Abstract
Background:
Hybrid closed loop (HCL) technology is now standard of care for women with type 1 diabetes in pregnancy in the United Kingdom, but there is minimal evidence to guide HCL use in the preconception period, peripartum, and postnatally. We used real-world data to assess whether HCL use offered benefits upon glycemia in the preconception, peripartum, and postnatal periods.
Methods:
This single-center retrospective observational study assesses the effect of HCL use upon HbA1c and continuous glucose monitoring (CGM) metrics, including time-in-range (TIR; 3.9-10.0 mmol/L; 72-180 mg/dL) or pregnancy time-in-range (TIRp; 3.5-7.8 mmol/L; 63-140 mg/dL) before (n = 46), during (n = 21), and after (n = 25) pregnancy. Data (mean (SD)) were analyzed using paired t tests (limit P < .05).
Results:
Preconception initiation of HCL was associated with a reduction of HbA1c from 62.4 (14.0) to 54.2 (7.7) mmol/mol at three to six months (7.9 (1.3) to 7.1 (0.7) %; P < .0001). The TIR increased from 49% at baseline to 65% at one week (P < .001) and 72% at six months (P < .001) after initiation. Time-below-range (TBR) fell from 3.2% at baseline to 2.1% at one week (P = .006) and 2.1% at three months (P = .042). Pregnancy initiation of HCL was associated with a reduction of HbA1c from 61.2 (14.6) to 48.1 (8.6) mmol/mol at three months (n = 36; P < .0001) and increased TIRp (37% baseline to 57% after one week; P < .0001). Patients using HCL postnatally at one month had TIR 70% and TBR 1.8%.
Conclusions:
When started preconception or in pregnancy, HCL significantly reduces HbA1c at three months and improves TIR by 15% to 20% within one week.
Introduction
Pregnancy in women with type 1 diabetes (T1D) is associated with increased maternal and fetal complications compared with women without diabetes. 1 Suboptimal maternal glycemic control can further elevate those risks, increasing the risk of congenital anomalies, preterm delivery, preeclampsia, operative delivery, and large-for-gestational age in infants.2-4 Hybrid closed loop (HCL) systems are now available to support patients to attain stricter pregnancy-specific glucose targets by adjusting insulin in real time throughout all stages of pregnancy.1,2 However, data are lacking on the effect of HCL upon preconception, perinatal, and postpartum outcomes in clinical practice.
Recent clinical trial data show that HCL can help people with T1D to lower HbA1c without raising hypoglycemic events. 1 In pregnancy, the recent Automated Insulin for Type 1 Diabetes in Pregnancy (AIDAPT) clinical trial demonstrated that HCL use was associated with a 10% improvement in maternal pregnancy-specific time-in range (TIRp) in women with T1D who started the HCL in early pregnancy. 2 In recognition of the benefits of HCL use, the National Institute of Health and Care Excellence (NICE) released a technology appraisal (TA 943) recommending HCL therapy for all adults with T1D with HbA1c > 7.5% (59 mmol/mol), with a five-year implementation plan, that set out high priority groups including pregnancy women or those planning pregnancy. 3 While HCL use is likely to be beneficial preconception and postnatally, there is limited evidence for its use in those settings.
Preconception care is imperative for women living with T1D and plays a pivotal role in improving health outcomes. 3 Effective preconception care can mitigate the risks of congenital anomalies, miscarriage, stillbirth, and neonatal mortality by achieving optimal glycemic control (HbA1c level below 48 mmol/mol (6.5%) for a minimum of three months) before conception.1,3,4 However, achieving and sustaining appropriate glycemic control during the preconception phase presents distinct challenges. There is limited evidence about the ability of HCL systems to attain prompt preconception glycemic targets without causing hypoglycemia, to support preparation for pregnancy.
The use of HCL in peripartum and postnatal care is, therefore, becoming more common, but there is minimal evidence to guide postpregnancy care. Achieving optimal glycemic control postnatally is often very difficult. Previous studies have shown some benefits of HCL use upon reducing rates of hypoglycemia.4,5 We aimed to assess the clinical effects of HCL use before, during, and after pregnancy in women with T1D using real-world data from our clinical center, where HCL use has been rapidly adopted.
