Abstract
The University of Chicago Monogenic Diabetes Registry (UCMDR) developed a participant portal to enhance engagement and data completeness in a large, longitudinal research study. Built in collaboration with the Center for Research Informatics (CRI), the portal integrates with REDCap to provide secure survey access, document exchange, and communication. Since its launch, 40% of invited participants have activated accounts. Among new enrollees, 88% of portal users completed their baseline survey, up from a historical 60%. Portal activation was higher among female participants (
Keywords
Introduction
The University of Chicago Monogenic Diabetes Registry (UCMDR) was formally founded in 2008 with the goal of establishing a systematic way of following participants with monogenic diabetes longitudinally. For the first several years, the UCMDR was maintained on a password-protected Microsoft Excel spreadsheet with clinical and genetic data from a few families. Due to rapid growth, it was transitioned into a REDCap database in 2011 and is now the largest monogenic diabetes registry in the United States with more than 1400 participants with a known genetic form of diabetes. In total, there are more than 4500 participants, which includes family member controls and those with suspected but unconfirmed monogenic diabetes. 1 Participants who enroll in the UCMDR are located throughout the entire United States, do not have to be University of Chicago Medical Center patients, and complete surveys electronically via REDCap. At time of enrollment, a baseline survey is delivered that includes questions about demographics, diabetes diagnosis history, birth history, diabetes clinical history, other medical problems, and family history of diabetes. A brief follow-up survey is then completed annually by participants to provide updates about possible changes in the status of their diabetes, medications, other medical problems, and changes in family history. Participant files are supplemented with medical records and genetic testing information when available. The UCMDR continues to play a prominent role in bridging evidence gaps within the various rare forms of monogenic diabetes, improving patient outcomes, and serving as a model for precision diagnosis and treatment for all forms of diabetes. 2
Nevertheless, the creation and maintenance of this legacy database has been challenging. Our understanding of monogenic diabetes has significantly changed since the origin of the UCMDR, and while the scientific understanding has grown considerably, the structure of the database and data collection tools has not always kept pace. Even with the implementation of periodic REDCap functionality improvements, an ongoing limitation was a suboptimal participant user experience, which has resulted in insufficient data completeness, and unsatisfactory survey completion rates hovering around 60%. For example, in the one-year period prior to portal implementation, 84 out of 131 new enrollees (64%) completed their baseline survey. Recognizing the importance of addressing this issue, we explored different solutions, including the possibility of a participant portal.
Patient-facing portals have become increasingly popular with more than 60% of hospitals giving patients access to view, download, and transmit their health data. The importance of such portals as drivers of satisfaction and engagement has been documented in several studies.3-6 We hypothesized that research registries could utilize this same technology to have timely, bidirectional communication with participants and to improve ongoing engagement in studies.
Robust participant data is of paramount importance to advancing our understanding and informing best practice recommendations for the various forms of monogenic diabetes. To increase data completeness and improve the research participant experience, we aimed to create a participant portal. Here, we describe the organizational and technical procedures that were undertaken in coordination with the UChicago Center for Research Informatics (CRI) to develop, pilot, and implement the UCMDR participant portal. We also review the preliminary results from participant feedback and utilization data and discuss future opportunities to augment portal interaction.
Methods
Initial REDCap Data Optimization
The University of Chicago Monogenic Diabetes Registry participant and operational data are maintained in a REDCap project and all surveys are delivered via automated REDCap functionality. Because study procedures are so deeply intertwined with REDCap, it was decided to design the portal in a way that interacts with REDCap instead of a standalone application. As such, as an initial precursor step to portal development, we undertook an internal optimization effort of our existing REDCap database. This six-month endeavor was multitiered. We performed a preliminary data clean-up phase to standardize fields and apply data validation requirements (eg, replacing open-text response with single- or multi-select questions that have defined permissible values). In addition, the baseline and follow-up surveys, which serve as our primary data capture instruments, were significantly upgraded to reflect advancements in monogenic diabetes research as well as REDCap functionality (eg, field embedding techniques with branching logic, action tags, data piping). Together, these upgrades streamline the user experience and ensure the collection of more complete and standardized data.
Engagement With UChicago CRI
After the internal database optimization phase was complete, a working group was established that included UCMDR and CRI team members. Initial meetings focused on the organizational and technical framework of the portal. Ultimately, the design was centered around the desired features and characteristics of the portal, including (1) serving as a central hub for participants to easily identify and complete all study tasks, (2) bidirectional communication capabilities, (3) user-friendliness. Working meetings and individual development effort progressed based on these criteria.
