Abstract
Background:
This study aimed to investigate the decline over time in the proportion of total daily insulin delivered as boluses in newly diagnosed youth with type 1 diabetes using a hybrid closed-loop system.
Method:
A secondary analysis was conducted using data from the CLOuD study, an open-label, multicenter, randomized, parallel hybrid closed-loop trial to investigate bolus patterns in youth with newly diagnosed type 1 diabetes.
Results:
Over the 48-month trial period, the proportion of total daily insulin delivered as carbohydrate-related boluses decreased from 58% to 34%. There was a decreasing trend in the median (interquartile range) amount of carbohydrates entered per day from 236 (204, 253) g to 184 (127, 232) g, and the number of carbohydrate-related boluses per day from 5.5 (4.6, 6.5) to 3.7 (2.9, 5.2) over the 48 months. Mean ± SD daily carbohydrate-related bolus insulin increased from 15.1 ± 6.6 to 22.0 ± 9.0 units/d, and the amount of insulin delivered per 10 g of carbohydrate more than doubled from 0.6 (0.5, 0.8) units to 1.3 (0.9, 1.5) units. The postprandial change in glucose (measured as the difference between peak glucose 30 to 180 minutes post carbohydrate-related bolus and glucose on carbohydrate-related bolus delivery) changed from 49 (45, 54) to 59 (53, 66) mg/dL.
Conclusions:
The decline in the proportion of total daily insulin delivered for as bolus is likely attributable to a combination of missed boluses and under-bolusing, while the closed-loop algorithm compensates for the missed or insufficient carbohydrate-related insulin delivery by increasing basal insulin delivery.
Introduction
Type 1 diabetes is a chronic autoimmune condition characterized by insulin deficiency resulting from immune-mediated β-cell destruction. Following type 1 diabetes diagnosis endogenous insulin production declines over time leading to increased exogenous insulin requirements.1,2 In a previous analysis of hybrid closed-loop insulin delivery in youth newly diagnosed with type 1 diabetes for a period of four years follow-up, we observed an increasing proportion of closed-loop insulin delivered as basal rather than bolus insulin. The factors underlying the decline in the proportion of total daily insulin delivered as bolus have not yet been investigated.
Mealtime bolusing is reported as a challenge of diabetes management in adolescents and missed mealtime boluses is frequently cited as a contributing factor to suboptimal glucose outcomes in this age group.3,4 We aimed to assess whether missed boluses or other factors could explain the reducing proportion of insulin delivered as boluses over time in this cohort.
The aim of this post hoc analysis was to evaluate bolus insulin patterns in newly diagnosed youth with type 1 diabetes using a hybrid closed-loop system, and to explore why the proportion of total daily insulin delivered as boluses decreases over time.
Methods
Study Design and Population
A secondary analysis of an open-label, multicenter, randomized, parallel hybrid closed-loop trial (CLOuD trial) was undertaken to assess bolus insulin delivery patterns in the first 48 months following type 1 diabetes diagnosis. Participants aged ≥10 and <17 years were recruited within 21 days of diagnosis of type 1 diabetes and were randomized to either hybrid closed-loop or standard insulin therapy for 24 months. Participants were offered a 24-month optional extension phase with the allocated treatment. Participants aged 16 years and parents/guardians of participants <16 years opting to continue in the extension phase were asked to reconsent. Written assent was obtained from participants <16 years. The present analysis includes only participants who used hybrid closed-loop therapy. The study was approved by an independent research ethics committee.
Closed-Loop System
The Cambridge model predictive control algorithm (version 0.3.71; CamDiab Ltd, Cambridge, UK) was implemented in two hardware configurations sequentially. The FlorenceM configuration consisted of an unlocked Android smartphone running the algorithm which communicated with a modified, next-generation sensor-augmented 640G Medtronic insulin pump (Medtronic MiniMed, California) through a proprietary translater, and a Medtronic continuous glucose monitor (CGM) transmitter with Guardian 3 sensor. The CamAPS FX configuration used an unlocked Android smartphone to house the CamAPS FX closed-loop app, a Dana Diabecare RS insulin pump (Sooil Development, Seoul, Korea) or YpsoPump (Ypsomed, Burgdorf, Switzerland), and either the Dexcom G6 (Dexcom, San Diego, California) or FreeStyle Libre 3 (Abbott Diabetes Care, Alameda, California) CGM.