Methods
All patients with a history of T1D who received preconception and/or antenatal care at University Hospitals of Leicester NHS Trust from June 2023 to October 2024 were identified retrospectively and included in this approved service evaluation. All women who reported planning pregnancy in the next six to 12 months were offered specific preconception care. This included information about pregnancy-specific glucose ranges, high-dose folic acid, and the option to start a HCL system in accordance with the high priority groups under NICE TA943 implementation guidance. 3
Clinical Data Collection
Data about preconception care and complications were collected from electronic clinical records. Baseline characteristics including body mass index (BMI) and ethnicity were gathered from a centrally hosted clinical computer system—SystmOne (TPP Ltd, London, UK). Glucose and insulin data from continuous glucose monitoring (CGM) and HCL systems were obtained from approved platforms (Clarity, LibreView, CareLink, Glooko). Retinopathy status was obtained from OptoMize eye screening software. Obstetric history and pregnancy outcomes were retrieved from the hospital E3 electronic medical records. Clinical information such as HbA1c was gathered from clinical lab system ICE.
Definitions of Outcomes
Neonatal hypoglycemia was defined as a plasma glucose level of less than 2.6 mmol/L within the first 24 hours of life. Neonatal intensive care unit (NICU) admission included babies admitted for a period of at least 24 hours within the first 28 days of life. Babies born before 37 weeks of gestation were considered preterm. LGA was calculated using GROW and defined as having a birthweight >90th centile.
Measurement of Glycemia
The HbA1c was measured locally using an IFCC-aligned Tosoh high-performance liquid chromatography analyzer. Continuous glucose monitoring metrics, including standard time-in-range for preconception and postpregnancy periods (TIR; 3.9-10.0 mmol/L; 72-180 mg/dL) or pregnancy-specific time-in-range during pregnancy (TIRp; 3.5-7.8 mmol/L; 63-140 mg/dL), were collected from Dexcom Clarity or Abbott Libreview for the two weeks prior to the start of HCL (baseline). The TIR, TIRp, time-below-range (TBR and TBRp) and time-above-range (TAR and TARp), average glucose, and Coefficient of Variation (CV) were recorded. For CGM data after HCL initiation and in the postnatal period, we identified the date of delivery and downloaded CSV files. For each time point during pregnancy, two weeks of CGM data was downloaded from Glooko or Carelink and relevant glucose metrics were calculated.
Statistical Analysis
Variables are presented as mean (SD) or n (%). Statistical analysis was performed using Stata version 16.0 (significance level P < .05) using paired t tests (Figure 1) and unadjusted linear/logistic regression. Analyses have also been provided with adjustment for maternal age, booking BMI, and ethnicity (white, black, or Asian). Missing data were not imputed.
