Abstract
Introduction:
Automated insulin delivery (AID) systems for type 1 diabetes (T1D) improve HbA1C, increase time-in-range, and reduce hypoglycemia. However, starting AID systems involves multiple steps, from decision to initiation. This study quantified time to AID initiation (TT-AID) and factors influencing the timeline.
Methods:
This retrospective study included adults with T1D at an academic diabetes center in Baltimore, Maryland who were on multiple daily injections and initiated an AID system for the first time since diagnosis from May 2022 to March 2025. Demographics and dates of AID decision, AID selection visit (optional), prescription, training, and initiation were extracted from electronic medical records. Time to AID initiation was measured, with differences by insurance and AID selection visit assessed using Wilcoxon rank-sum and log-rank tests.
Results:
Participants included 114 adults with T1D [median age 38.9 years, 57% male, 21% Black, 75% commercial insurance, median diabetes duration 10.2 years (IQR = 3.5, 18.1)]. The median TT-AID was 89.5 days (IQR = 49, 132). The longest delay was between decision and training [median: 82.5 days (IQR = 43, 122)]. Patients attending the optional AID selection visit had significantly longer TT-AID compared with those who did not [112 (IQR = 79, 144) vs 55 (IQR = 35, 98) days,
Conclusion:
Adults took roughly 3 months to initiate AID, with longer delays among those with public insurance and those attending AID selection visits. Streamlining AID system initiation may reduce delays and optimize outcomes.
Keywords
Introduction
Over the last decade, there has been an increase in use of diabetes technologies such as continuous glucose monitors (CGMs), insulin pumps, and automated insulin delivery (AID) systems.1,2 These diabetes technologies significantly reduce the burden of glucose monitoring and insulin administration, leading to lower HbA1c levels, increased time-in-range, and reduced risk of acute complications, including hypoglycemia and diabetic ketoacidosis.3,4 Ultimately, integration of CGMs with insulin pumps, leading to AID functionality, makes AID a powerful tool for achieving optimal glucose control and enhancing the quality of life for individuals with diabetes.5-7
The process of initiating an AID system is complex, involving multiple steps including decision to start an AID system, AID selection, prescription, training with either a diabetes educator or with a vendor representative, and finally AID initiation. 8 At our adult diabetes center, the AID initiation process begins with the patient and provider making a shared decision to manage the patient’s diabetes with an AID system. If the patient would like to learn more about each AID system, the provider recommends an optional AID selection visit with a diabetes educator for the patient to learn about the different AID types. Once the patient chooses a specific AID system, the provider then orders the AID. If the patient chooses Omnipod 5, the prescription is sent directly to a pharmacy where the patient can pick up their supplies. For other AID systems, the order goes through a Durable Medical Equipment (DME) supplier, and the patient communicates with the vendor to initiate the process. Automated insulin delivery system trainers are notified once AID ships out for Tandem, Medtronic and BetaBionics AID, or, for Omnipod 5, when the patient scans the QR code or opens the controller. The patient is then trained to use the chosen AID system by either a diabetes educator or a vendor representative, based on AID type and patient preference. 8 Following AID system training, the patient starts the AID. 8
While AID systems offer significant benefits for diabetes management, they are not universally adopted. The American Diabetes Association (ADA) recommends AID for management of all individuals with diabetes. 9 However, a significant portion of individuals with diabetes continue to rely on traditional multiple daily injections (MDIs). 10 As of 2024, only approximately 30% of adults with type 1 diabetes (T1Ds) in the United States utilize AID systems. 11 While limitations to access and patient-related factors delaying AID adoption persist, the complex AID initiation process potentially adds barriers.8,12-15 Clinical observation shows it is not uncommon for patients to wait several months or even longer for AID initiation. However, there is limited data describing the time it takes to start an AID system. Therefore, the primary aim of this study is to identify time to AID system initiation (TT-AID), defined as total duration from the time decision is made to pursue an AID system to actual AID use. We also aim to delineate time intervals between each sequential step within the AID initiation process and to identify factors contributing to variability in this timeline. We hypothesized that sociodemographic factors may act as significant bottlenecks, leading to delayed initiation of this beneficial technology.
Methods
Study Design and Participants
This retrospective study included adult patients with T1D, age 21 years and older, who decided to start an AID system, an insulin pump integrating data from a CGM through a closed-loop algorithm, at the Johns Hopkins Diabetes Center from May 1, 2022, to January 22, 2025. We included patients who were on MDIs and were starting an AID system for the first time since diagnosis. We also only included patients starting AID systems after May 2022, as this was when Omnipod 5 became available for early pharmacy prescription release. Patients previously on any insulin pump before May 2022 or who had other types of diabetes were excluded. This study was approved as a human subjects research study by the Johns Hopkins Institutional Review Board (IRB00181094) with a waiver of consent.