Participant Portal Technical Architecture
The portal is a web application that securely captures data in and displays data from the 21 Code of Federal Regulations Part 11 compliant instance of REDCap housed at the UChicago Kenwood Data Center and managed by the CRI.7,8 A separate portal database server that is also hosted in the HIPAA-compliant UChicago Kenwood Data Center supports user registration and linkage to REDCap participant information, authentication, authorization, and auditing. All web-based information transmission between the portal and REDCap and between the portal and end users are encrypted (Figure 1). The portal is accessible on both computers and mobile devices and has been designed with mobile-friendly features.
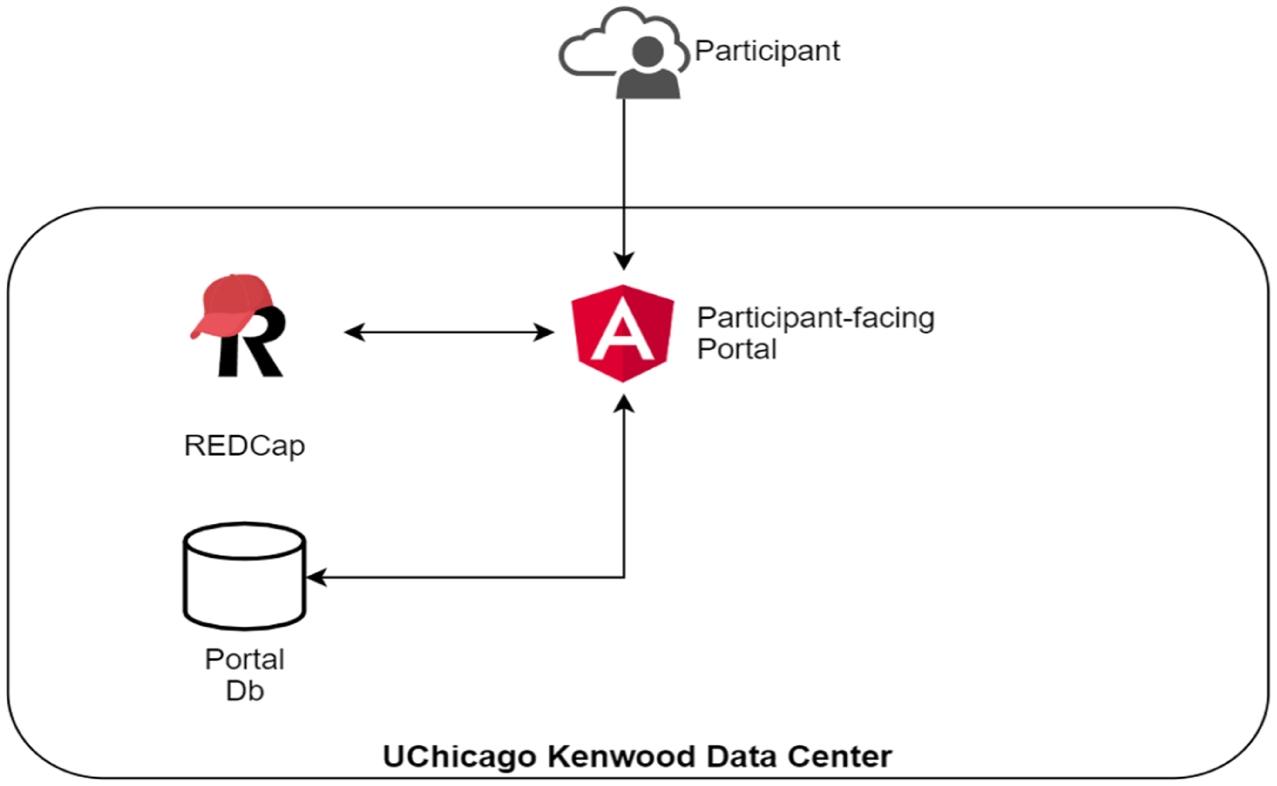
Participant portal technical architecture.
Prototype Testing Program
Following an initial development phase of nine months, a viable product was in place, and a two-month testing program was executed. This included an initial testing phase completed by internal UCMDR staff and subsequent testing by a five-person cohort of actively enrolled research participants. For each stage, detailed instructions were delivered to test all aspects of portal functionality including survey completion, document uploads, and document downloads. Feedback at each stage was collected and incorporated into iterative portal modifications.