Both configurations used an algorithm housed in a mobile app to automatically adjust the insulin infusion rate every 8 to 12 minutes. All insulin delivered by the algorithm was considered basal insulin. The system was initialized by entering the user’s weight and total daily dose, while insulin sensitivity and active insulin time were automatically calculated and adjusted as necessary. The algorithm used adaptive learning with respect to total daily insulin dose, diurnal variation, and insulin delivery around meals. Insulin pump basal rates were preset by the user and only used if the system was not in automode. These do not influence the algorithm insulin delivery. The insulin to carbohydrate ratio (ICR) was entered into the system at set-up and used by the algorithm for meal bolus calculations. All “non-carbohydrate”-related insulin delivery was manually administered by the user.
Data Analysis
Metrics were calculated daily for each participant and then summarized over three-month (91‑day) periods for the first 24 months and six-month (182-day) periods for the second 24 months. Time periods were calculated from the day after diagnosis. All metrics were summarized with equal weighting across participants. To be included in the analysis, data were only considered if participants spent at least 50% of each 24-hour period in closed-loop mode. Additionally, a minimum of 30 days of data were required for inclusion in each 91-day period and at least 60 days for each 182-day period.
Metrics included total daily carbohydrate-related bolus insulin (defined as a bolus delivered within 5 minutes of a carbohydrate entry), the proportion of total insulin delivered as carbohydrate-related boluses, number of carbohydrate-related boluses per day, total daily carbohydrates entered per day, units of insulin delivered per 10 g of carbohydrate (ICR), postprandial change in glucose, and time in target glucose range between 70 and 180 mg/dL. Insulin delivered per 10 g of carbohydrate was calculated by dividing the total bolus insulin delivered during each 91- or 182-day period by the total amount of carbohydrate entered during that same period and multiplying that by 10. Based on typical timing of postprandial glucose excursions, postprandial change in glucose was defined as the difference between the peak glucose level (found within 30 to 180 minutes after a carbohydrate-related bolus) and the glucose at the time of the carbohydrate-related insulin bolus delivery. 5
Data analysis was completed using R Studio (R Foundation for Statistical Computing, Vienna, Austria). Data are presented as median (interquartile range, IQR) or mean ± standard deviation (SD).
Results
A total of 48 participants (23 female and 25 male) aged 10 to 16.9 years were included in the analysis (Table 1). One participant was excluded as an outlier due to behavioral (deliberate carbohydrate restriction) factors impacting on insulin requirements. The trends in carbohydrate-related insulin boluses and glycemic outcomes are shown in Figure 1.
Demographics.
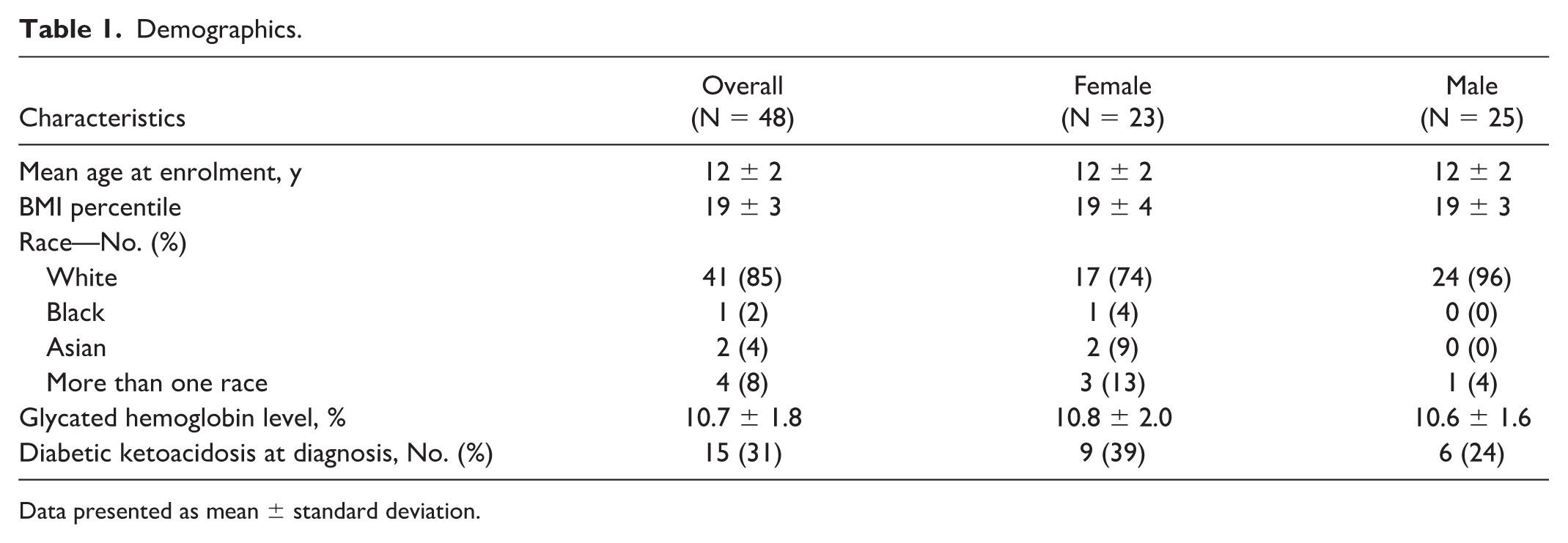
Data presented as mean ± standard deviation.