Data Collection
Data were collected from review of electronic medical records (EMRs) from January 2, 2025, to March 22, 2025. Extracted data included age, sex, race, ethnicity, and insurance type (commercial or public). The area deprivation index was obtained by using University of Wisconsin’s Area Deprivation Index tool, which calculates percentile ranking based on census-derived socioeconomic indicators.
16
Steps of AID initiation process were identified from the process map published by our group.
8
Dates of AID initiation process collected include
For patients who initially chose to start an AID system but later declined, reasons for discontinuation were recorded. Data were entered into REDCap and reviewed by two independent study team members.17,18
Outcome Measures
The primary outcome was time to automated insulin delivery system initiation (TT-AID) defined as the time interval, in days, from documented shared decision to initiate AID to date of actual AID initiation.
Secondary outcomes included differences in TT-AID based on insurance type (commercial vs public), prescription pathway (pharmacy vs DME suppliers), and AID selection visit completion. Time to AID initiation within 6 months of AID decision was also included. This 6-month timeframe reflects typical follow-up interval in diabetes care and represents a clinically meaningful window when AID initiation is expected to occur.
Statistical Analysis
Summary data are presented as medians with interquartile ranges (IQRs) for continuous variables, while categorical variables are expressed as counts and percentages. Differences in patient characteristics between groups were tested using chi-square test or Fisher exact test for categorical variables and two-sample
To calculate TT-AID, we determined interval between date of decision to initiate an AID system and AID initiation date as shown in the device portal. For patients (n = 31) who received training from a vendor representative, portal connection date was imputed for training date since these are typically the same. For patients (n = 3) lacking portal connection, EMR-documented AID initiation date was utilized. Median and IQR were calculated for each process. Wilcoxon rank-sum tests compared median time between those with an AID selection visit and those without.
For time to event analysis, participants who had not yet initiated an AID system were considered right-censored. Time-to-event analysis was also conducted with arbitrary right-censoring at 6 and 9 months after decision date to better illustrate early effects. Kaplan-Meier method was used to estimate survival function and visualize time-to-event distributions between groups. Median TT-AID and corresponding 95% log-log confidence intervals are presented. Group differences in survival distributions were assessed using log-rank test.
Multivariable Cox proportional hazard model was used to test association between age, sex, race, insurance type, AID prescription/order pathway, and duration of diabetes with TT-AID. Proportional hazards assumptions for each variable were assessed using both standard residual-based diagnostics and resampling-based approach and were satisfied. The significance level of
