Abstract
A panel of experts in the use of continuous glucose monitoring (CGM) data in the treatment of diabetes met in Burlingame, California on October 27, 2025 to discuss the utility of the glycemia risk index (GRI) for clinical care research and population health management. The GRI composite metric is a single number (on a 0-100 percentile scale—lower is better) based on an expert-determined weighting of the seven individual components in the existing ambulatory glucose profile (AGP). The GRI describes the quality of glycemia based on glucose values collected in a 14-day CGM tracing, thus providing additional insights into CGM profiles beyond the AGP. During the meeting, the mathematical derivation of the GRI metric was presented along with its use for adult and pediatric individuals with diabetes and cancer who require medications that can adversely affect the glucose concentration. Examples where the GRI provided useful insights into the quality of CGM tracings were also discussed by the expert panel. In addition, a new smartphone application, the GRI Calculator, was presented. This app calculates the GRI of a CGM tracing and provides visualization of sequential CGM tracings for a specific individual. The GRI provides a reference measurement for the accuracy of artificial intelligence (AI) models assigning levels of glycemic quality to CGM tracings intended to match the assessments of clinicians. The GRI is now part of the data visualization panel for the Integration of Connected Diabetes Device Data into the Electronic Health Record (iCoDE-2) project, which standardizes both CGM and insulin dosing data. Further exploration of the potential value of the GRI for non-insulin users needs to be undertaken. The panel unanimously recommended that CGM manufacturers and developers of data visualization software for CGMs add the GRI to their data platforms for insulin users.
Keywords
Introduction
Diabetes Technology Society (DTS) hosted a meeting on the glycemia risk index (GRI), a composite metric that describes the quality of glycemia, on October 27, 2025, in Burlingame, CA, USA, prior to the 2025 Diabetes Technology Meeting (DTM). The event brought together 44 international panelists from adult endocrinology, pediatric endocrinology, primary care, diabetes care and education, epidemiology, and engineering, as well as industry observers from four of the largest manufacturers of continuous glucose monitoring (CGM) systems. The aim of the event was to discuss the status of use of the GRI for clinical and research purposes, as well as its potential value to be added to a CGM manufacturer’s data platform.
The GRI describes the quality of glycemia in a 14-day CGM system by incorporating the seven metrics of an ambulatory glucose profile (AGP) report (Table 1). These metrics are weighted according to the opinions of 330 experienced international healthcare professionals who were experienced in the interpretation of CGM data. Use of time in range (TIR) or time in tight range (TITR) reflects overall glycemic control but these metrics do not differentiate between values that are mildly versus substantially outside the target range (70-180 mg/dL). Originally described by Klonoff et al 1 in 2022, the GRI was developed to quantify clinician judgment, provide actionable scores of the risk of hypoglycemia and hyperglycemia, and provide graphical display (called the GRI Grid) that could be used by clinicians and researchers to determine the glycemic effects of prescribed and investigational treatments. Although multiple metrics describe various features of CGM tracings, the GRI is the only composite statistic that accounts for all seven of the metrics presented in an AGP report ranked according to the opinions of an international panel of international experts in interpreting CGM tracings.
The Seven Metrics of an Ambulatory Glucose Profile Report.

Abbreviations: CV, coefficient of variation.
Mathematical Derivation of the GRI
Michael Kohn, MD, MPP (UCSF, San Francisco, CA), first reviewed the derivation of the GRI. This metric was designed to be a single number that describes the quality of glycemia by incorporating the frequency and magnitude of hypoglycemia and hyperglycemia. To capture expert clinician opinion into a statistical model, 225 fourteen-day CGM tracings from users with type 1 diabetes (T1D) on automated insulin delivery, T1D on open-loop control systems, T1D using multiple daily injections (MDIs) of insulin, and adults with type 2 diabetes (T2D) on MDIs of insulin were reviewed and ranked by 330 international clinicians experienced in reading CGM reports. Each tracing was reviewed by 22 of these clinicians. Clinician rankings were then converted into percentile scores. Using best-fit regression analysis, the weight for each metric was calculated. Finally, a model was developed that simplified the seven coefficients into a formula using four of the seven metrics. 1
Principal component analysis (PCA) revealed that two dominant dimensions of the tracings, hypoglycemia and hyperglycemia, explained most of the variability in clinician rankings. Subsequent regression modeling identified four AGP metrics that best predicted the expert clinician consensus. These were % Very Low (VLow) (<54 mg/dL), % Low (54-<70 mg/dL), % High (>180-250 mg/dL), and % Very High (VHigh) (>250 mg/dL). The final derived equation for the GRI was GRI = 3.0 (%VLow) + 2.4 (%Low) + 1.6 (%VHigh) + 0.8 (%High). This model achieved an R2 value of .904 with a root mean square error (RMSE) of 8.95, indicating a high agreement between the model and clinician rankings (Figure 1a). The use of a combination of hypoglycemia and hyperglycemia was deemed consistent with engineering principles, where the best way to optimize a phenomenon with two components (eg, glycemia) is to identify the two adverse components (such as hypoglycemia and hyperglycemia) and then decrease both factors as much as possible. A low GRI score reflects low levels of both factors and is the optimal pairing. A high GRI score reflects a combination of high values of both factors and is a greater risk combination. The score is presented on a scale of 0 to 100 percentile points. A GRI report consists of four boxes—one for the percentage of time spent in each of the four out-of-range glucose values (the overall calculated GRI score), and a figure presenting the exposure (risk) to hypoglycemia and hyperglycemia. It should be noted that the two percentages of times below range used to calculate the GRI do not reflect the number of episodes of hypoglycemia that a patient experienced, but rather reflect the percentage of time spent in mild or severe hypoglycemia, just as is the case with the two hypoglycemic metrics in an AGP report.