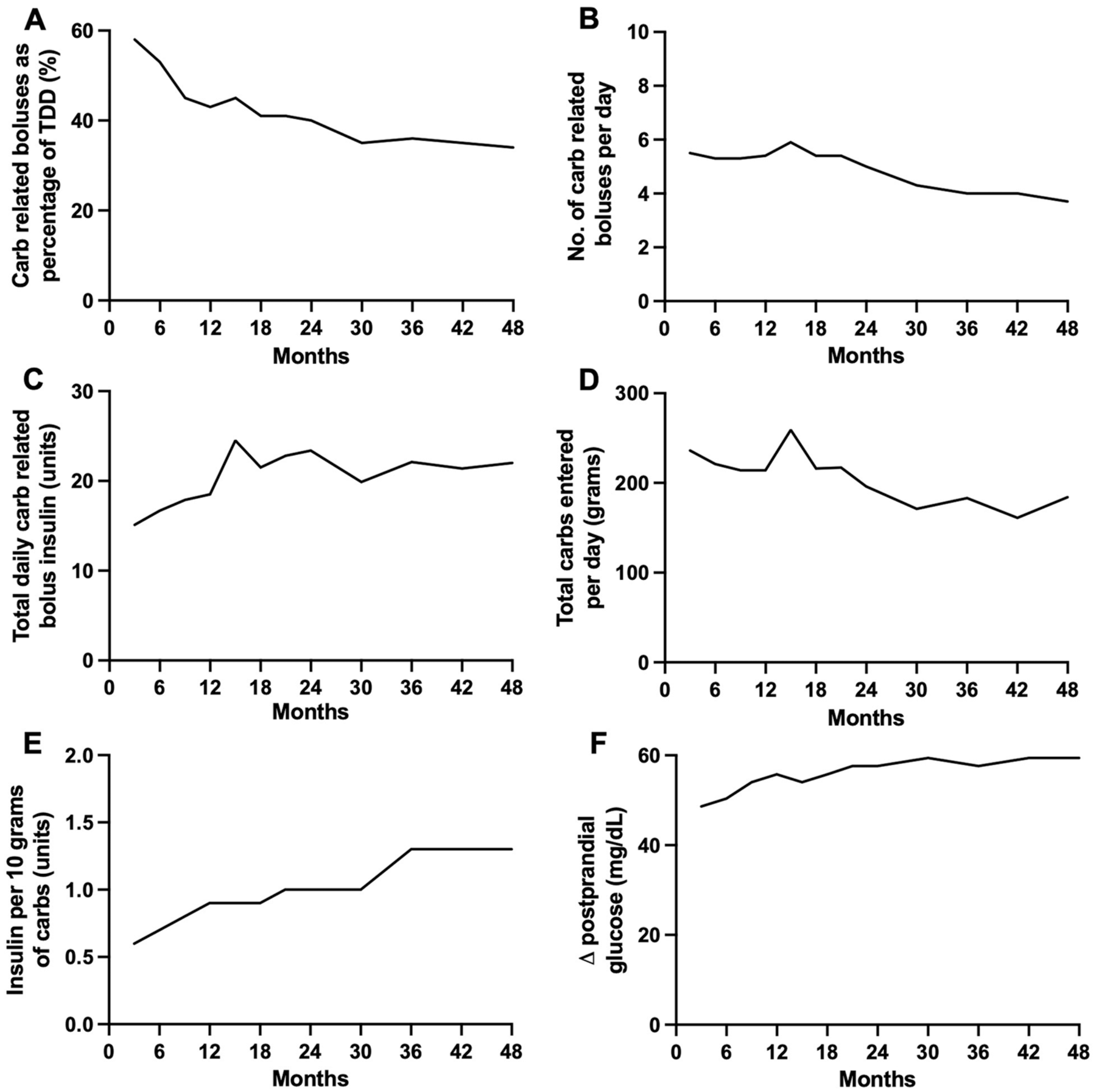
Trends in carbohydrate-related insulin boluses and glycemic outcomes during hybrid closed-loop insulin delivery over the 48 months following diagnosis of type 1 diabetes: (a) Proportion of total daily insulin as carbohydrate-related bolus insulin (%; mean). (b) Carbohydrate-related bolus frequency (per day; median). (c) Total daily carbohydrate-related bolus (units; mean). (d) Total carbohydrate entered per day (g; median). (e) Insulin delivered per 10 g of carbohydrate (median). (f) Change in postprandial glucose calculated as difference between peak glucose 30 to 180 minutes post carbohydrate-related bolus and glucose on carbohydrate-related bolus delivery (mg/dL; median).