Results
Patient Characteristics
Initial review of EMR identified 1686 adults with International Classification of Diseases, 10th Revision (ICD-10) diagnosis codes for T1D followed at our adult diabetes center. Of these, 965 patients were excluded due to insulin pump use prior to May 2022, 142 for other types of diabetes, and 464 who did not decide to start using an AID system between May 2022 and January 2025. One additional patient was excluded who did not have an AID decision date recorded in the EMR (Figure 1). A total of 114 patients were included in the study.
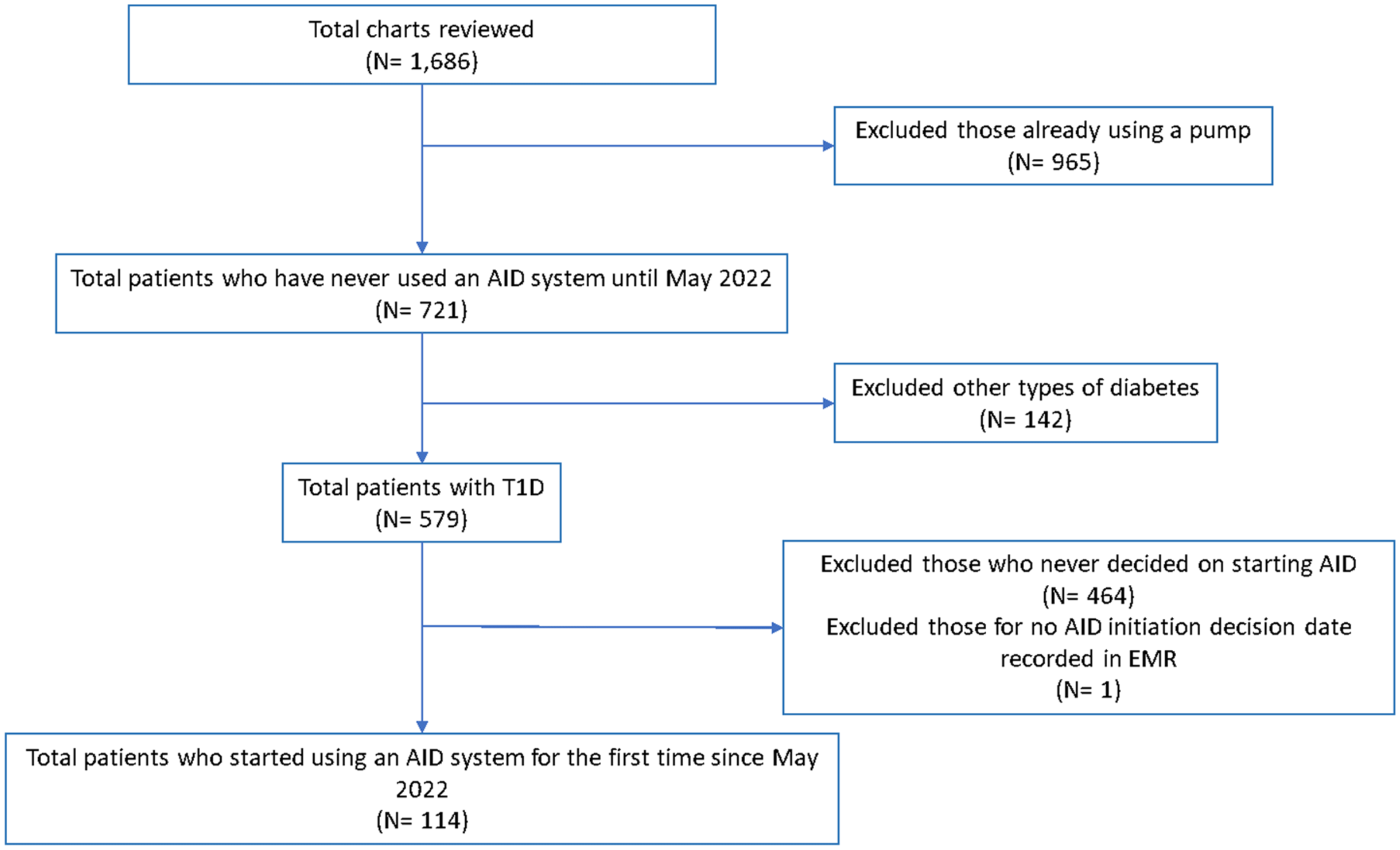
Patient inclusion.
Demographic and Clinical Characteristics
As shown in Table 1, in this cohort the median age was 38.9 years (IQR = 30.0, 56.1), 57% were male, 69% were non-Hispanic White, 21% non-Hispanic Black, and 4% identifying as Hispanic. Most patients had commercial insurance (75%), and median area deprivation index was 4.0. The median duration of diabetes was 10.2 years (IQR = 3.5, 18.1) and median HbA1c was 7.7% (IQR = 6.6, 8.9). Patients started the following AID systems: Omnipod 66.7%, Tandem 16.7% (12.3% T:Slim and 4.4% Tandem Mobi), iLet Bionic Pancreas 8.8%, and Medtronic 7%. Patients with public insurance were more likely to be female (58.6% vs 37.6%,
Characteristics of Those Deciding to Start an AID System.
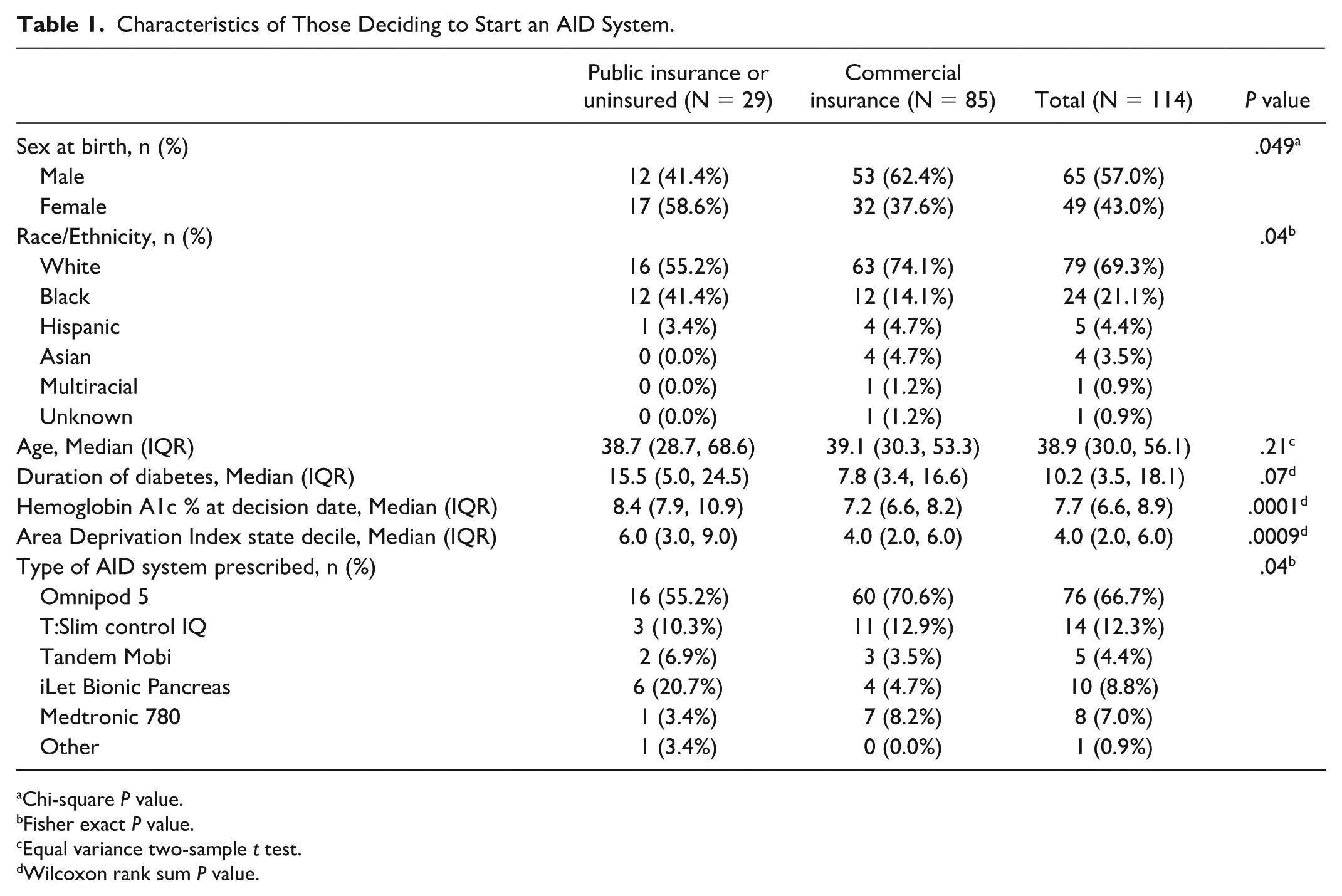
Chi-square
Fisher exact
Equal variance two-sample
Wilcoxon rank sum
TT-AID System Initiation