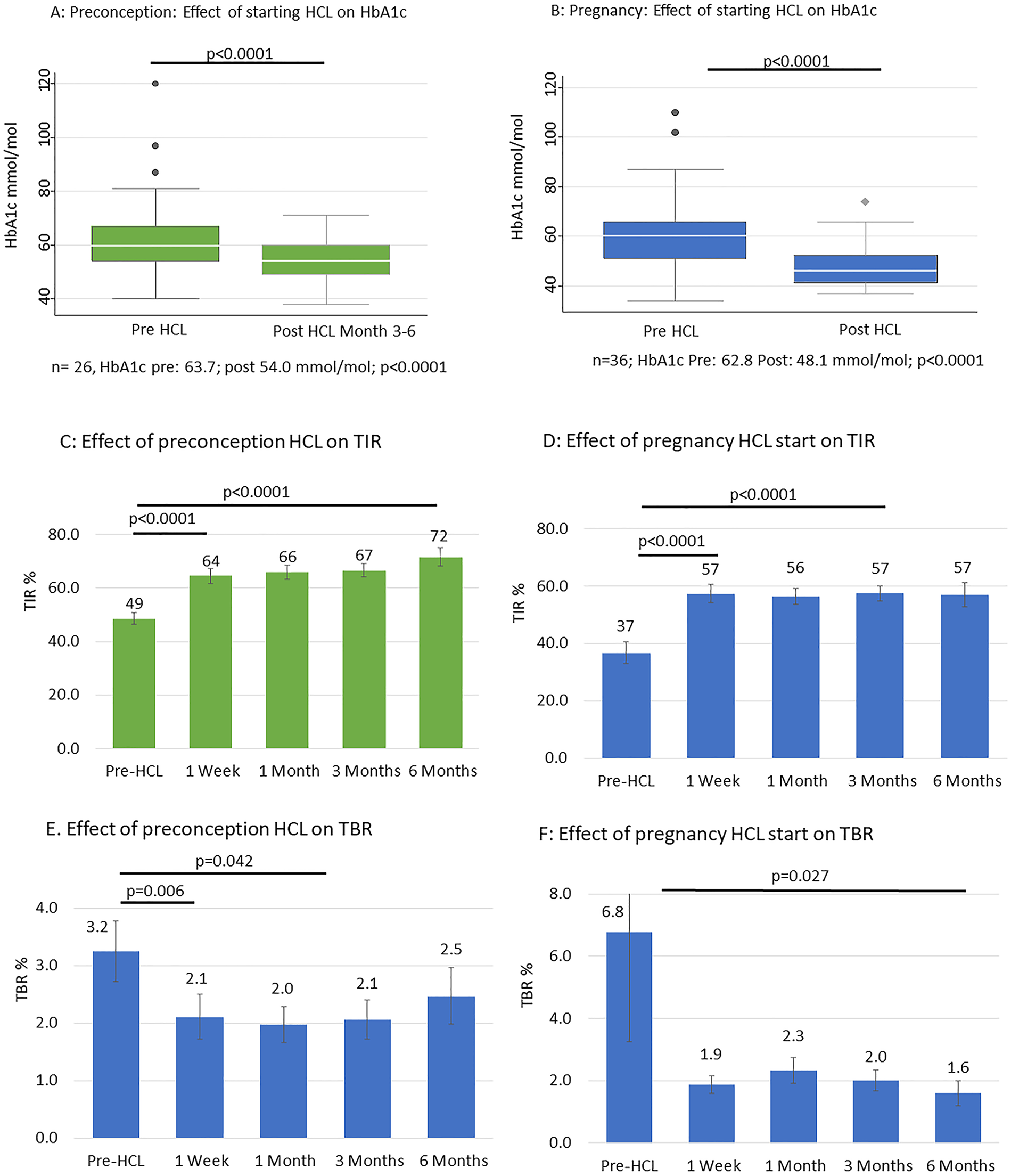
Effect of HCL initiation preconception (green) or in pregnancy (blue) upon HbA1c, time in range (TIR) and time below range (TBR). Error bars show standard error of the mean.
Results
Baseline Characteristics
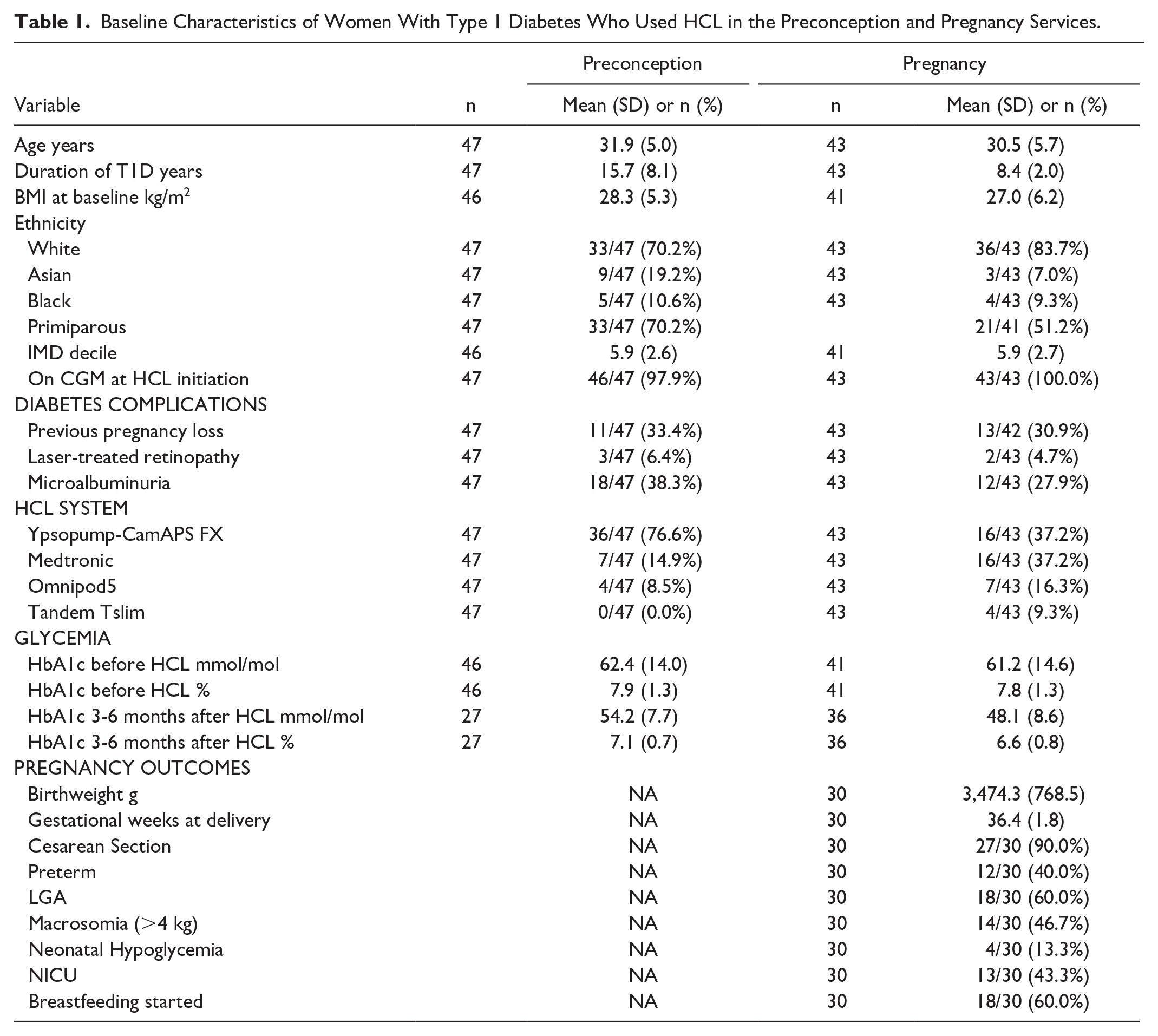
In our service, 47 women started HCL in the preconception period and 43 women used HCL in pregnancy (25 pregnancy HCL start; 18 preconception HCL start; Table 1). Women with HCL use preconception had similar baseline characteristics in terms of age, BMI, ethnicity and parity to women who started HCL in pregnancy, but had a longer duration of T1D (mean (SD): 15.7 (8.1) years vs 8.4 (2.0) years) and higher baseline time in range (48.6% (14.6) preconception cohort; 34.0% (20.1) pregnancy cohort). Women who started HCL in the preconception period were more likely to be using the Ypsopump−CamAPS system, while pregnancy HCL users used a range of different HCL systems.
Baseline Characteristics of Women With Type 1 Diabetes Who Used HCL in the Preconception and Pregnancy Services.
HCL Use and Glycemia in the Preconception Period
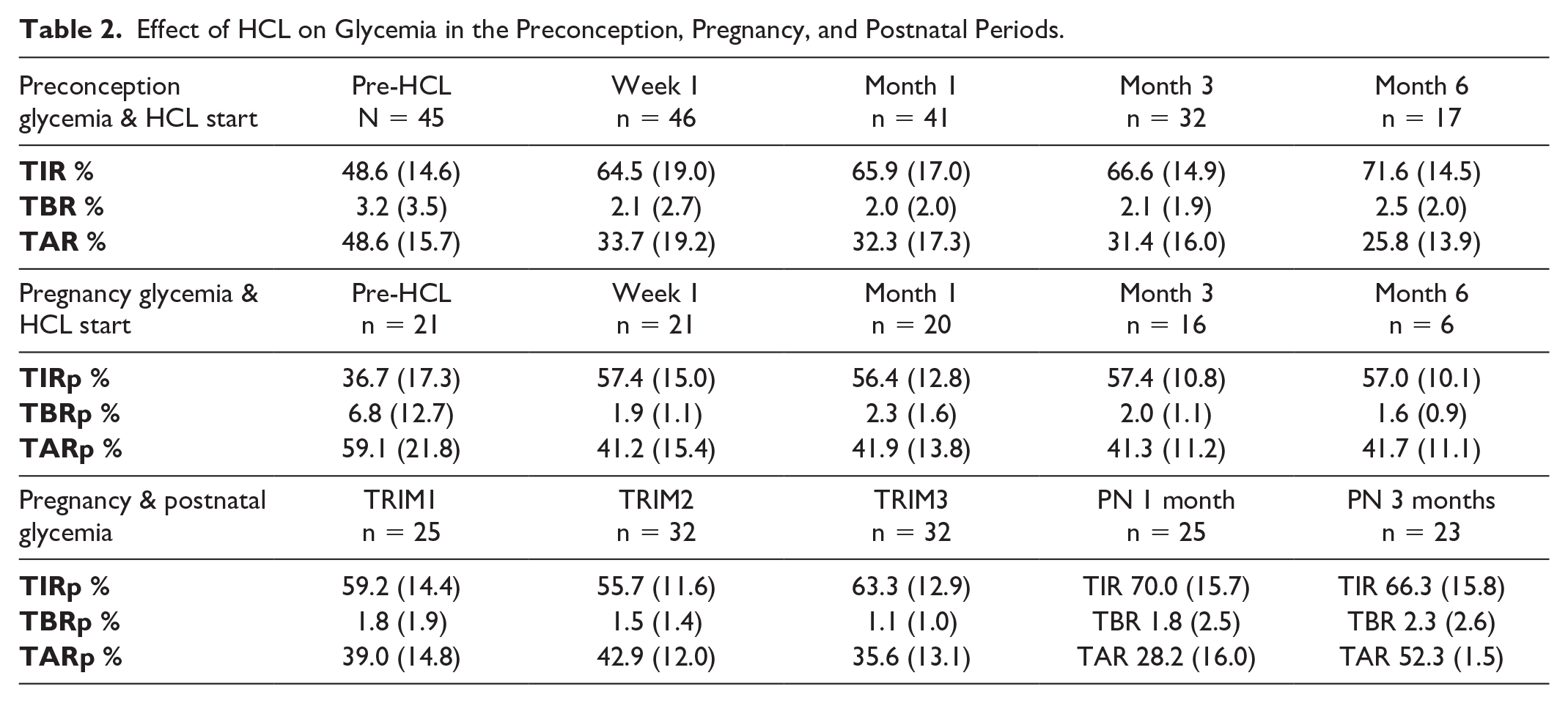
Starting HCL in the preconception period was associated with a reduction in HbA1c from 62.4 (14.0) to 54.2 (7.7) mmol/mol at three to six months (7.9 (1.3) to 7.1 (0.7) %; P < .0001; Figure 1; Table 2) and an increase in TIR (pre-HCL 48.6% (14.6) to 64.5% (19.0) after one week (P < .001) and 71.6% (14.5) at six months (P < .001). The TBR reduced from 3.2% (3.5) at baseline to 2.1% (2.7) at one week (P = .006) and 2.1% (1.9) at three months (P = .042).