Bolus Insulin Delivery
The proportion of total daily dose of insulin accounted for by carbohydrate-related insulin boluses declined from 58% at zero to three months to 34% at 42 to 48 months (Table 2, Figure 1a). The median (IQR) number of carbohydrate-related boluses administered per day decreased from 5.5 (4.6, 6.5) in the first three months to 3.7 (2.9, 5.2) in the final three months (Table 2, Figure 1b).
Bolus Patterns and Glycemic Outcomes.
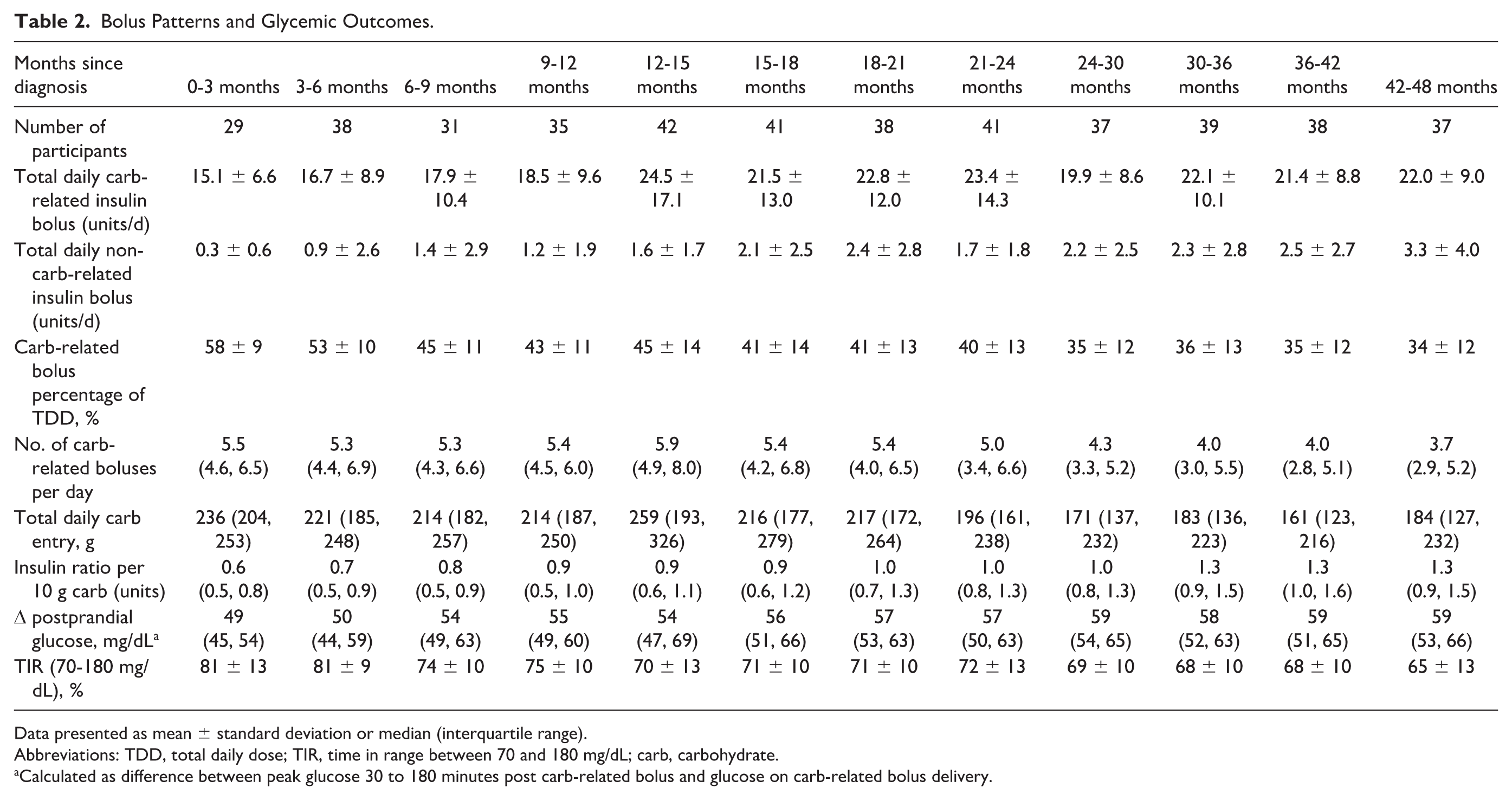
Data presented as mean ± standard deviation or median (interquartile range).
