift nonequivalence in NMR depends directly on the structural changes resulting from complexation between the chiral selector and each enantiomer. 24,25 Smaller chemical shift differences in the two enantiomers of amino acids were shown than those in their corresponding methyl ester derivatives, although amino acids usually showed larger enantioselectivities than their corresponding methyl esters in chiral HPLC. 27 In addition, the enantiomers of phenylalanine methyl ester and leucine methyl ester were not resolved by chiral HPLC using 18-C-6-TA-derived CSP, whereas they showed high chemical shift nonequivalences (0.29 and 0.16 ppm) of their α-protons in the presence of 18-C-6-TA as determined by NMR, respectively. 16,27 These results indicate that NMR analytical method using 18-C-6-TA as a CSA could be complementary to the HPLC analytical technique using 18-C-6-TA-derived CSP. The chiral discrimination of the analytes in the presence of 18-C-6-TA is influenced by the side chains of amino acids. Therefore, the enantiodiscriminations of analytes with aromatic side chain, charged group, or other hetero nuclei moiety on their stereogenic centers were more effective than those of analytes with alkyl chains, showing larger chemical shift differences. As shown in Figure 2, the chemical shift nonequivalence [ΔΔδ(d-l)] of the α-proton of phenylalanine is 0.21 ppm, whereas those of leucine, serine, and alanine are 0.10, 0.15, and 0.10 ppm, respectively (Table 1).

1H NMR spectra of various racemic compounds in the presence of 1 equivalent of 18-C-6-TA; (a) Phe, (b) Leu, (c) Ser, and (d) Ala. All α-proton regions of the racemic compounds were annotated. NMR, nuclear magnetic resonance.
The NMR analytical method using 13-C-6-TA was usefully applied to the determinations of the enantiomeric purities of these analytes. As examples, 1H NMR experiments of the α- or β-proton of enantiomerically enriched phenylglycine (d:l = 1:10) and diphenylalanine analytes (d:l = 20:1 and 1:15) in the presence of (+)-18-C-6-TA were performed. 25,27 The d/l integration ratios of the α- or β-proton peak of these enantiomerically enriched analytes in the NMR spectra indicate the enantiomeric purities of the corresponding analytes. In the search for the origin of the chiral recognition mechanism of α-amino acid enantiomers in the presence of 18-C-6-TA as a chiral selector, detailed studies for the elucidation of noncovalent chiral interactions in solution were performed. Based on the data obtained by molecular dynamics calculations together with nuclear Overhauser effect information, a plausible chiral recognition mechanism between the (d)- or (l)-phenylglycine enantiomer and (+)-18-C-6-TA was proposed. 24 These results showed that the ammonium moiety held inside the cavity is bound by 3 +NH —O hydrogen bonds in a tripod arrangement; hence chiral discrimination is achieved by secondary lateral interactions between the substituents (the carboxylic acid moiety and the polyether ring) on 18-C-6-TA and the analyte (the carbonyl group and the phenyl moiety, which are hydrogen bonding and hydrophobic interactions, respectively). Among several chiral analytical methods, therefore, the NMR technique has a strong advantage for elucidation of chiral recognition mechanism in the solution state between each enantiomer and chiral selector as a CSA. Independently, Wenzel and Machida reported 1H NMR results of their chiral discrimination using (+)-18-C-6-TA for chiral primary amines using NMR spectroscopy. 28-30 For the determination of enantiomeric purity of these analytes, the NMR analytical method using 18-C-6-TA as a CSA shows lower precision, accuracy, and sensitivity than the HPLC analytical method using 18-C-6-TA-derived chiral column. 21,27 However, owing to the advantages due to the practical simplicity of this method, it is expected that this NMR analytical method using 18-C-6-TA as a CSA can be practical and useful for determinations of the enantiomeric purities of chiral primary amines such as amino acids and their analogs including hydrophobic compounds. In particular, it should be emphasized that the NMR method in chirotechnology can be employed as the most powerful technique for elucidation of related chiral recognition mechanisms.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Ministry of Science, ICT & Future Planning (2015R1A4A1041219 and 2017R1A2B2008483).