Effect of HCL on Glycemia in the Preconception, Pregnancy, and Postnatal Periods.
HCL Use and Glycemia in Pregnancy
Pregnancy initiation of HCL was associated with a reduction of HbA1c from 61.2 (14.6) to 48.1 (8.6) mmol/mol at three months (7.8 (1.3) to 6.6 (0.8) %; n = 36; P < .0001). Pregnancy initiation was associated with an increase in TIRp (pre-HCL 36.7% (17.3) to 57.4% (15.0) at one week (P < .0001) and 57.4% (10.8) at three months (P < .0001; n = 15; Figure 1). The TBRp also reduced from a baseline of 2.7% (11.8) to 1.7% (1.3) at six months (P = .027; n = 15).
Comparison of HCL Initiation in Pregnancy or Preconception Period Upon Pregnancy Outcomes and Postnatal Health
Most women (29/30) used the HCL during labor and delivery (27/30 had a cesarean delivery). One woman required a variable rate insulin infusion (VRII) due to intercurrent illness. All other patients used HCL during labor and delivery. No significant differences in glycemia or clinical outcomes were observed between women who started HCL in the preconception period compared with during pregnancy (Table 3). Women who started an HCL in the preconception period had a nonsignificantly higher TIRp (by around 5%) in each trimester and a nonsignificantly lower TBR in the second trimester (Table 3).
Effect of Preconception Versus Pregnancy HCL Start Upon Antenatal and Postnatal Glycemia and Pregnancy Outcomes.