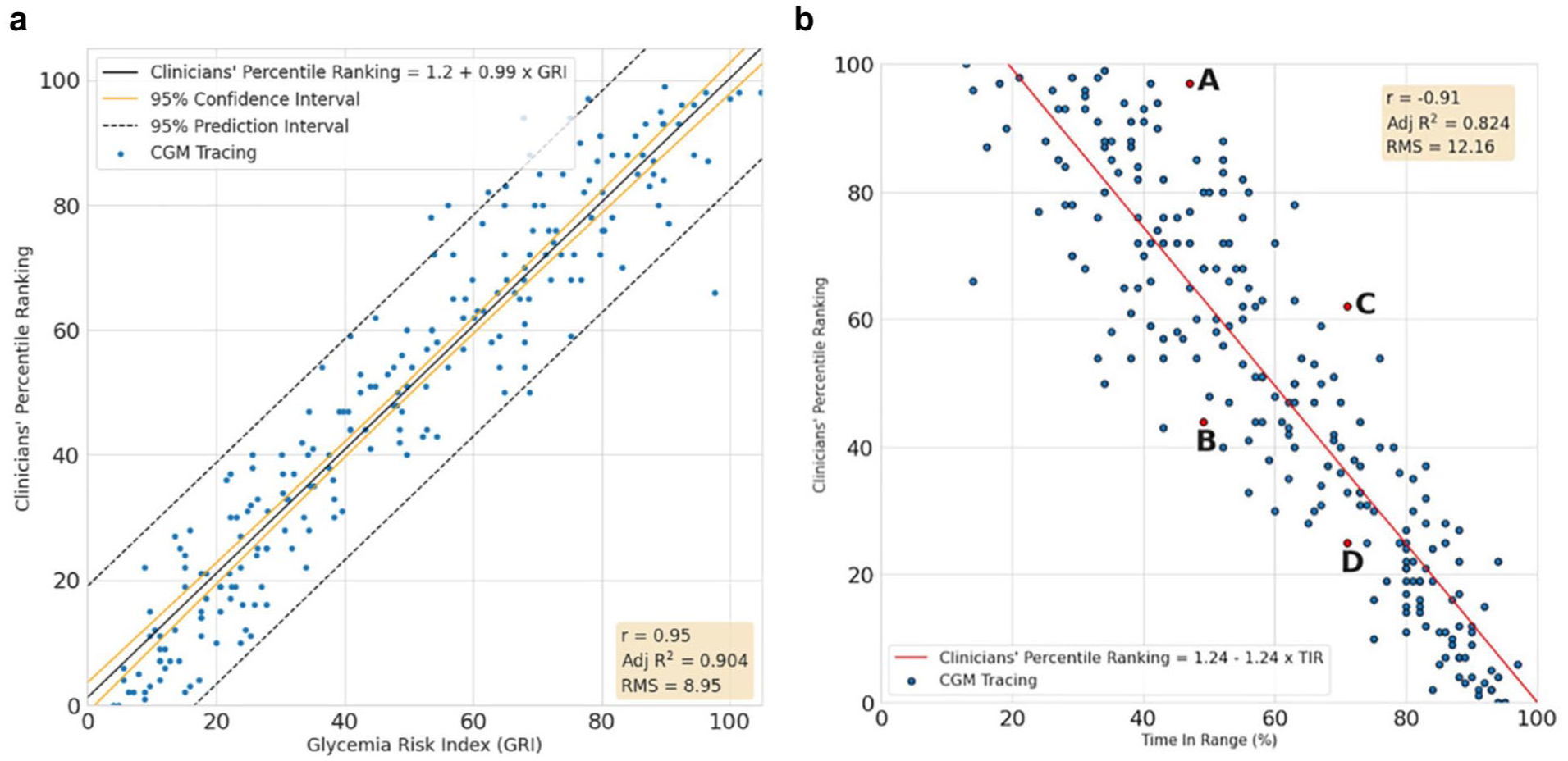
(a) Relationship between clinician ranking and the glycemia risk index (GRI). Linear regression curve demonstrating a strong relationship between GRI and clinician-ranked continuous glucose monitoring (CGM) profiles (R2 = .904). Higher GRI values correspond to the clinician’s opinion of poorer glycemic quality. Reproduced from Klonoff et al. 1 (b) Relationship between clinician ranking and time in range (TIR). CGM tracings with the same TIR percentage can receive markedly different clinician rankings. At around 50% TIR, expert clinician percentile rankings ranged from high risk (97) to substantially better perceived control (44) (points A and B). Similarly, when TIR was approximately 70%, the clinicians’ percentile ranking could be as high as 62 or as low as 25 (points C and D). These examples demonstrate that TIR alone does not fully reflect glycemic quality. Reproduced from Klonoff et al. 1
In the original GRI article, the percentage of time in the target glucose range (TIR, 70-180 mg/dL) achieved an R2 value of .824 and RMSE of 12.16 (compared with the GRI values 0.904 and 8.95, respectively), which indicated that the TIR metric in this study did not fit clinician rankings as closely (Figure 1b). While TIR captures the percentage of readings that fall within the 70 to 180 mg/dL, it fails to quantify the severity of outlier durations of dysglycemia. Furthermore, clinicians were shown to have based their rankings on more than just TIR, because clinician rankings with the same TIR could vary widely in their quality of glycemia. In addition, using the Steiger’s test for dependent correlations, Kohn found that the GRI was more strongly associated with the coefficient of variation (CV) than the TIR (P < .0001). Overall, GRI appears to closely capture expert clinician rankings by using a single metric that can summarize glycemic quality.
The Value of the GRI Metric
Guillermo Umpierrez, MD (Emory University, Atlanta, GA) discussed the clinical value of the GRI, focusing on how it complements traditional CGM metrics such as TIR, time above range (TAR, >180 mg/dL), and time below range (TBR, <70 mg/dL). While the TIR, TAR, and TBR measure the percentage of glucose readings within or outside target thresholds, they do not express the clinical severity of those excursions, especially when very low or very high readings are worse than just low or high readings, respectively. The GRI bridges this gap by combining both magnitude and duration of hypoglycemia and hyperglycemia into a single, weighted value.
Umpierrez highlighted that the GRI is a complementary metric that can quantify risk intensity where TIR cannot. Scores below 20 reflect optimal glycemic stability, values between 20 and 40 represent mild to moderate variability, and scores above 40 indicate marked glycemic instability and higher clinical risk.
In a 2023 longitudinal study of adults with T2D without diabetes-related retinopathy (DR) at baseline, Wang et al 2 found a positive association between GRI and risk of DR in people with T2D. For every 1 standard deviation increase in GRI, the risk of DR was increased by 20% after adjustment for confounding variables. A 2025 retrospective analysis in France 3 evaluated 136 individuals with T1D before and after initiation of hybrid closed loop (HCL). Receiver operating characteristic (ROC) analysis identified that a GRI of less than 26 was the optimal threshold for identifying participants who achieved efficacy and safety targets (specificity 92%, negative predictive value 93%). 3 Based on these findings, if the GRI were to be automatically calculated by CGM platforms for HCL users, then individuals with T1D who require further support could be automatically identified. However, the group did not discuss in detail the target GRI according to which treatment a patient is receiving. Unlike fasting glucose or hemoglobin A1c (HbA1c), the GRI is not intended to diagnose prediabetes or diabetes. Since GRI is correlated with all seven of the metrics presented in an AGP report, including time in target range, and HbA1c correlates only with one of these metrics (time in target range), it would not be expected that a GRI level would be closely correlated with a HbA1c concentration. 1 A 2023 retrospective study also found that GRI correlated strongly with TIR across 202 pediatric and adult individuals using intermittent scanned CGM (isCGM) 4 (Figure 2). In conclusion, these findings suggest that beyond mirroring clinician assessment, the GRI might have value in predicting real-world outcomes and response to therapeutic interventions.
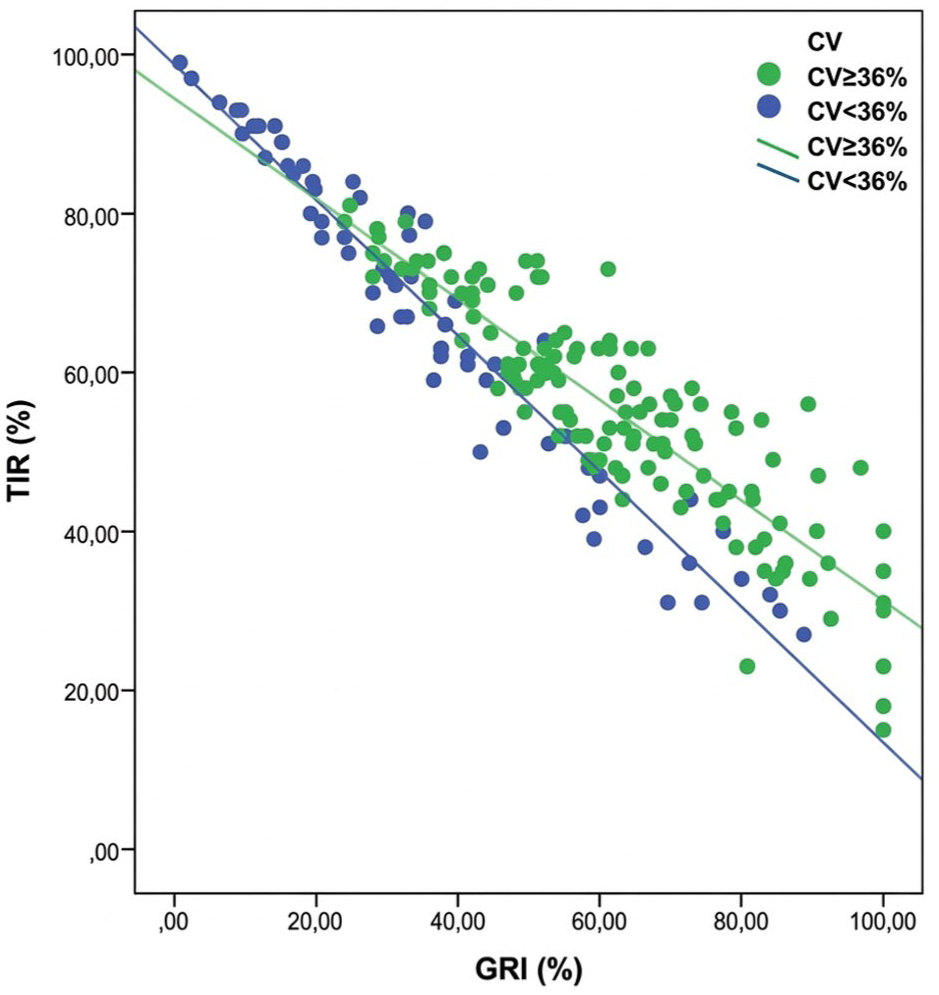
Scatter plot showing the correlation between glycemia risk index (GRI) and time in range (TIR).