Abbreviations: TDD, total daily dose; TIR, time in range between 70 and 180 mg/dL; carb, carbohydrate.
Calculated as difference between peak glucose 30 to 180 minutes post carb-related bolus and glucose on carb-related bolus delivery.
The absolute number of units of insulin delivered for carbohydrate intake per day increased by approximately 1.5-fold from 15.1 ± 6.6 units/d at zero to three months to 22.0 ± 9.0 units/d at 4248 months. This increase occurred most rapidly during the first 15 months, rising from 15.1 ± 6.6 to 24.5 ± 17.1 units/d (Table 2, Figure 1c). Non-carbohydrate-related bolus delivery was low overall but also increased from 0.3 ± 0.6 units/d at months 0 to 3 after diagnosis to 3.3 ± 4.0 units/d during months 42 to 48 (Table 2).
Carbohydrate Entry
Total carbohydrate entered into the closed-loop app per day decreased over the study period by approximately 22% from 236 (204, 253) g to 184 (127, 232) g (Table 2, Figure 1d).
The ratio of insulin delivered per 10 g of carbohydrate increased by just over twofold over the 48 months from 0.6 (0.5, 0.8) units in months 0 to 3 to 1.3 (0.9, 1.5) units in months 42 to 48 (Table 2, Figure 1e).
Glucose Outcomes
Despite the increasing ratio of insulin delivered per 10 g of carbohydrate, the postprandial glucose excursion increased by approximately 20% over the 48 months from 49 (45, 54) mg/dL at 0 to 3 months to 59 (53, 66) mg/dL at 42 to 48 months (Table 2, Figure 1f). Time in target glucose range (70 to 180 mg/dL) showed a trend toward a decrease from 81 ± 13% at zero to three months to 65 ± 13% at 42-48 months (Table 2).
Subgroup Analysis (Time in Range ≥70%)
We completed a subanalysis of individuals with a mean time in target glucose range of ≥70% (n = 30). The trends in this cohort reflect the same trends observed in the overall population in this study. However, the small sample size limits the interpretation of this finding.
Discussion
Our analysis suggests that the decreasing proportion of total daily insulin delivered as boluses may be driven by a combination of reduced carbohydrate-related bolus frequency and inadequate adjustment of carbohydrate ratios or carbohydrate estimation.
The initial number of carbohydrate-related boluses (5.5 times per day) was comparable to a prior study analyzing bolus delivery patterns in adults with type 1 diabetes and an HbA
The total amount of insulin delivered for carbohydrate intake each day increased despite fewer boluses. This was reflected in the 1.5-fold rise in insulin ratio per 10 g of carbohydrate, suggesting that insulin-to-carbohydrate ratios are being adjusted over time. This increasing in insulin requirements is expected immediately following a type 1 diabetes diagnosis and later due to insulin resistance often occurring during growth and puberty.1,2,9 The increase in postprandial glucose excursions over the 48-month study period, alongside a decreasing proportion of bolus insulin relative to total daily dose despite adjustments to insulin-to-carbohydrate ratios, suggests that these adjustments were inadequate. The insulin-to-carbohydrate ratios likely remained too weak, or carbohydrate entry is increasingly underestimated over time, potentially due to growing portion sizes in this population.