Median TT-AID, that is, time from decision to initiate an AID system to AID use, was 89.5 days (IQR = 49, 132), with the longest time segment being from decision to AID training 82.5 days (IQR = 43, 122).
Among patients who attended an AID selection visit, median time from decision to selection visit was 24 days (IQR = 10, 59). The median time from decision date to AID prescription date was 58 days (IQR = 21, 98), and from prescription date to AID training was 42 days (IQR = 20.5, 68.5). Patients who had an AID selection visit had median time from decision to AID initiation of 112 days (IQR = 79, 144), as shown in Figure 2a.
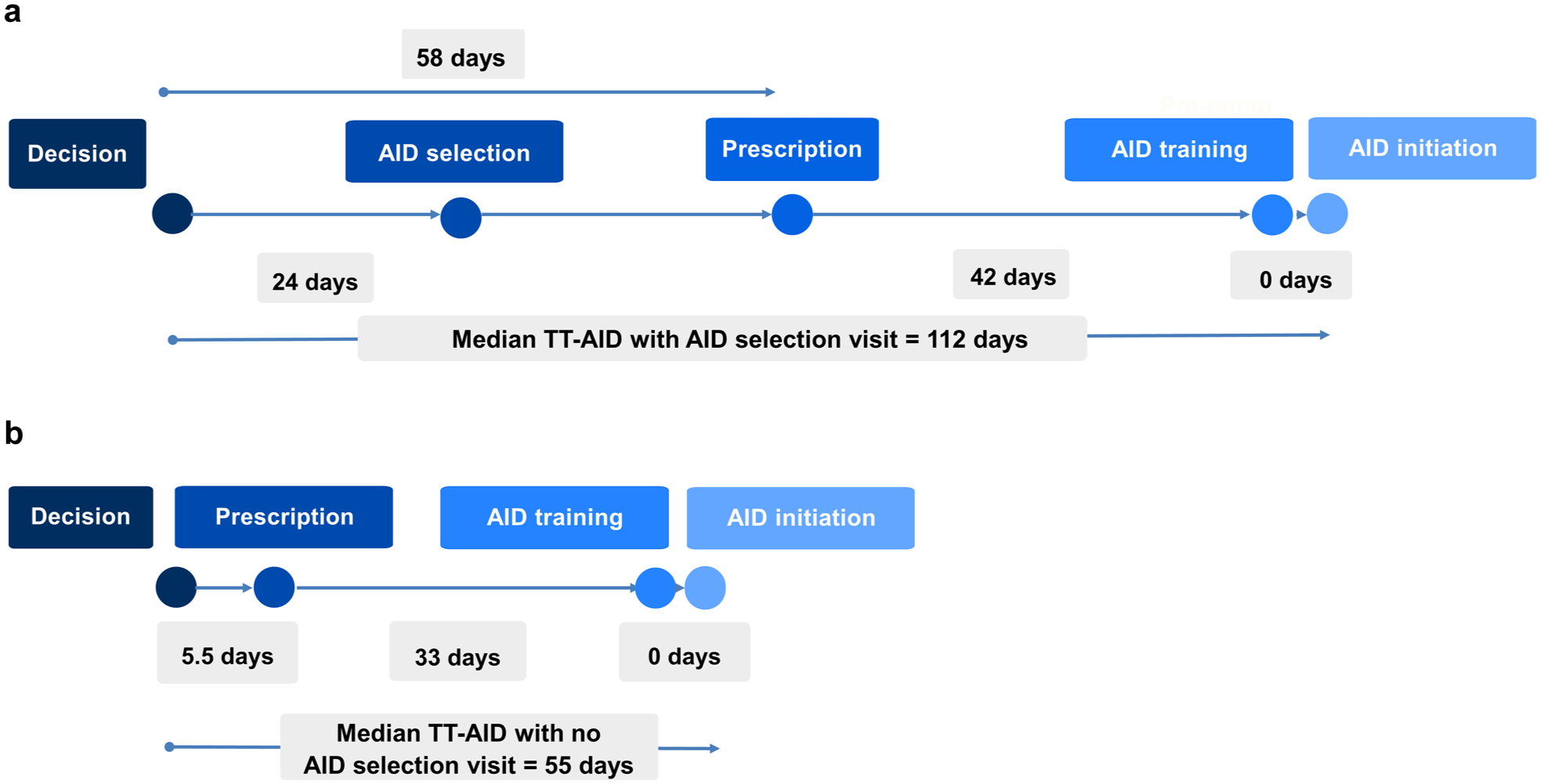
(a) Median time taken at each stage of AID initiation with AID selection visit. (b) Median time taken at each stage of AID initiation with no AID selection visit.
For patients who did not attend the AID selection visit (selected AID system at visit with provider), median time from decision to prescription date was 5.5 days (IQR = 1, 22), and prescription date to AID training was 33 days (IQR = 24, 55). The overall median time from decision to AID initiation in this group was 55 days (IQR = 35, 98); shown in Figure 2b.
Although both groups had comparable durations from prescription to AID training, patients who participated in the AID selection visit had significantly longer times from decision to prescription (58 vs 5.5 days,
Time to Automated Insulin Delivery System Initiation Stratified by AID Selection Visit.
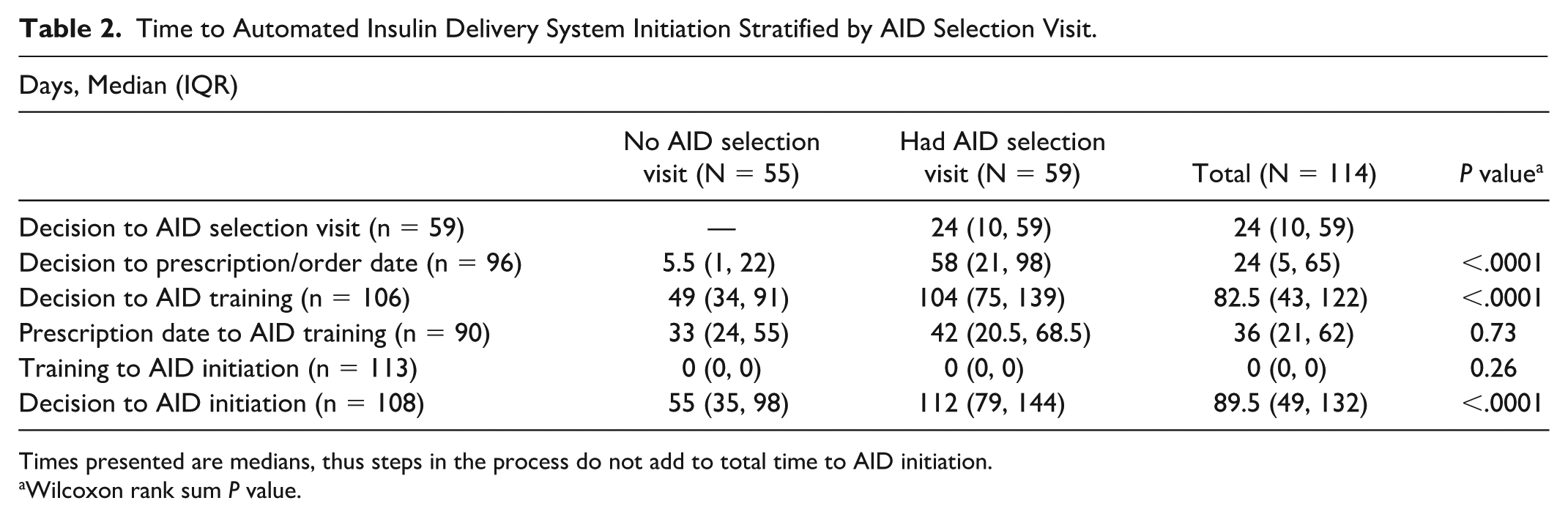
Times presented are medians, thus steps in the process do not add to total time to AID initiation.