GRI for Clinical Care From the Endocrinologist Perspective
Ralph Oiknine, MD (Diabetes and Endocrinology Specialists, Inc, Chesterfield, MO), discussed the practical applications of the GRI in his routine endocrinology practice. He emphasized that while more advanced CGM analytics continue to be developed, clinicians value a simple, intuitive metric that can summarize glycemic outcomes without requiring extensive data interpretation. The GRI fits that description.
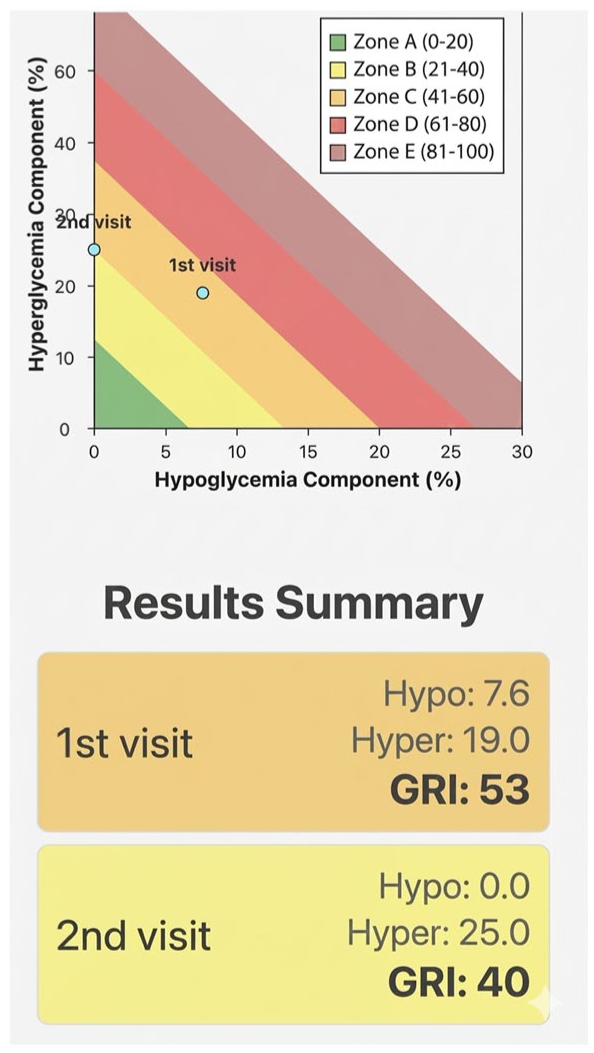
Oiknine described the integration of the GRI into SweetSpot, a diabetes management platform that aggregates data from CGM and insulin delivery devices across various platforms. This centralized digital system displays the GRI alongside the AGP, which allows clinicians to monitor the progression of glycemic risk from one visit to the next. Using the pictorial GRI grid, clinicians can assess the quality of glycemia at-a-glance as it evolves over time (Figure 3), which is something they cannot do by only looking at AGP reports. He concluded that overall, the GRI enhances the value of AGP metrics, allowing busy clinicians to conduct a more thorough assessment of glycemic quality.
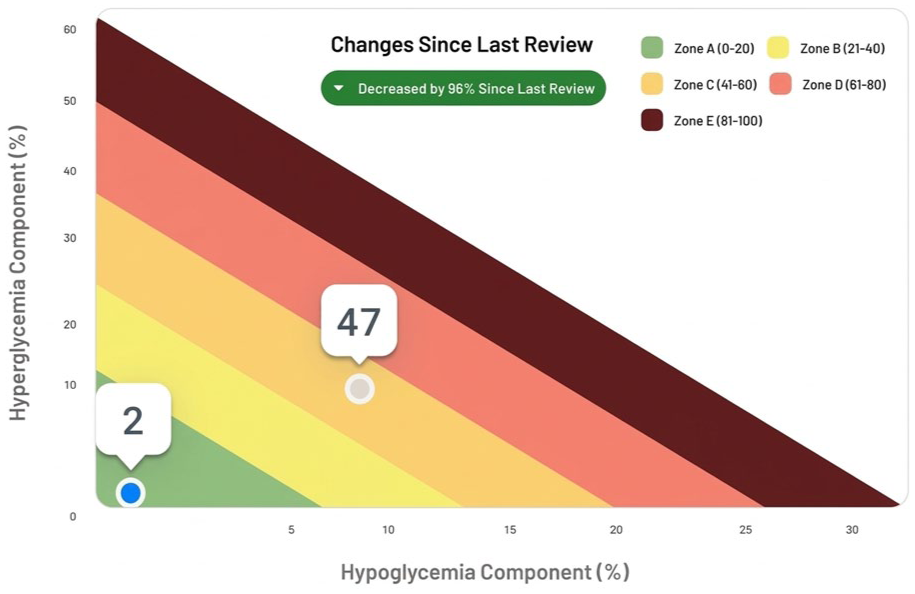
Example of the GRI displayed within the SweetSpot platform.
GRI for Clinical Care of Patients With Cancer
Steven Weitzman, MD, FACE (MD Anderson Cancer Center, Houston, TX), illustrated the potential role of the GRI in enhancing assessment and guiding therapy for individuals with cancer and diabetes. People with cancer have about a 40% higher risk of new‑onset diabetes compared with cancer‑free controls. 5 Chemotherapy can lead to diabetes through drug-induced insulin resistance or beta-cell injury. 6 Weitzman presented a case of a man in his 50s with T2D, who developed immune checkpoint inhibitor–related thyroiditis, and then developed immune checkpoint inhibitor–associated autoimmune diabetes mellitus. This required a change from oral agents to MDIs of insulin. Three sequential CGM tracings from this patient demonstrated progressive improvement of glycemic management (Table 2). It could be possible to eventually develop a specific GRI for diabetes patients with various accompanying diseases, or with either prediabetes or pregnancy. Patients using insulin who need additional support can currently be identified by TBR and TIR, by collapsing these metrics into a single number, and the GRI serves a similar purpose. However, in the original GRI article, a model based on an optimized combination of TBR and TIR was built, and this composite metric did not fit the clinicians’ rankings of the quality of glycemia in the 225 study tracings as well as the GRI. Although it would be possible to calculate a ratio of total TBR divided by total time in the target range as a measure of glycemia, this type of ratio is not well standardized or comparable to a GRI level. The use of a ratio of TBR divided by TIR has been proposed as a metric of overall glycemia; 7 however, this metric has not been validated, and it does not account for changes in glycemia from interventions that either increase or decrease both the TBR and TIR.
Three Sequential CGM Tracings From an Individual With Diabetes, Who Is Being Treated for Cancer.