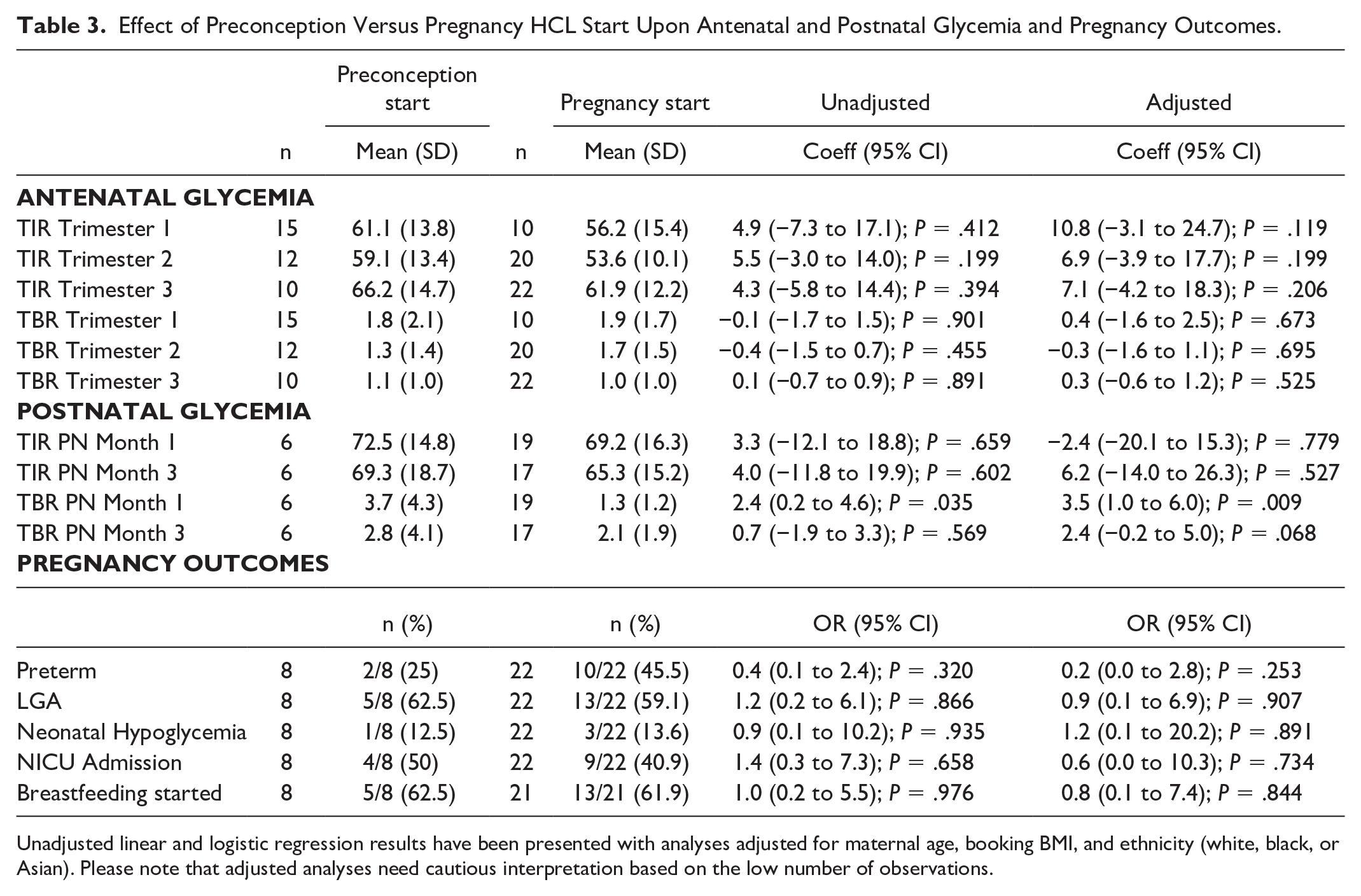
Unadjusted linear and logistic regression results have been presented with analyses adjusted for maternal age, booking BMI, and ethnicity (white, black, or Asian). Please note that adjusted analyses need cautious interpretation based on the low number of observations.
Pregnancy outcomes appeared similar when comparing women with a preconception versus pregnancy HCL start. However, preterm delivery rates were nonsignificantly higher (45.5%) in women with a pregnancy HCL start compared with women who started in the preconception period (25.0%). There were no significant differences in rates of LGA, neonatal hypoglycemia, or admission to the NICU.
Postnatal Outcomes
Mean hospital stay was 2.75 days (SD: 2.38) with a range from one to 12 days. Longer stays were linked to preterm delivery, neonatal admission, and transitional care for the newborn or mother awaiting medical investigations. Rates of breastfeeding were similar in women with a pregnancy versus preconception HCL start. The HCL use in the postpartum period was associated with near-optimal glycemic control with a TIR of 70.0% (15.7) at one month and 66.3% (15.8) at three months. The TBR was 1.8% (2.5) at one month and 2.3% (2.6) at three months (Tables 2 and 3).
Discussion
Statement of Principal Findings
This single-center, real-world, observational study demonstrates that the benefits of HCL upon glycemia in pregnant women with T1D described in a clinical trial setting are also evident in real-world clinical practice, in a socioeconomically diverse, multiethnic population. Indeed, in our practice, we identified a 15% improvement in TIR in one week after preconception initiation and a 20% improvement in TIRp after HCL initiation in pregnancy, suggesting that the benefits may be even more marked in real-world clinical practice, where the baseline HbA1c may be higher than that seen in randomised controlled trial (RCT) settings.2,6 We found benefits of HCL use upon glycemia in the preconception period, which was not previously studied in RCT settings.2,5 Our data overall confirm that HCL use is feasible and beneficial in diverse populations, across language and cultural barriers. Among our preconception cohort, 19% of affected women were Asian and 11% were black, showing that HCL initiation is acceptable and beneficial to women with diverse cultural and socioeconomic backgrounds. Our results confirm that HCLs offer a highly effective and safe method for optimizing glycemia in the preconception, pregnancy, and postnatal periods.
