Abstract
Six metabolites (
Endophytes are defined as microorganisms that inhabit internal tissues of plants without causing any apparent disease symptoms. 1–4 They live with their host in a mutualistic relationship during at least a part of their life span and are thought to protect their host against pathogens. 5 Endophytic fungi are also considered to be a pivotal and prolific source of bioactive secondary metabolites with promising medicinal, agricultural, or industrial applications. 6
Psidium guajava (family Myrtacea) is a fruit tree wildly distributed throughout the tropical and subtropical areas. Its leaves are used as antidiabetic, hepatoprotective, antioxidant, hemostatic, and anti-inflammatory agents in folk medicine. 7 Chemical investigations on the leaves of P. guajava led to the isolation of several sesquiterpene- and monoterpene-based meroterpenoids with unprecedented skeletons. 8 However, little is known about secondary metabolites of endophytes harbored inside the healthy tissues of P. guajava.
During our ongoing search for new biologically active compounds from fungi,
9–12
we isolated the endophyte Alternaria atrans MP-7 from the leaves of P. guajava. In the present study, a new fusaric acid derivative, atransfusarin (
Fractionation of the chloroform soluble extracts of culture of A. atrans MP-7 grown on solid rice medium by multiple Sephadex LH-20 and MCI column chromatography (CC) yielded one new fusaric acid (
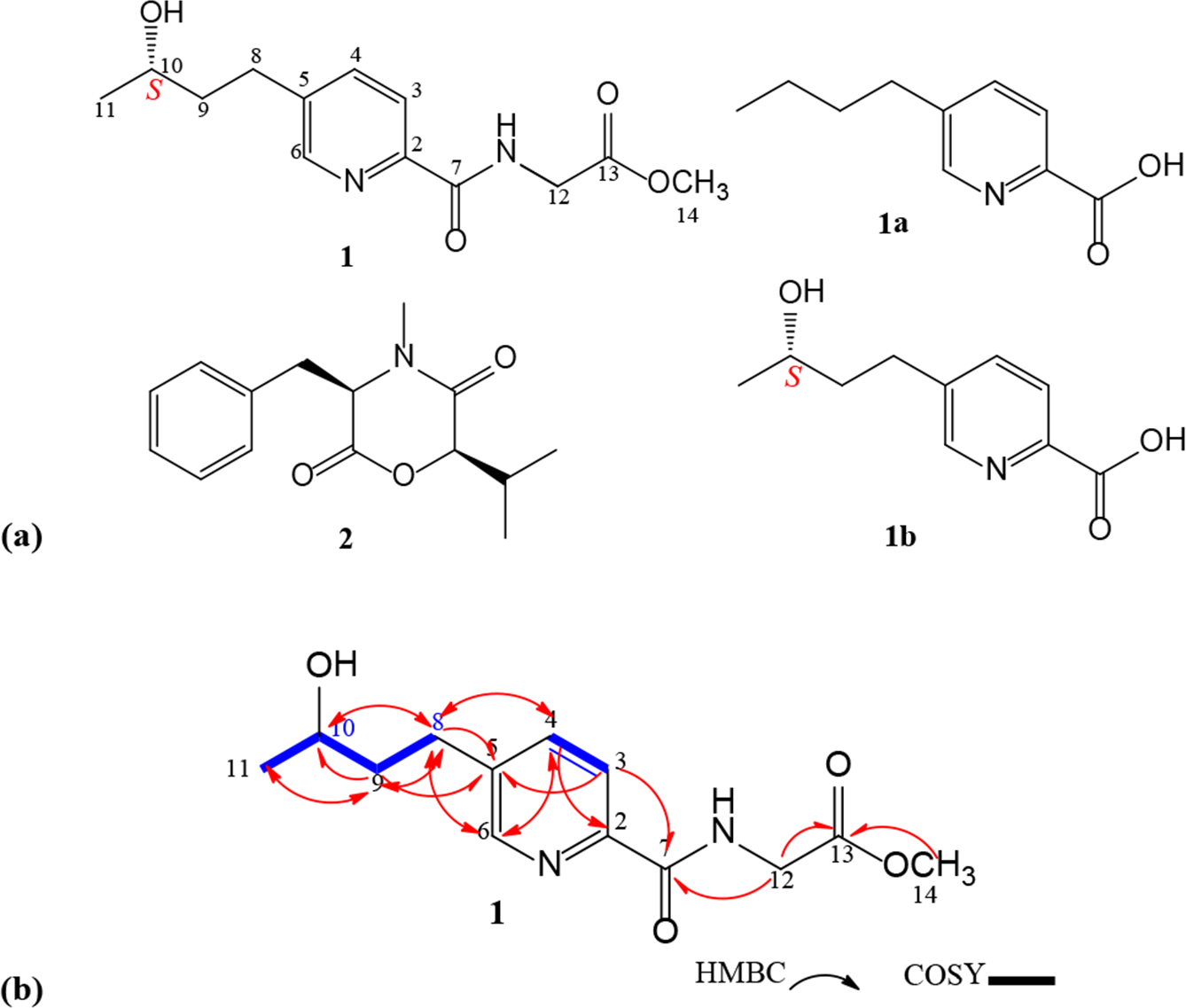
(a) Structures of compounds 1, fusaric acid (1a), (S)-(+)-fusarinolic acid (1b), and 2; (b) Key HMBC and COSY correlations of compound 1.