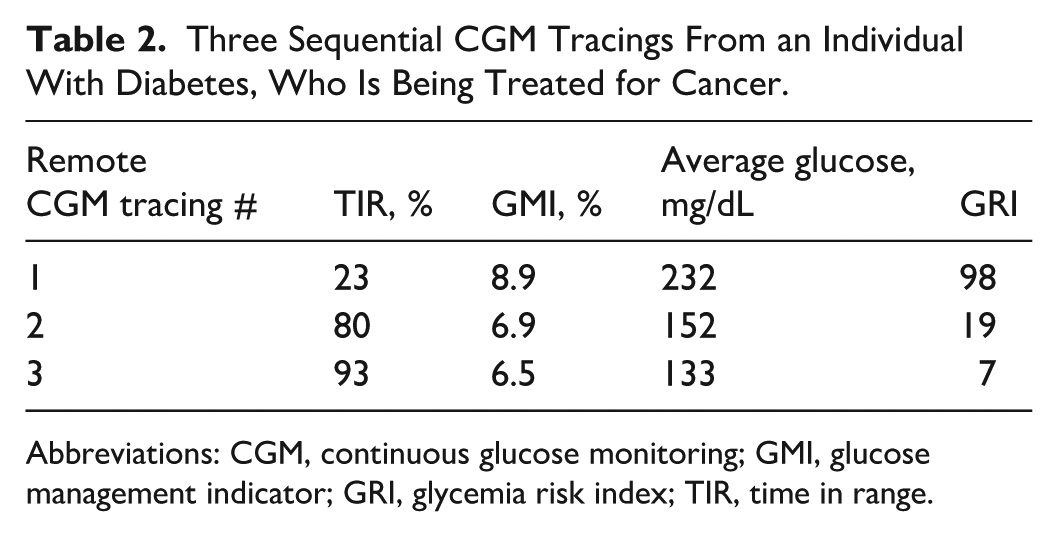
Abbreviations: CGM, continuous glucose monitoring; GMI, glucose management indicator; GRI, glycemia risk index; TIR, time in range.
Although TIR improved substantially over time, the GRI’s large numeric decline from 98 to 7 captured the magnitude of risk reduction more dynamically than either the TIR or glucose management indicator (GMI). The GRI-derived weighting of both hypoglycemia and hyperglycemia by the GRI makes it valuable for both remote and inpatient oncology settings. Weitzman concluded that the GRI provides a quantitative measure of glycemic management in complex cancer care, facilitating earlier and safer diabetes interventions.
GRI for Clinical Care of Diabetes
Veronica Brady, PhD, FNP-BC, BC-ADM, CDCES, FADCES (University of Texas Health Science Center, Houston, TX, and President of the Association of Diabetes Care & Education Specialists), presented the perspective of Certified Diabetes Care and Education Specialists (CDCES), by focusing on the role of the GRI in diabetes education. Brady emphasized that diabetes care and education specialists are often frontline users of digital diabetes tools, and their role is to help individuals and healthcare professionals interpret CGM data and integrate technology into daily management.
Using an example of her reports from current practice (Figure 4), Brady described how the GRI can simplify CGM interpretation for the CDCES by providing an at-a-glance summary of glycemic stability over time. She suggested that the GRI Calculator app can help the CDCES to communicate complex data in a format that is intuitive for individuals and easy to share with healthcare professionals.

Example of a report currently used by a Certified Diabetes Care and Education Specialists (CDCES) (left) shown alongside the GRI calculator (right).
At the same time, she cautioned that the GRI must be used with a contextual understanding of the times of high and low glucose levels. While the GRI provides a single number summary of glycemic risk, it may oversimplify glycemic patterns. Miscommunication may occur if scores are presented without explanation, and workflow burden may increase as new digital metrics are introduced. She also noted that the GRI has not yet been validated for certain populations, including pregnant women and non-insulin users. Brady concluded that, with adequate training, the GRI could enhance individual engagement and strengthen coordination between the CDCES and other members of the healthcare team.
The GRI Calculator App
Jane Seley, DNP MPH MSN GNP BC-ADM CDCES CDTC FADCES (Weill Cornell Medicine, New York, NY), presented the GRI Calculator App, a novel mobile application designed to allow healthcare professionals to rapidly compute and visualize GRI scores from CGM data. The app transforms the GRI formula into a tool for both clinical and educational use. It is available for iOS at the Apple App Store and for Android at the Google Play Store.
The GRI Calculator app calculates a composite risk score from four inputs: percentages of time Very Low (<54 mg/dL), Low (54-69 mg/dL), High (181-250 mg/dL), and Very High (>250 mg/dL). These data can be entered manually or uploaded as a comma-separated values (CSV) file. Each overall GRI result is assigned a corresponding risk zone (A through E), ranging from little to no risk to severe risk, in five equal quintiles of glycemia. The app also allows entry of multiple tracings across multiple visits, and automatically generates a trend plot that compares GRI values over time (Figure 5). Integrating the GRI Calculator into diabetes care can facilitate a quick review of CGM uploads and visualizations of overall glycemic risk trajectories without requiring advanced data analysis.
Example screenshot from the GRI Calculator App visualizing multiple visits.
GRI for Population Health
Viral Shah, MD (Indiana University, Indianapolis, IN), discussed the potential of the GRI as a tool for population health and clinical research. He emphasized that while no glycemic metric is perfect, the GRI has an advantage when evaluating outcomes across diverse populations and interventions, compared with more traditional metrics such as TIR and HbA1c.
In a pooled analysis of 33 intervention studies published between 2022 and September 2025, Shah and colleagues compared the delta-GRI and delta-TIR values across therapeutic interventions. In more than 80% of the articles included in the analysis, the delta-GRI exceeded the delta-TIR. 8 The study demonstrated that although both GRI and TIR are reported on a 0 to 100 scale, these two metrics use different units, which complicates the comparison of the two metrics. The study indicated that the delta-GRI is typically numerically larger than the delta-TIR and that the GRI typically improves following beneficial interventions.
Shah noted that the agreement between a 90-day GRI measurement and a shorter measurement progressively increases over the 90 days (Figure 6). He also cited ongoing work that suggests the GRI may predict long-term diabetes complications since glycemic variability is linked to microvascular damage.9,10
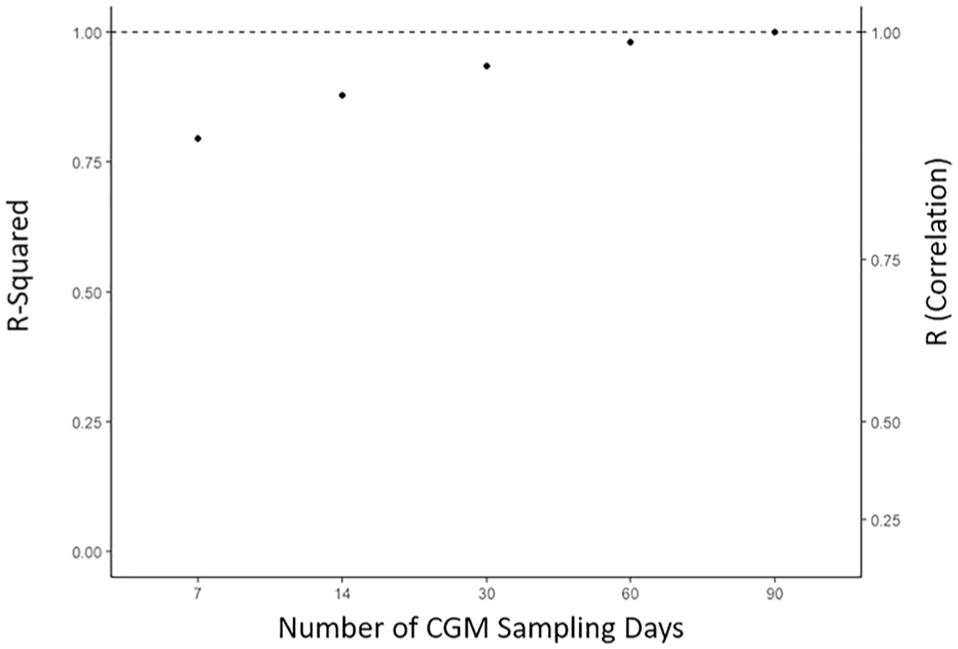
Relationship between continuous glucose monitoring (CGM) sampling duration and agreement with 90-day glycemia risk index (GRI).