Strengths and Weaknesses of Study
This was a single-center observational cohort with limited patient numbers and variability in the timing of antenatal booking and HCL initiation. This was not a randomized controlled trial, which has been done before. 2 The cohort was observed under standard clinical care in a public health care system and included individuals from diverse ethnic, cultural, educational, and socioeconomic backgrounds, making the findings broadly applicable to people with T1D. Our population was spread across deprivation deciles and included patients of minoritized ethnicity, who are often underrepresented in clinical trials. Notably, standard clinical care revealed that many women with pronounced hyperglycemia previously labeled as being “under-engaged” with services achieved excellent results with HCL therapy. Our findings reinforce existing evidence-based recommendations, from NICE, 3 supporting HCL therapy during pregnancy as the optimal standard of care for individuals with type 1 diabetes, 2 and that benefits can be seen in diverse groups according to ethnicity, culture, language, and education.
Our population originally used multiple different HCL systems, as there was no evidence to support one choice over another for pregnancy prior to November 2023, when the AIDAPT results were published. Virtually all patients who initiated HCL for pregnancy planning or in pregnancy since then were started on the Ypsopump-CamAPS system. As it takes six to nine months to obtain pregnancy outcome data, we did not have sufficient data to segregate results by HCL system. The different HCL systems in use and inclusion of two different ranges for CGM data (TIRp (pregnancy) 3.5-7.8 mmol/L; TIR prepregnancy 3.9-10.0 mmol/L) make it difficult to compare TIR across the two cohorts. This study did not have sufficient numbers of patients to assess the accuracy or algorithm performance of the different systems.
Relevance to Other Studies: Preconception HCL Use
The HCL use has rarely been studied in the preconception period. Our preconception cohort showed 15% improvement in TIR one week after starting HCL in the preconception period, with a total increase of 23% at six months. Although there are no recommendations for TIR targets to demonstrate readiness for pregnancy, on average women achieved 72% TIR six months after starting an HCL, which is similar to achieving HbA1c <48 mmol/mol (<6.5%) in alignment with national guidance. 3
Being ready for pregnancy after six months of preconception care is hugely beneficial to mothers, who are juggling factors such as life circumstances, caring responsibilities, age, fertility, and glycemia as they prepare for pregnancy. This rapid reduction in hyperglycemia enables early modification of the most important risk factor for pregnancy complications in people with type 1 diabetes, glycemia, likely to result in benefits upon rates of preterm birth, large birth weight, and the requirement for neonatal care input.2,7
Relevance to Other Studies: Pregnancy HCL Use
The multicentre AIDAPT RCT 2 compared HCL use with the CamAPS system to standard care in 220 women with T1D in pregnancy. Overall, HCL users had a 10% improvement in TIRp. While patient numbers were limited and multiple HCL systems are currently in use in our study, we provide a real-world confirmation that HCL yields comparable benefits in routine clinical care for pregnant women with T1D. Indeed, our population had a 20% improvement in TIRp within one week of starting HCL in pregnancy. Our results demonstrating a rapid improvement in glycemia are consistent with AIDAPT results, showing that the initiation of HCL therapy during pregnancy (<14 weeks) caused rapid improvements in glycemia, within one week of commencing HCL therapy. 2 Improvements in TIR have been demonstrated in children (two to six years), as early as within one day of initiation of HCL therapy. This rapid modification of risk is essential in reducing adverse pregnancy outcomes in people with type 1 diabetes.
Despite a substantial improvement in glycemia in the first week of HCL use, many of our patients did not obtain recommended TIRp targets of >70% in pregnancy and HbA1c <6.5% (48 mmol/mol) in the preconception setting. This may be due to differences in baseline glycemia in pregnancy. Our pregnancy cohort had a baseline HbA1c 7.8% and TIRp 37%. Several individual patients in our pregnancy cohort started HCL with a baseline TIRp of zero and achieved substantial benefits with HCL use (consistent with the spread of data in Figure 1a and b). In the AIDAPT trial, women in the intervention group had a baseline HbA1c of 7.6% with baseline TIRp 49%. Women in the intervention group attained TIRp 68% on average during the intervention phase, a treatment difference of 10.5% compared with the standard care group. Conversely, in the CRISTAL trial, baseline HbA1c was 6.5% and TIRp was 59%, with TIRp rising to 67% in the intervention group over the study period. Our results suggest that while initiation of HCL systems gives immediate benefits to women in pregnancy, choice of system or device optimization (for example, through changing settings or patient bolusing behaviors) may be important to help users attain optimal glycemic targets, and hence optimal pregnancy outcomes.