Wilcoxon rank sum
Automated insulin delivery system training was conducted by vendor representatives for 34 patients, while the remaining 75 received training with internal educators. There was no significant difference in TT-AID (
Time to Automated Insulin Delivery System Initiation by Insurance Type
When focusing on short-term AID initiation, by right-censoring AID initiations taking place after 6 months, Kaplan-Meier time-to-event analysis showed differences in TT-AID between patients with public or no insurance versus commercial insurance. Time-to-event analysis demonstrated individuals with commercial insurance had shorter median TT-AID, 86 (69, 98) days versus 122 (67, 195) days,
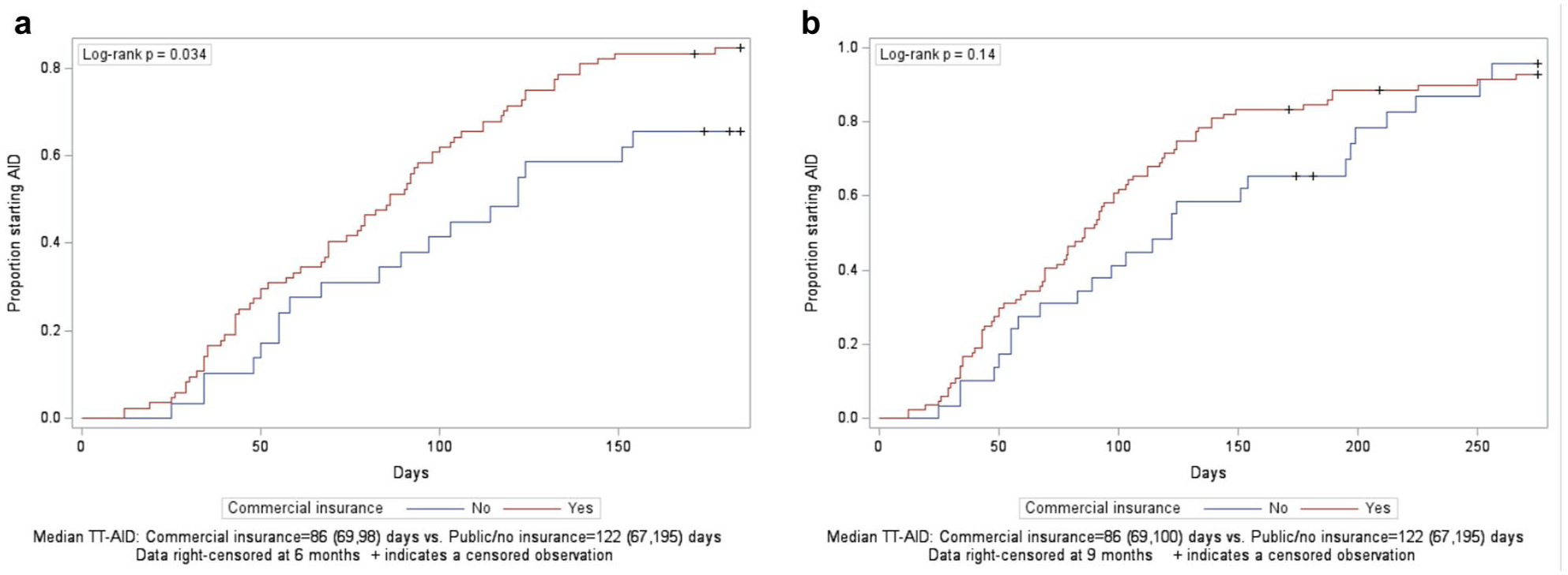
Inverse Kaplan-Meier curve illustrating the cumulative proportion of adults starting AID therapy, stratified by insurance type. (a) Represents data right-censored at 6 months. + indicates censored observation. (b) Represents data right-censored at 9 months. + indicates censored observation.
Factors Associated With TT-AID
A multivariable Cox proportional hazards model including insurance type, age, duration of diabetes, sex, race/ethnicity, and AID pickup at pharmacy was fit with right censoring at 6 months to demonstrate early effects influencing TT-AID. In the first 6 months after decision, those with commercial insurance had a 92% higher hazard of TT-AID, indicating faster TT-AID (hazard ratio [HR] = 1.92; 95% confidence interval [CI] = [1.13, 3.27];
Cox Proportional Hazard Model for the Hazard Rate of Time to Automated Insulin Delivery System Initiation.