A survey of 28 CDCESs and other healthcare professionals found that 75% were interested in integrating GRI into their clinical workflows, highlighting enthusiasm among clinicians to adopt the new metric. 12 Shah concluded that while the GRI remains relatively new, it could become a valuable tool in population health.
Use of AI to Interpret the GRI
Elizabeth Healey, PhD (Harvard University, Boston, MA), discussed how the GRI is beginning to be used in artificial intelligence (AI) for research and diabetes management. As there is increasing interest in using AI systems to interpret medical data, it is necessary to consider how to make AI decision-making more transparent and aligned with clinician priorities. Because the GRI quantifies clinician trade-offs between hypoglycemia and hyperglycemia, it provides a lens for evaluating how AI models could assess glycemic risk.
To evaluate how closely AI reasoning aligns with clinician judgment, Healey’s team used 100 simulated adult T1D patient datasets to create CGM profiles for three treatments: closed-loop control therapy, open-loop control with continuous subcutaneous insulin infusion, and MDI. Healey and colleagues then conducted pairwise comparisons of these 100 profiles using five large language models (LLMs) developed by OpenAI and Anthropic. The models tested included GPT-3.5, GPT-4.1, GPT-o4-mini, Claude Haiku, and Claude Sonnet. This study framework is summarized in Figure 7.
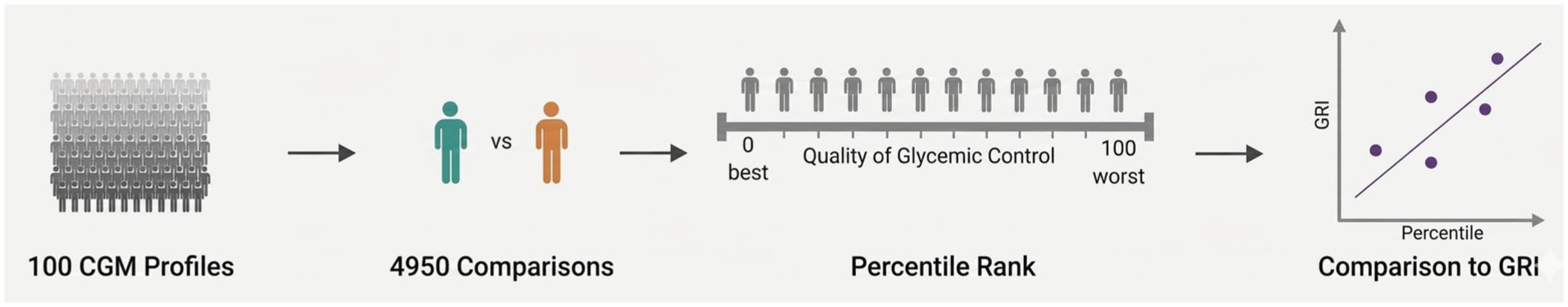
Alignment study schematic illustrating the study by Chandak et al. 13
Each model ranked the CGM tracings by glycemic quality, and those rankings were compared with GRI-based and TIR-derived rankings. Following the calculation of Spearman correlation coefficients between each LLM’s rankings and the two reference rankings, Healey found that an older LLM (GPT-3.5) demonstrated greater correlation with the TIR (r = .84 for MDI treatment) than GRI (r = .79 for MDI treatment). A newer model (GPT-o4-mini) demonstrated near-perfect alignment with the GRI (r = .99 for MDI treatment), surpassing the correlation with TIR (r = .97 for MDI treatment). This result was similar across data simulated with all three treatment modalities, suggesting that newer AI models synthesize the tradeoffs between hypoglycemia and hyperglycemia in a way that is similar to the GRI.
To understand the source of the decisions in these models, investigators performed a perturbation analysis, where they varied one AGP value while holding others constant to observe how each model weighted different values. The findings revealed that the AI models underweight mild hypoglycemia compared with the GRI. In contrast, the GRI slightly overweights mild hypoglycemia relative to mild hyperglycemia to reflect clinician caution regarding low glucose events.
Healey concluded that the GRI can function as a “gold-standard” reference for evaluating AI decision-support systems because it quantifies expert clinician decision-making. As medical AI becomes more pervasive, benchmarking new models against the GRI will allow for a transparent, quantifiable way to ensure that LLM judgment reflects clinician priorities.
The iCoDE-2 Dashboard
Juan Espinoza, MD (Ann & Robert H Lurie Children’s Hospital of Chicago, Chicago, IL), discussed the Integration of Connected Diabetes Device Data into the Electronic Health Record (iCoDE-2), a two-year project involving over 120 participants from industry, academia, United States federal agencies, and patient advocacy groups. 14 The goal of iCoDE-2 is to develop technical specifications for integrating insulin delivery data into electronic health records (EHRs) and establish standardized guidelines for consistent presentation of data.
The Insulin Dosing Profile (IDP) is a one-page EHR-friendly summary that presents insulin-delivery and CGM data side-by-side over a 14-day window with the goal of supporting clinical decision-making (Figure 8). It hosts metrics of CGM patterns, including the GRI, thereby allowing clinicians to interpret glycemic quality and insulin dosing data on a single page. 14
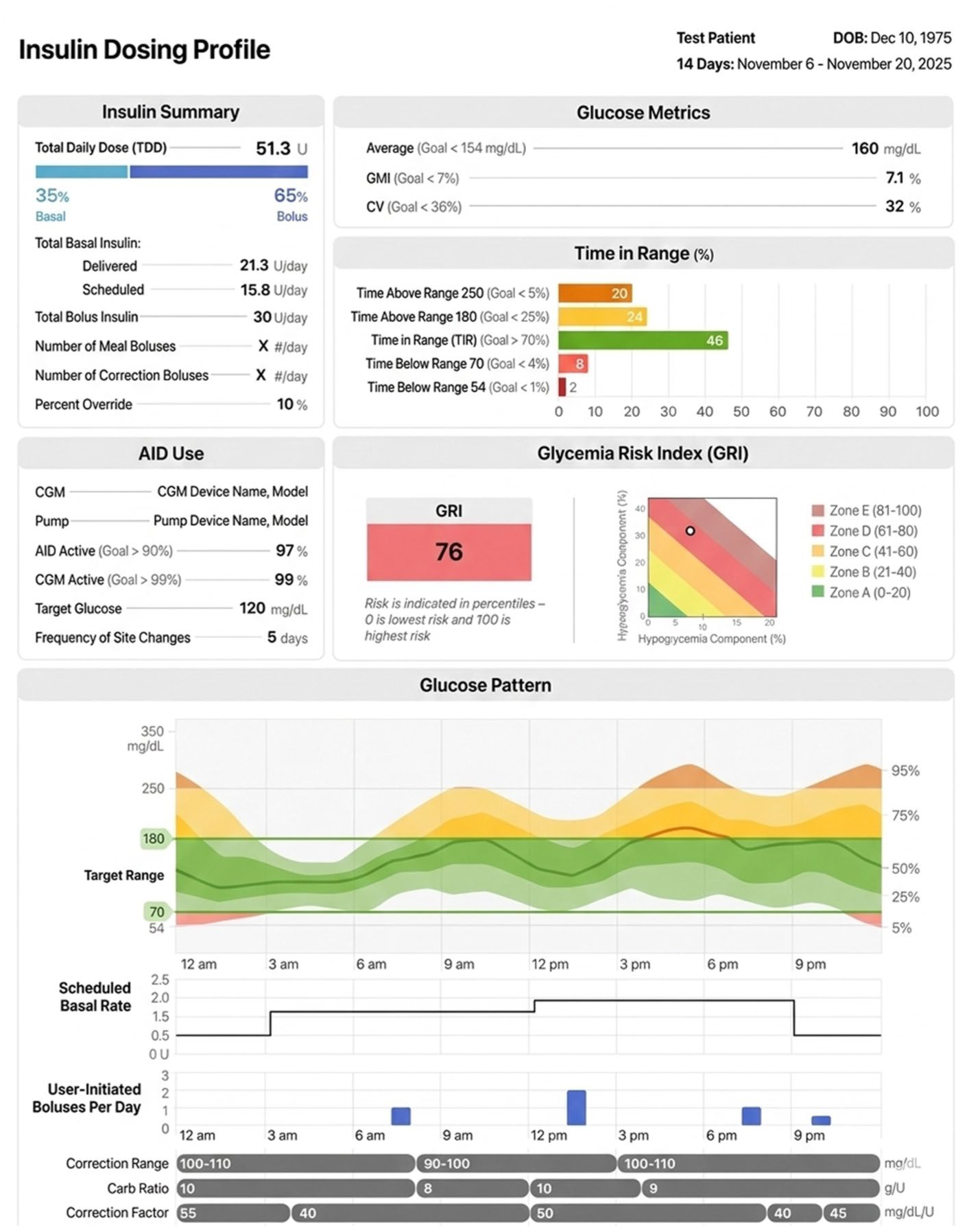
The Insulin Dosing Profile (IDP) is one-page insulin and CGM report developed through the iCoDE-2 project.