Changes in basal insulin delivery directly reflect the closed-loop algorithm adjustments. In contrast, bolus insulin delivery is user-initiated and is primarily associated with carbohydrate intake. The decreasing proportion of total daily insulin accounted for by carbohydrate-related boluses, combined with the observed trends, suggests the algorithm is delivering insulin to compensate for missed boluses or insufficient insulin delivery surrounding carbohydrate intake. The algorithm compensates for suboptimal carbohydrate-related bolusing and is likely to be delivering insulin later in response to postprandial glucose spikes rather than insulin ideally having been administered as a premeal bolus. While hybrid closed-loop systems can partially compensate for inaccurate or even missed meal boluses, this is not always sufficient to stop postprandial glucose excursions due to the delay in insulin action.
Strengths of the present analysis include the long duration of hybrid closed-loop insulin delivery following diagnosis of type 1 diabetes in youth. Limitations include the small sample size (N = 48), limited ethnic diversity (85% white), the absence of dietary records and weight measurements for participants and the lack of pubertal status and psychosocial context which may impact on the results.
Conclusions
In conclusion, over the 48 months following type 1 diabetes diagnosis, the decline in the proportion of total daily insulin delivered as boluses is attributable to a combination of reduced carbohydrate-related bolus frequency and inadequate adjustment of carbohydrate ratios or carbohydrate estimation. The closed-loop algorithm partially compensates for missed or insufficient carbohydrate-related bolus insulin; however, additional education surrounding carbohydrate counting, meal blousing, and ICR adjustments may be beneficial.
Footnotes
Acknowledgements
None
Abbreviations
carb, carbohydrate; CGM, continuous glucose monitor; ICR, insulin-to-carbohydrate ratio; IQR, interquartile range; SD, standard deviation; TDD, total daily dose; TIR, time in range.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JW has received speaker honoraria from Ypsomed and Novo Nordisk and is a consultant at CamDiab. JMA has received consultancy fees from CamDiab. MEW reports patents related to closed-loop and being a consultant at CamDiab. SH serves as a member of Medtronic advisory board, is a director of Ask Diabetes Ltd providing training and research support in health care settings, and reports having received training honoraria from Medtronic and Sanofi and consulting fees for CamDiab. TR receives consultancy fees from Abbott Diabetes care, has received honoraria from Novo Nordisk for delivering educational meetings, and has received honoraria from Sanofi for delivering educational meetings and participating in advisory boards. REJB reports receiving speaker honoraria from Eli Lilly and Springer Healthcare, reports sitting on the Novo Nordisk UK Foundation Research Selection Committee on a voluntary basis, acted as an independent advisor for Provent Bio, and received speaking honoraria from Sanofi and Medscape, which were donated to an education research fund. FMC has received honoraria for speakers fees and Advisory Board attendance from Abbott, Lily, Sanofi, Medtronic, and Insulet. RH reports having received speaker honoraria from Eli Lilly, Dexcom, and Novo Nordisk, receiving license fees from BBraun; patents related to closed-loop, being consultant to Abbott Diabetes Care, and being director at CamDiab. CKB is a consultant at CamDiab and has received speaker honoraria from Ypsomed. CR, AT, AG, DE, and NT report no conflicts of interest related to the present paper.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the National Institute for Health Research (NIHR) EME Programme (14/23/09), the Helmsley Trust (2016PG-T1D045 and 2016PG-T1D046), JDRF (22-2013-266 and 2-RSC-2019-828-M-N), and Diabetes UK (24/0006785). CR is funded by a Diabetes UK PhD Studentship. RH is a recipient of the EASD-Novo Nordisk Foundation Diabetes Prize for Excellence. CKB is a recipient of the EFSD/Novo Nordisk Foundation Future Leaders Award. Additional support for the artificial pancreas work from National Institute for Health Research Cambridge Biomedical Research Centre and National Institute for Health Research Oxford Biomedical Research Centre. Abbott Diabetes Care supplied free glucose monitoring devices, and Dexcom and Medtronic supplied discounted continuous glucose monitoring devices. Medtronic supplied discounted insulin pumps, phone enclosures, continuous glucose monitoring devices, and pump consumables. The views expressed are those of the author(s) and not necessarily those of the NIHR, the Department of Health and Social Care, or other funders.