Relevance to Other Studies: Peripartum HCL Use
Previously in our center, all women with type 1 diabetes were put on to a VRII during labor and delivery except for those having an elective Cesarean section. Following the initiation of HCLs, widely used during labor and delivery, only one patient needed a VRII during the study data collection period. The vast majority of patients used HCL for glycemic control during delivery, resulting in more control for patients and with beneficial implications for staffing in maternity services.
Relevance to Other Studies: Postnatal HCL Use
Postnatal glucose control in women after the AIDAPT trial showed continued benefits from HCL use, with HCL users achieving 72% TIR, with no increase in hypoglycemia. 8 Similar benefits were seen in our multiethnic, diverse population. Indeed, the use of HCL postpartum in our cohort was associated with around 70% TIR and low levels of hypoglycemia, below that of recommended targets, 9 and significantly lower than at baseline, prior to HCL initiation. During the postnatal period, there are multiple barriers to achieving 70% TIR (3.9-10 mmol/L). 9 There may be less incentive to achieve optimal glycemia, less time to focus on diabetes management due to the demands of infant feeding and sleep deprivation, and additional fear relating to hypoglycemia during childcare. 10 Breastfeeding, in particular, is a time at which people with type 1 diabetes fear hypoglycemia,4,11 and women with diabetes are less likely to breastfeed than those without diabetes. 5 Hypoglycemia detected via CGM (<3.9 mmol/L) is known to occur within three hours of breastfeeding, in 5% to 14% of mothers with type 1 diabetes. The use of closed loop therapy postpartum is recognized to reduce the incidence of hypoglycemia following breastfeeding. 5
Meaning of the Study and Implications for Clinical Care
Initiation of HCL in women in the preconception period or in pregnancy is feasible within a multiethnic, diverse population in an NHS setting. The HCL use was associated with a rapid improvement in glycemia, suggesting that the benefits of HCL use may be even greater in the real world compared with results from clinical trials. Patients who had previously been labeled as “under-engaged” made rapid progress on HCL systems. Women from Asian or Black backgrounds, often underrepresented in clinical trials, had excellent results from HCL systems in clinical practice.
We had insufficient number of observations to examine pregnancy outcomes, but there were lower preterm rates (25% vs 46%) if HCL was started preconception. However, women who choose to attend for preconception care are fundamentally different to women who have unplanned pregnancies; data between groups are difficult to compare directly. Future work should examine pregnancy outcomes in more detail, comparing preconception and pregnancy HCL start to assess whether there are truly benefits to starting in the preconception period.
Conclusions
The HCL use in the preconception, pregnancy, and postnatal periods is feasible and beneficial to women with type 1 diabetes from an ethnically and socioeconomically diverse population.
Footnotes
Acknowledgements
The authors would like to thank the multidisciplinary diabetes in pregnancy team at University Hospitals of Leicester NHS Trust, the diabetes pump team, and our patients. This research was supported by the National Institute for Health Research (NIHR) Leicester Biomedical Research Centre (BRC). The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care.
Abbreviations
AIDAPT, Automated Insulin for Type 1 Diabetes in Pregnancy clinical trial; BMI, Body mass index; CV, Coefficient of variation; HCL, Hybrid Closed-Loop; NHS, National Health Service; NICE, National Institute of Health and Care Excellence; T1D, Type 1 Diabetes; TAR, Time above range; TARp, Time above pregnancy-specific range; TBR, Time-below-range; TBRp, Time below pregnancy-specific range; TIR, Standard time-in-range 3.9-10.0 mmol/l; 72-180 mg/dl; TIRp, Pregnancy-specific time-in range; 3.5-7.8 mmol/l; 63-140 mg/dl; SD, Standard deviation.
Author Contributions
C.L.M. is the guarantor for this analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: C.L.M. is supported by the European Foundation for the Study of Diabetes—Novo Nordisk Foundation Future Leaders’ Award (NNF19SA058974). C.L.M.’s work on type 1 diabetes in pregnancy is supported by Diabetes UK under project grant number 22/0006456.
Data Availability
Data are available upon request, subject to approval from study steering groups.