The iCoDE-2 working group is now focused on disseminating, validating, and adopting the technology across the industry. Espinoza concluded that embedding the GRI within standardized insulin-data visualization is a step toward better data-driven diabetes care.
GRI and Pediatrics
Mark Clements, MD, PhD (Glooko, Palo Alto, CA) and David Maahs, MD, PhD (Stanford University, Stanford, CA), reviewed the clinical use of the GRI in pediatric populations. They focused on its correlation with HbA1c, youth self-management habits, and potential integration into digital pediatric care models.
In a study conducted by Panfil et al, 15 GRI strongly correlated with both current and future HbA1c measurements. In another study, GRI was found to correlate with the seven habits of pediatric diabetes self-management: (1) monitoring glucose frequently through CGM use or checking blood glucose at least four times per day, (2) administering three or more insulin boluses daily, (3) utilizing insulin pump therapy, (4) delivering bolus doses before meals, (5) reviewing glucose trends between clinic visits, (6) adjusting insulin doses between appointments, and (7) consumption of fruits and vegetables. 16 Lower GRI scores were associated with better diabetes self-care, suggesting that the GRI can reflect the quality of day-to-day glycemic management (Figure 9).
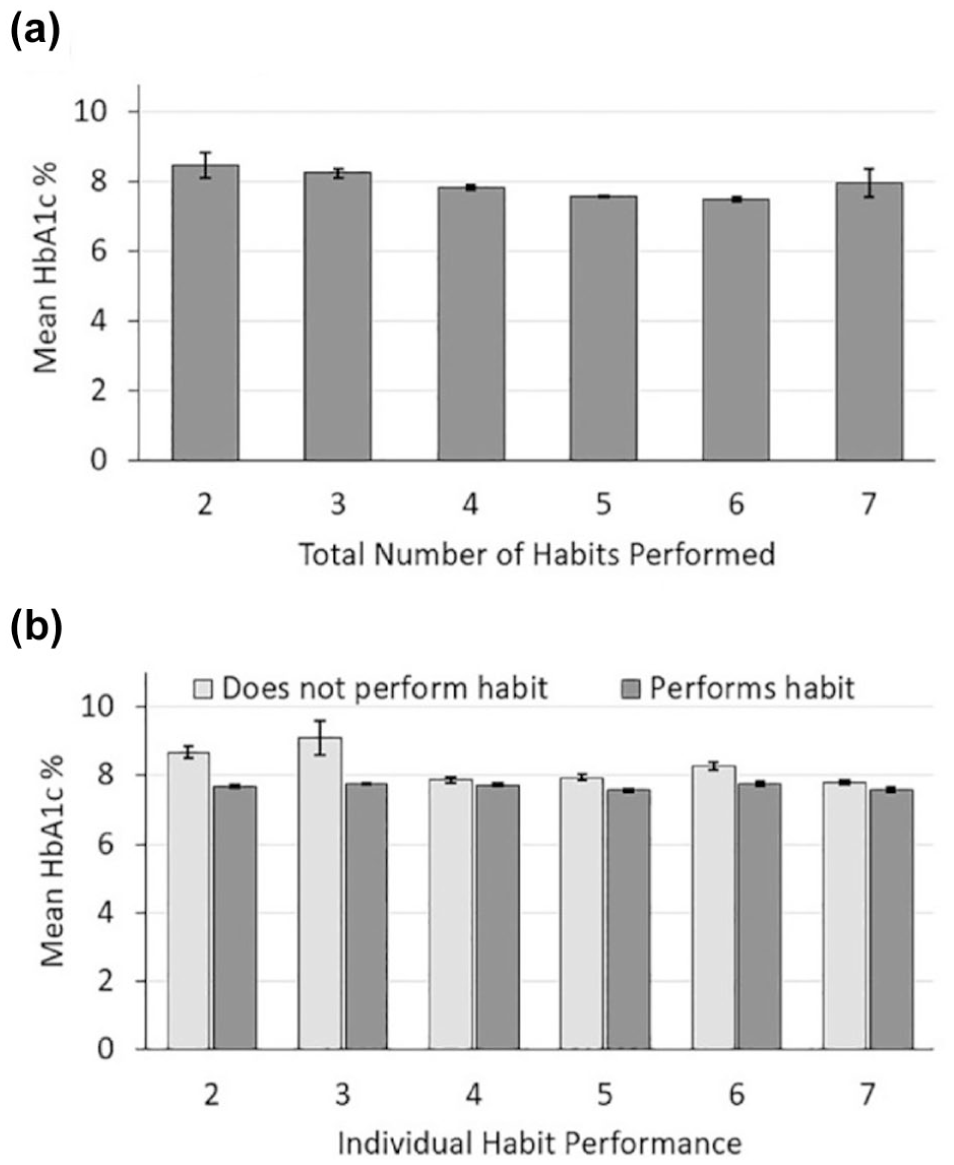
Mean glycemia risk index (GRI) by number of self-management habits performed in youth with type 1 diabetes (T1D).
Both of these presenters discussed the possibility of incorporating the GRI into existing data platforms, including the Timely Intervention for Diabetes Excellence (TIDE) 17 platform, which uses CGM data to automatically identify individuals who need extra attention at scheduled clinician visits or special interventions between clinician visits. They also referred to the Teamwork, Targets, Technology and Tight Control (4T) 18 intervention program as a platform to test the GRI’s ability to monitor treatment progress in youth with T1D. Clements and Maahs concluded that embedding the GRI into pediatric diabetes care frameworks could allow for quantification of glycemic risk.
Discussion
The panel agreed on four points during the discussion (see Table 3). Specifically, the expert panel members agreed that the new generation of diabetes metrics, including GRI, should extend beyond time-based CGM measures to capture the magnitude and frequency of glycemic excursions. Across the presentations, participants viewed the GRI as a bridge between data analytics and clinician judgment, which offers a single value summary of glycemic quality that aligns with expert clinician opinion.
Key Discussion Points and Consensus.