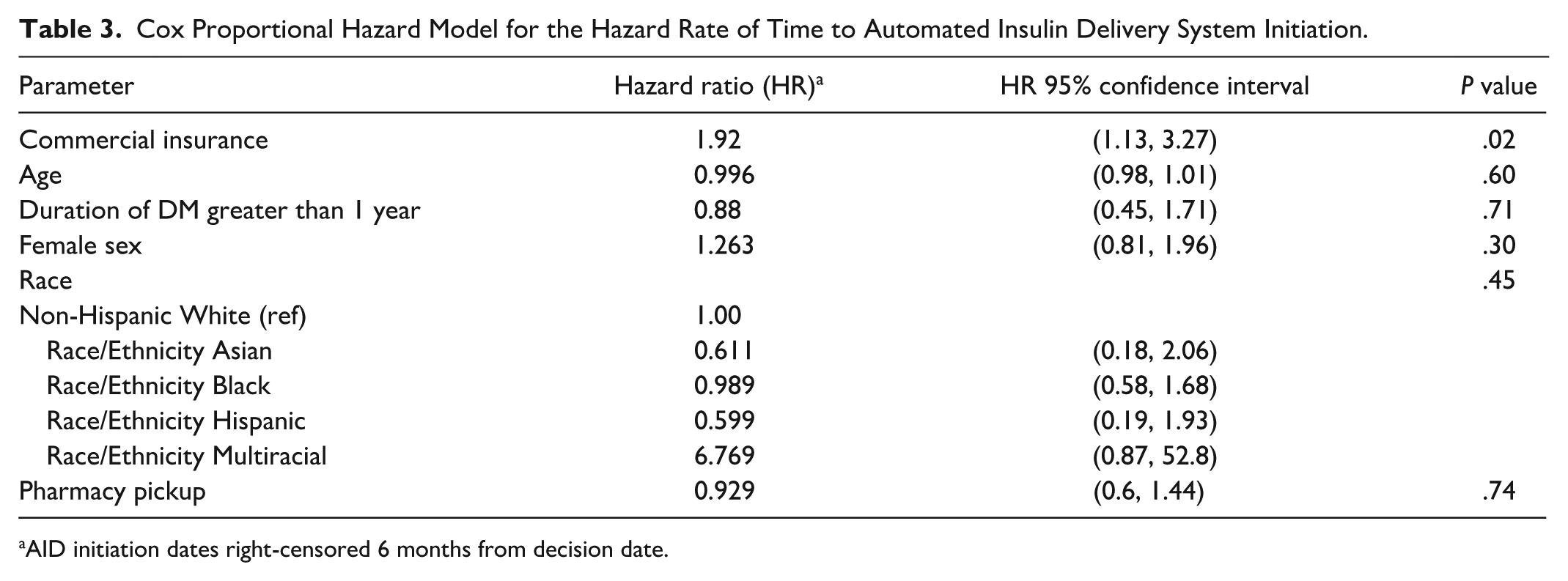
AID initiation dates right-censored 6 months from decision date.
Missing Data
Order dates for AID systems were missing from 18 subjects. Of these, 15 AID were ordered through DME and the date was unavailable in EMR, 2 obtained devices with another provider’s prescription, and 1 discontinued the AID initiation process, with no documented reason identified in the EMR. Automated insulin delivery system training dates were missing for 39 subjects. Of these, 34 trained with vendor representative and 5 did not continue to start an AID. There was no statistically significant difference in TT-AID for those with missing order dates.
Discussion
This retrospective study revealed that it took over 3 months from the time a patient decided to start an AID system to actual AID use, and the longest time was between decision to AID training. To our knowledge, this study is among the first to quantify time taken for AID initiation and identify factors delaying the complex AID initiation process. In this study, we showed that patients who attended an extra AID system selection visit and those with public health insurance had a significantly longer TT-AID.
Patients who attended an AID selection visit experienced a significantly longer TT-AID compared with those who did not attend, but rather selected an AID at their regular diabetes clinic visit with their provider. While the AID selection visits prolonged TT-AID, education provided at these visits is important.19-21 These education sessions help patients make informed choices about their AID system options and provide anticipatory guidance on management of AID-related issues. Longer TT-AID may reflect time taken to make appointments, limited appointment availability, and increased time required to choose an AID. Therefore, potential solutions might include streamlining the process while also incorporating patient education and AID system selection. A quality improvement initiative at a tertiary children’s hospital in Canada reduced pump wait times by conducting shortened readiness workshops and by using digital tools. 22 These findings support the use of flexible, patient-centered models to provide education on diabetes technologies. Asynchronous virtual modules or group sessions that could maintain informed decision-making and improve efficiency could expedite access to this beneficial technology.