Compound
Furthermore, 1 oxygenated methine at δH 3.83 (1H, m, H-10); 2 methyl groups at δH 1.25 (3H, d, J = 5.3 Hz, H3-11) and 3.79 (3H, s, 14-OMe); and 3 methylene groups at δH 1.77 (2H, d, J = 6.4 Hz, H2-9), 2.73–2.86 (2H, m, H2-8), and 4.26 (2H, d, J = 5.3 Hz, H2-12) were observed (Table 1). The COSY correlations of H2-8/H2-9/H-10/H3-11 (Figure 1(b)), together with the HMBC correlations from H2-8/C-5, C-10, H2-9/C-8 and C-10, and H3-11/C-10, and H-4 and H-6/C-8 indicated a hydroxy-containing butyl moiety at C-5 and the OH group attached to C-10. Further HMBC correlations of H2-12/C-7 and C-13, H3-14/C-13 suggested a methyl acetate moiety linked to C-7 via an NH group (Figure 1(b)). From the above data, the structure of
1D and 2D NMR Data of Compound 1 (CDCl3).

The absolute configuration of the chiral center C-10 in
All isolated compounds (
Antifungal Activity of Compounds (1–6).
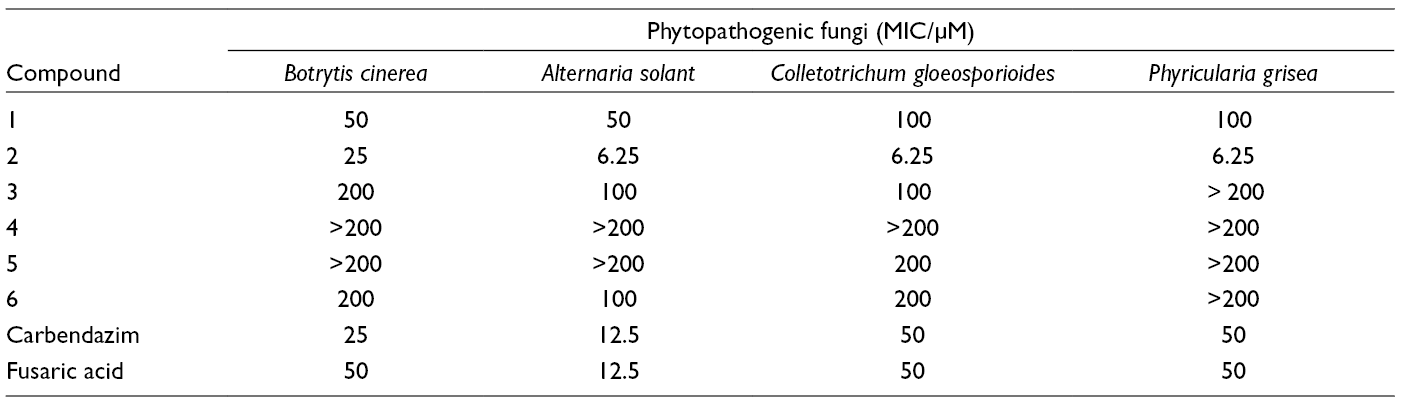
In this study, 6 compounds were purified from an endophytic A. atrans MP-7, in which compound
Experimental
General
Optical rotations were measured on a Rudolph Autopol III automatic polarimeter (Rudolph Research Analytical, Hackettstown, New Jersey). UV spectra were obtained on an UV−vis Thermo Evolution 300 spectrometer (Thermo Scientific, Waltham, Massachusetts). IR spectra were measured with a Bruker TENSOR 27 spectrophotometer (BrukerOptics, Ettlingen, Germany) in KBr pellets. ESIMS data were obtained on a Thermo Fisher LTQ Fleet instrument spectrometer (Thermo Scientific, MA, USA). HRESIMS was performed on a VG Autospec-3000 spectrometer (VG, Manchester, UK). NMR spectra were recorded on a Bruker Avance Ⅲ 500 spectrometer (Bruker BioSpin, Rheinstetten, Germany), δ in ppm rel. to trimethylsilane as internal standard, J in Hz. High performance liquid chromatography analysis and semipreparation was performed on a Waters 1525 instrument (Waters Corp., USA). Column chromatographywas conducted on silica gel (SiO2: 200–300 mesh, Qingdao Marine Chemical Group, Co., Qingdao, China), Sephadex LH-20 (Amersham Pharmacia Biotech, Uppsala, Sweden), MCI Gel (Mitsubishi Chemical Corp., Tokyo, Japan), and Lichroprep RP-18 gel (40, 63 µm, Merck, Germany). GF254 plates (Qingdao Marine Chemical Inc., Qingdao, China) were used for thin-layer chromatography (TLC). The fractions were monitored by TLC, and spots were visualized by heating silica gel plates sprayed with 5% H2SO4 in EtOH.