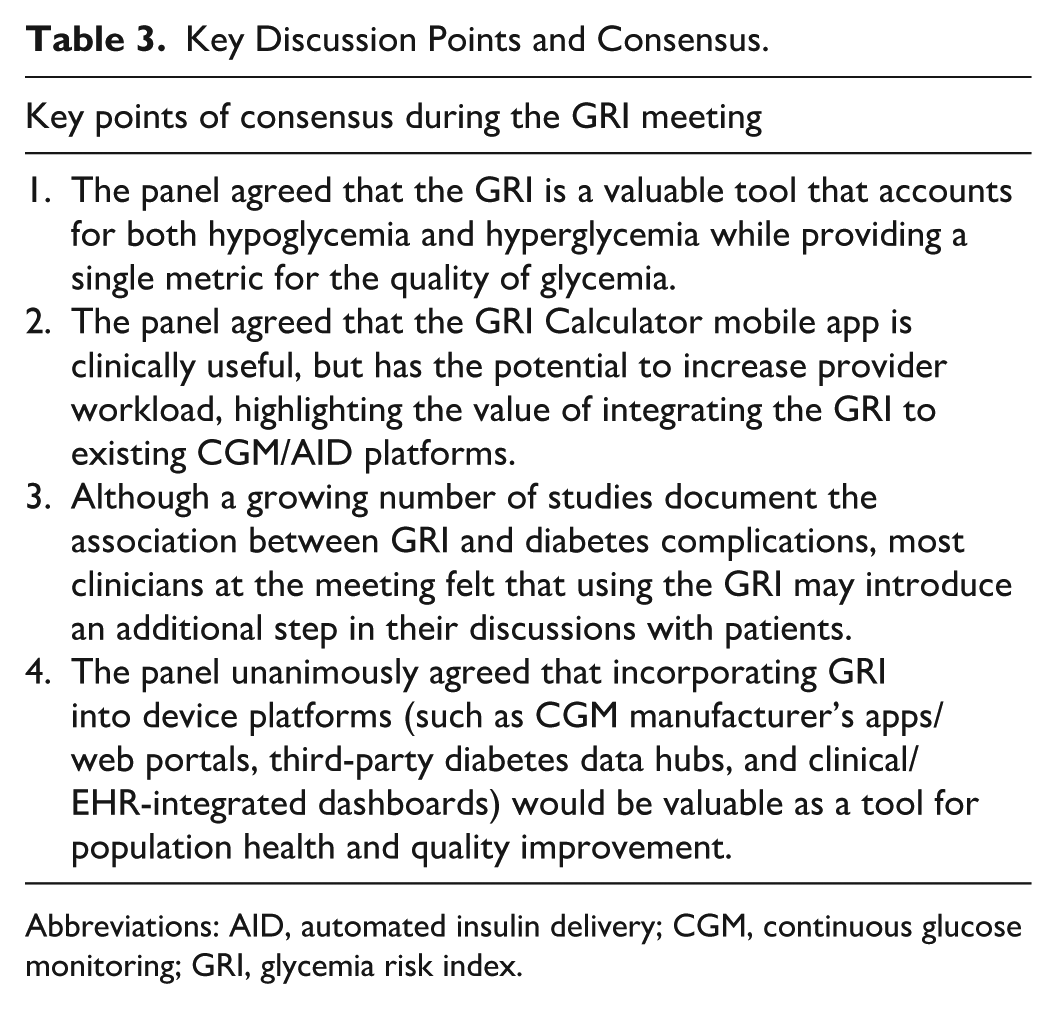
Abbreviations: AID, automated insulin delivery; CGM, continuous glucose monitoring; GRI, glycemia risk index.
The group identified no particular GRI value consistent with a poor glycemic control that indicated a need to refer an individual to a diabetes specialist, and that each clinician should decide for themselves which GRI level warrants referral to specialist services. One participant suggested that a GRI of more than 40 should warrant such referral, but no other participant either agreed or disagreed with this recommendation.
Participants at the meeting also affirmed that the GRI complements existing metrics such as TIR, TBR, and TAR. Clinical studies have also demonstrated that the GRI accurately mirrors clinician rankings of CGM profiles and correlates with outcomes, including predicting the risk of DR and responses to therapy.
Examples across endocrinology, oncology, and primary care practices illustrate how the GRI can be applied to support clinicians involved in diabetes care. The GRI can also be used as a tool for the education of clinicians and individuals choosing to wear a CGM. In addition, pediatric studies have shown that the GRI may have relevance in younger populations, suggesting that it could become a standard for quantifying glycemic risk across all ages, especially if a specific GRI based on pediatric data can be developed. With data visualization projects like iCoDE-2, there are pathways in place for embedding the GRI within the EHR. 19 The GRI can also serve as a benchmark for evaluating AI models, quantifying whether the decisions LLMs make align with clinician opinion. 13
Participants acknowledged several current limitations to the GRI’s adoption. As the metric is relatively new, it is not yet widely adopted in routine diabetes care. Because the GRI was developed using data primarily from adults using insulin, multiple speakers questioned whether the GRI could be generalized to adults not using insulin. Others noted that there are no specified cutoff values that can guide clinical decisions and more data are needed to define these thresholds. Broader clinical adoption would require more validation across diverse groups, including pediatric populations, pregnant women with gestational diabetes or pregestational diabetes, and people with prediabetes or normoglycemia, all of whom have different targets than adult diabetes patients using insulin, and might need their own GRIs based on expectations for those groups. Some participants expressed concern that introducing another metric could increase the workload of clinicians and diabetes care and education specialists to learn about the GRI if they did not understand how to use the metric. An additional concern was raised that some patients may not know how to interpret the GRI without proper education, and the metric must be explained carefully to avoid adding to diabetes distress. Some participants questioned whether a single composite value risks oversimplifying the glycemic patterns. They determined that while the GRI may be useful for population health, for patients not doing well, clinicians should still review the full AGP and device settings to make clinical decisions.
Finally, the GRI meeting participants agreed that the GRI is valuable for clinical, educational, and AI validation purposes. The group unanimously supported incorporating the GRI into software dashboards, reports, and platforms offered by CGM manufacturers, enabling clinicians and researchers to track improvements in diabetes care and population health.
Footnotes
Acknowledgements
The authors thank Annamarie Sucher-Jones for her expert editorial assistance.
Abbreviations
4T, Teamwork, Targets, Technology and Tight Control; AGP, ambulatory glucose profile; AI, artificial intelligence; AID, automated insulin delivery; CDCES, certified diabetes care and education specialist; CGM, continuous glucose monitoring; CLC, closed-loop control therapy; CSII, continuous subcutaneous insulin infusion; CSV, comma-separated values; CV, coefficient of variation; DR, diabetes-related retinopathy; DTM, Diabetes Technology Meeting; DTS, Diabetes Technology Society; EHR, electronic health record; GMI, glucose management indicator; GRI, glycemia risk index; HbA1c, hemoglobin A1c; HCL, hybrid closed loop; iCoDE-2, integration of connected diabetes device data into the electronic health record; IDP, Insulin Dosing Profile; iSCGM, intermittent scanned CGM; LLM, large language model; MDI, multiple daily injection; PCA, principal component analysis; RMSE, root mean square error; ROC, receiver operating characteristic; T1D, type 1 diabetes; T2D, type 2 diabetes; TAR, time above range; TBR, time below range; TDD, total daily dose; TIDE, Timely Intervention for Diabetes Excellence; TIR, time in range; TITR, time in tight range; US, United States; VHigh, very high; VLow, very low.