In the first 6 months after AID system initiation decision, our study identified that patients with commercial insurance had shorter TT-AID compared with those with public insurance. Disparities in diabetes technology use based on insurance are well-described.13,23,24 Existing literature demonstrates that patients with public insurance face barriers related to complex eligibility requirements and delays in prior authorization (PA) approvals, often leading to further delays in use of diabetes technology.25,26 Furthermore, in our cohort, patients with public insurance were more likely to live in higher area deprivation index deciles, indicating greater socioeconomic disadvantage compared with those with commercial insurance. Individuals living in socioeconomically deprived areas often face multiple barriers to accessing diabetes technologies, including limited health care access, unreliable transportation, complex PA processes, and financial constraints that can lead to missed appointments and delayed treatment. 27 While association between TT-AID and insurance type persisted in the multivariable Cox model, we did not find a significant association between TT-AID and higher area deprivation index. Similarly, in our cohort, those with public insurance were more likely to be female. However, sex was not associated with a longer TT-AID in the multivariable model. This aligns with prior studies showing no consistent link between sex and diabetes technology uptake after adjusting for socioeconomic factors.2,24
Interestingly, we found that 9 months after AID initiation decision date, gaps between TT-AID based on insurance closed, suggesting that patients experience notable delays early after AID initiation decision. A recent article highlighted that AID therapy should be initiated early after diagnosis to reduce risk of disparities and improve glycemic outcomes. 28 Delays occurring within 6 months from AID decision, observed among publicly insured patients, reflect a critical window that quality improvement initiatives could focus on.2,24 Ongoing efforts to reduce disparities in diabetes technology use include a multicenter study evaluating the implementation of an EMR-based best practice advisory designed to standardize prescribing practices and promote equitable access to diabetes technologies. 29
We found no significant difference in TT-AID based on prescription pathway. We initially hypothesized that the pharmacy route, which involves fewer administrative steps, may be easier, thereby taking less time for patients to acquire an AID. 30 These findings suggest that the pharmacy route does not lead to faster initiation but may still help reduce patient and provider burden related to the AID acquisition paperwork. 30
One of the strengths of this study is that it is among the first to examine the duration it takes for individuals to begin AID systems from AID initiation decision, and the factors affecting AID initiation. We also investigated multiple AID systems and prescription pathways, allowing us to capture a broader view of available options for patients. Furthermore, we included a diverse cohort of patients and incorporated area deprivation index in our analysis to assess sociogeographic disparities in TT-AID. However, a key limitation of this study is its single-center design. Since our results only reflect the AID acquisition process within one health system, these findings may not be generalizable to other settings. A unique factor at our center is the ability to provide flexibility in training options. Patients can be trained by a vendor representative or an in-house diabetes educator. While specific steps may vary, the overall process remains consistent across most centers. Therefore, the findings from our study could serve as a starting point for future research and may inform other centers as they navigate similar optimization processes. This study captures experiences of patients starting AID therapy for the first time and does not account for those transitioning to an AID system from non-AID or different AID systems. In addition, the retrospective nature of the study restricts our ability to establish causality. Data are also limited by what is available in EMR, which includes missing data points, and may not fully capture nuances such as patient decision-making, provider recommendations, or other systemic barriers to AID initiation not documented in EMR.
Conclusions
This study identified the long TT-AID and two contributors to delayed AID system initiation: attending an additional AID system education visit and having public insurance. Future quality improvement efforts should explore targeted interventions to streamline this process, reduce TT-AID, and improve overall AID uptake. In addition, prospective studies with larger and more diverse samples could help further evaluate sociodemographic and AID-related factors contributing to AID initiation timelines.
Footnotes
Abbreviations
A1C, hemoglobin A1c; AID, automated insulin delivery; ADA, American Diabetes Association; CGM, continuous glucose monitor; CI, confidence interval; DME, durable medical equipment; DM, diabetes mellitus; EMR, electronic medical record; HR, hazard ratio; ICD-10, International Classification of Diseases, 10th Revision; IQR, interquartile range; IRB, institutional review board; MDI, multiple daily injections; NIH, National Institutes of Health; PA, prior authorization; QR, quick response; REDCAP, research electronic data capture; T1D, type 1 diabetes; TT-AID, time to automated insulin delivery initiation.
Authors’ Note
This study was presented as a poster at the American Diabetes Association 85th Scientific Sessions in Chicago, IL 2025.
Author Contributions
RMW, NM, AS, and EAB conceived of the study. YV, NP, and YCK collected data. EAB completed the statistical analyses. EAB, YV, and NP created figures and tables. YV, RMW, and EAB wrote the manuscript. All authors made critical contributions to the manuscript. All authors edited, reviewed and approved the manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RMW reports receiving research support from Lilly Diabetes, Novo Nordisk and Sanofi outside the submitted work. NM reports receiving research support from Breakhtrough T1D and Samsung Research America outside the submitted work. YV, NP, YCK, EAB, and AS have no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Institutes of Health under award R01-DK134955 to RMW and NM.
Data Availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