Fungal Material
The stems and leaves of P. guajava were collected from Nanning city, Guangxi Province of China, in August 2011, and authenticated by Mr Z.-H. Wu. A voucher specimen (0095460) was deposited at the herbarium of the Northwest A&F University. The endophytic fungal strain MP-7 was isolated under aseptic conditions from the healthy leaves of P. guajava.
The leaves were washed twice with sterile, refined water and dried, followed by surface sterilization using 70% ethanol (2 × 1 min, each) under the flow hood. After drying, the disinfected leaves were dissected into small pieces of 0.5 cm length and placed on a potato dextrose agar (PDA) plate (containing 0.005% Rose bengal and 0.01% kanamycin) followed by incubation for several days at 25°C. Strain MP-7, grown on the plate, was isolated and maintained on another PDA plate.
The strain was identified according to the phylogenetic taxonomy with sequence alignment of internal transcribed spacer and had a genetic closeness of 100% to the A. atrans strain. The strain was thus defined as A. atrans MP-7 and kept in one of the authors’ lab.
Extraction and Isolation
The fungal strain was cultivated axenically on solid rice medium, prepared by autoclaving 40 g of rice and 60 mL of water in a 500 mL Erlenmeyer flask. Fermentation was carried out in 200 Erlenmeyer flasks for 28 days at 25°C under static conditions. After 28 days of fermentation, the fungal culture in each flask was exhaustively extracted with MeOH overnight (3 × 500 mL) followed by filtration and solvent evaporation under reduced pressure. The obtained crude extract was dissolved in 90% MeOH and defatted with petroleum ether to give a crude MeOH extract, which was dissolved in 50% aq. MeOH and partitioned with chloroform. The chloroform soluble portion (45.3 g) was subjected to a silica gel column eluted with CHCl3-MeOH (100:0, 50:1, 20:1, 10:1, 0:100, v/v), providing 5 fractions 1–5.
Fraction 1 (2.3 g) was subjected to MCI gel (30% MeOH-H2O), followed by purification with Sephadex LH-20 (MeOH) CC, to yield compounds
Brine Shrimp Bioassay
Metabolites were tested in vitro for the brine shrimp (A. salina) bioassay, processed according to the previously identified method. 17,18 The final concentrations of the tested compounds were 10, 50, and 100 µg/mL. Toosendanin was used as the positive control and DMSO as the negative control. The lethality of each concentration was recorded.
Antifungal Bioassay
The test phytopathogenic fungi used in this study were B. cinerea, A. solani, C. gloeosporioides, and P. grisea (Cooke) Sacc. All the fungi were isolated from infected plant organs at the Northwest A&F University.
Antifungal activity was assessed by the microbroth dilution method in 96-well culture plates using a PD medium. 3,19,20 The test compounds were made up to 2 mg/mL in DMSO. Carbendazim (Aladdin Chemistry Co., Ltd, Shanghai, China), a commercial fungicide, was used as a positive control, and the solution of equal concentration of DMSO was used as a negative control. The tested fungi were incubated in the PD medium for 18 hours at 28°C ± 0.5°C at 150 rpm, and spores of different microorganism concentrations were diluted to approximately 1 × 106 CFU with PD medium. Test compounds (10 µL) were added to 96-well microplates, and 90 µL of PD medium was added. Serial dilutions were made in the 96-well round-bottom sterile plates in triplicate in 50 µL of PD medium, and then 50 µL of the fungal suspension was added. After incubation for 48 hours at 28°C ± 0.5°C, MIC was taken as the lowest concentration of the test compounds in the wells of the 96-well plate in which no microbial growth could be observed.
Atransfusarin (1)
Colorless crystal.
[α]D 25 +5.2 (c 0.10, MeOH).
UV (MeOH) λmax: 245 nm.
IR (KBr) υmax: 3442, 1744, 1663, 1530 cm–1.
1H (500 MHz) and 13C NMR (125 MHz): Table 1.
HRESIMS (positive): m/z 267.1325 [M + H]+ (calcd for C13H19N2O4, 267.1339), 289.1155 [M + Na]+, 555.2447 [2M + Na]+.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Program of Unified Planning Innovation Engineering of Science & Technology in Shaanxi Province (No.2015KTCQ02-14) and the Natural Science Foundation of Shaanxi Province (2014JZ2-001).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