ORCID iDs
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GEU has received research support for Emory University from Bayer, Corcept, Abbott, Glucotrack, and Dexcom, and has participated in advisory boards for Dexcom, Corcept, Glucotrack, and Glycare. VNS’ institute has received research funding from Lilly, Enable Bioscience, Zucara Therapeutics, Cystic Fibrosis foundation, and Breakthrough T1D. VNS has received honoraria from Sanofi, Novo Nordisk, Lilly, Dexcom, Insulet, Tandem Diabetes Care, Medtronic, Sequel Med Tech, Abbott Diabetes, Roche, Biomea Fusion, and T1D Scout for advising, consulting, or speaking outside of this submitted work. VB has nothing to disclose. MAC is an employee of Glooko. JE receives funding from the Helmsley Charitable Trust, and is a consultant for Sanofi, Glooko, and Dexcom. EH is supported by T32HD040128 from the NICHD/NIH. MAK has nothing to disclose. DMM has had research support from the NIH, JDRF, NSF, and the Helmsley Charitable Trust and his institution has had research support from Medtronic, Dexcom, Insulet, Bigfoot Biomedical, Tandem, and Roche. DMM has consulted for Abbott, Aditxt, the Helmsley Charitable Trust, LifeScan, MannKind, Sanofi, Novo Nordisk, Eli Lilly, Medtronic, Insulet, Dompe, Biospex, Provention Bio, Kriya, Enable Biosciences, and Bayer. RO is the founder and chief medical officer of SweetSpot. JJS has nothing to disclose. SPW has nothing to disclose. HKA reports research support and honorarium through the University of Colorado from Medtronic, Dexcom, Roche, and Tandem Diabetes Care. GA reported personal fees from Eli Lilly, Dexcom, Insulet, Medscape, and Tandem Diabetes Care, and grants from AbbVie, Bayer, Fractyl Health, Insulet, MannKind, and Tandem Diabetes Care, outside the submitted work. MEA has served on an advisory panel for Abbott, Medtronic, Insulet, VitalAire, and Sanofi; has received honoraria for speaking and consultancy from Abbott, Eli Lilly, Medtronic, Novo Nordisk, Sanofi, and VitalAire; and has received research fund from Medtronic and Sanofi. EC has served on the scientific advisory board of Novo Nordisk, Eli Lilly, MannKind, Arecor, Portal Insulin, Provention Bio, Tandem, Sanofi, and Ypsomed. FJD has received patent royalties at UCSB and Harvard from licenses granted to Dexcom, Roche and Insulet. OE have received research grants through his organization T1D Exchange from Medtronic, Lexicon, Vertex, Lilly, and Sanofi. SVE has nothing to disclose. L.E. receives salary support from NIDDK. LE’s institution has received research support from Breakthrough T1D, The Leona M. and Harry B. Helmsley Charitable Trust, Sanofi, Medtronic, MannKind, Tandem Diabetes Care, and Abbott. She has served on the advisory board of Sanofi, Abbott, MannKind, Diabetes Center Berne, Sequel, and Medtronic. She has received consulting fees from Jaeb, Ypsomed, and Tandem Diabetes Care, and has received honorarium fees from ADA, Endocrine Society, Children with Diabetes (MannKind-sponsored event), Med Learning Group (Sanofi-sponsored grant), Tandem Diabetes Care, Medtronic, and Insulet. GF is general manager and medical director of the IfDT (Institut fürDiabetes-Technologie, Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany), which carries out clinical studies, eg, with medical devices for diabetes therapy on its own initiative and on behalf of various companies. GF/IfDT have received research support, speakers’ honoraria or consulting fees in the last three years from Abbott, Ascensia, Bionime, Boydsense, Dexcom, Glucoset, Insulet, i-SENS, Lilly, Menarini, Novo Nordisk, Perfood, Pharmasens, Roche, Sinocare, Terumo, Vertex, and Ypsomed. LH has received honoraria from Roche Diagnostics, Lifecare, Medtronic, Liom, Dexcom, OneTwenty, Perfood, Boydsense, PharmaSens, Unomedical, and Sinocare for lectures and participation in advisory boards. LH is a shareholder in Profil Institut für Stoffwechselforschung GmbH, diateam GmbH, and Science Consulting in Diabetes GmbH. IBH has received research support from MannKind, Dexcom, and Tandem. IBH is a consultant for Abbott, Roche, and Hagar. VCH has nothing to disclose. DK has served as a consultant for Sanofi, Evidation Health, and Better Therapeutics and has stock options for Glooko. SHK is a consultant for TeCure and received an education grant from Novo Nordisk. BK reports receiving research support from Dexcom, Inc and Tandem Diabetes Care handled by the University of Virginia and patent royalties from Dexcom, Inc and Tandem Diabetes Care handled by the University of Virginia’s Licensing and Ventures Group. RAL has received consulting fees from Abbott Diabetes Care, Adaptyx Biosciences, Biolinq, Capillary Biomedical, Deep Valley Labs, Gluroo, Portal Diabetes, Sanofi, and Tidepool. He has served on advisory boards for Provention Bio, Lilly, and Rezolute. He receives research support from his institutions from Insulet, Medtronic, Sinocare, and Tandem. ML has received honoraria or been consultant for Boehringer Ingelheim, Eli Lilly, Nordic Infucare, Novo Nordisk, and Rubin Medical and received research grants from Eli Lilly and Novo Nordisk. JKM is a member of advisory boards of Abbott Diabetes Care, Becton-Dickinson, Biomea Fusion, Dexcom, Eli Lilly, Embecta, Medtronic, myLife, Novo Nordisk A/S, Pharmasens, Roche Diabetes Care, Sanofi-Aventis, Tandem, and Viatris and received speaker honoraria from A. Menarini Diagnostics, Abbott Diabetes Care, Dexcom, Eli Lilly, Medtrust, MSD, Novo Nordisk A/S, Roche Diabetes Care, Sanofi, Sinocare, Viatris, and Ypsomed. She is a shareholder of decide Clinical Software GmbH and elyte Diagnostics and serves as CMO of elyte Diagnostics. NM receives research support from the NIH, Breakthrough T1D, Helmsley Charitable Trust, and Samsung Research America. TM is a consultant for Dexcom, Amylyx, MARS, January AI, Regeneron, Recordati. SAM has nothing to disclose. DAO has nothing to disclose. MP has been serving as advisory board member of AstraZeneca, Eli Lilly, MannKind, Medtronic Diabetes, Pfizer, Sanofi, DOMPE, LifeScan, Novo Nordisk, Insulet, ProventionBio, Merck, Ascensia, Bayer, Embecta, and Tandem. He has received consulting fee from: Qulab Medical, ProventionBio. The Institute he is heading received research grants from Eli Lilly, Medtronic Diabetes, Novo Nordisk, Pfizer, Sanofi, DreaMed Diabetes, NG Solutions, DOMPE, Lumos, GWAVE, OPKO, ProventionBio, AstraZeneca, Omega Galil. MP is stock owner: DreaMedDiabetes and NG Solutions.
PP has received consulting fees from Sanofi. DS has nothing to disclose. MPS is a co-founder and a member of the scientific advisory board of Personalis, Qbio, January AI, SensOmics, Protos, and Mirvie. He is on the scientific advisory board of Danaher, GenapSys, and Jupiter. JW has nothing to disclose. JCW has nothing to disclose. EEW has received consulting fees from Abbott Diabetes Care, Ascensia, Bayer, Boehringer, Ingelheim, Embecta, GlaxoSmithKline, Lilly, Medtronic, Renalytix, and Sanofi. EEW has received honoraria from Abbott Diabetes Care, Bayer, Boehringer Ingelheim, GlaxoSmithKline, Lilly, Medtronic, Renalytix, and Sanofi. EEW is on the Speakers’ Bureau from Abbott Diabetes Care, Bayer, Boehringer, Ingelheim, GlaxoSmithKline, Lilly, Renalytix, and Sanofi. MMS has nothing to disclose. AYD has nothing to disclose. DCK is a consultant for Afon, Atropos Health, Embecta, Glooko, Glucotrack, Lifecare, Novo Nordisk, SynchNeuro, and Thirdwayv.