Portal Live Launch
The final production version of the participant portal was launched in February 2023. Participants who meet eligibility criteria for the portal receive an email that contains a unique registration link and instructions for portal account activation. An initial blast of registration emails was sent out to 455 historical participants who were portal eligible (ie, had confirmed monogenic diabetes, had an active consent form on file, and had complete data fields required for identity verification). All prospective participants newly enrolled into the study can opt in to receive a portal invitation email. Participants who opt-out of the portal continue with study procedures through traditional methods via an email prompt and complete identical surveys as portal users.
Portal Feedback Survey
To capture user experience data, a feedback survey is emailed to portal users 90 days after account creation. There are four questions that use a Likert scale asking about usability and opinions on utility and features of the portal.
Results
Overall User Metrics and Uptake Results
To date, there have been 615 total portal invitation emails sent out to research participants. Two hundred forty-nine distinct participants have set up their portal accounts (40%). Seventy-one participants have accessed surveys through the portal, 32 participants downloaded a previously completed survey, and 12 participants have used the portal to upload a document for the study team to review. Complete participant portal usage metrics are displayed in Table 1.
Overall Participant Portal Usage Summary Among 249 Portal Users.
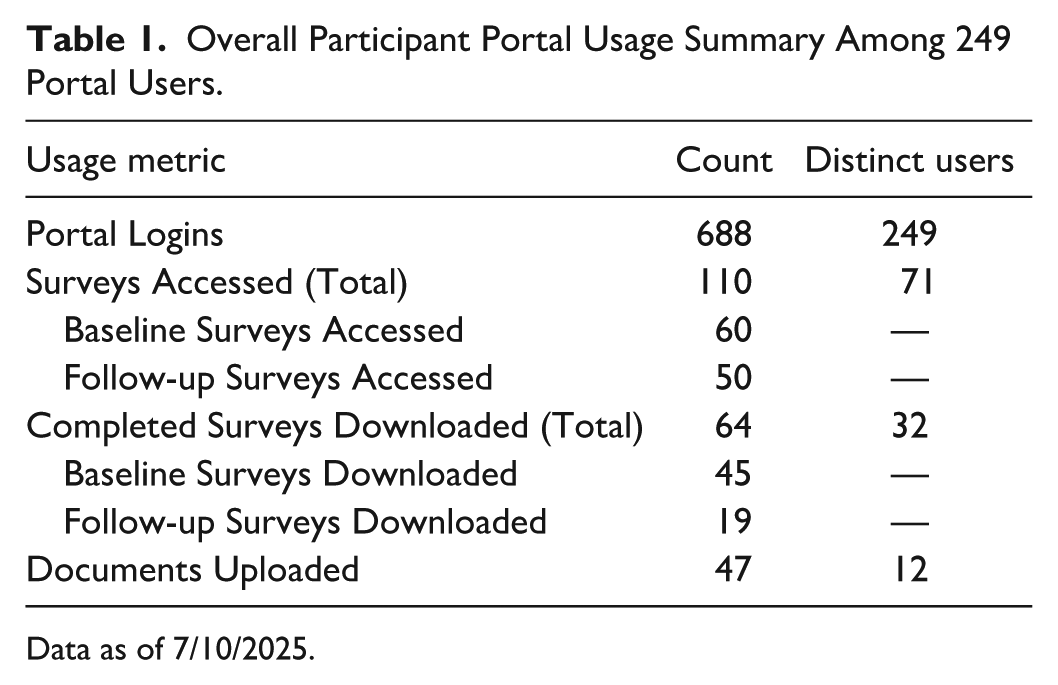
Data as of 7/10/2025.
To gain insight into the prospective portal opt-in and account set up rate in a normal enrollment process, we evaluated enrollments in a one-year timeframe from 7/1/2024 to 7/1/2025. In this period, there were 122 portal-eligible participants who enrolled in our study. Among these, 39 (32%) opted out of portal use, 33 (27%) opted in but did not activate their accounts, and 50 (41%) opted in and successfully activated their accounts. Of the 50 participants who activated their account, 44 completed their baseline survey (88%). Demographic and socioeconomic characteristics of new enrollees were examined to better understand factors associated with portal engagement (Table 2). Biological sex was significantly associated with portal engagement (
Demographic and Socioeconomic Characteristics of New Enrollees.
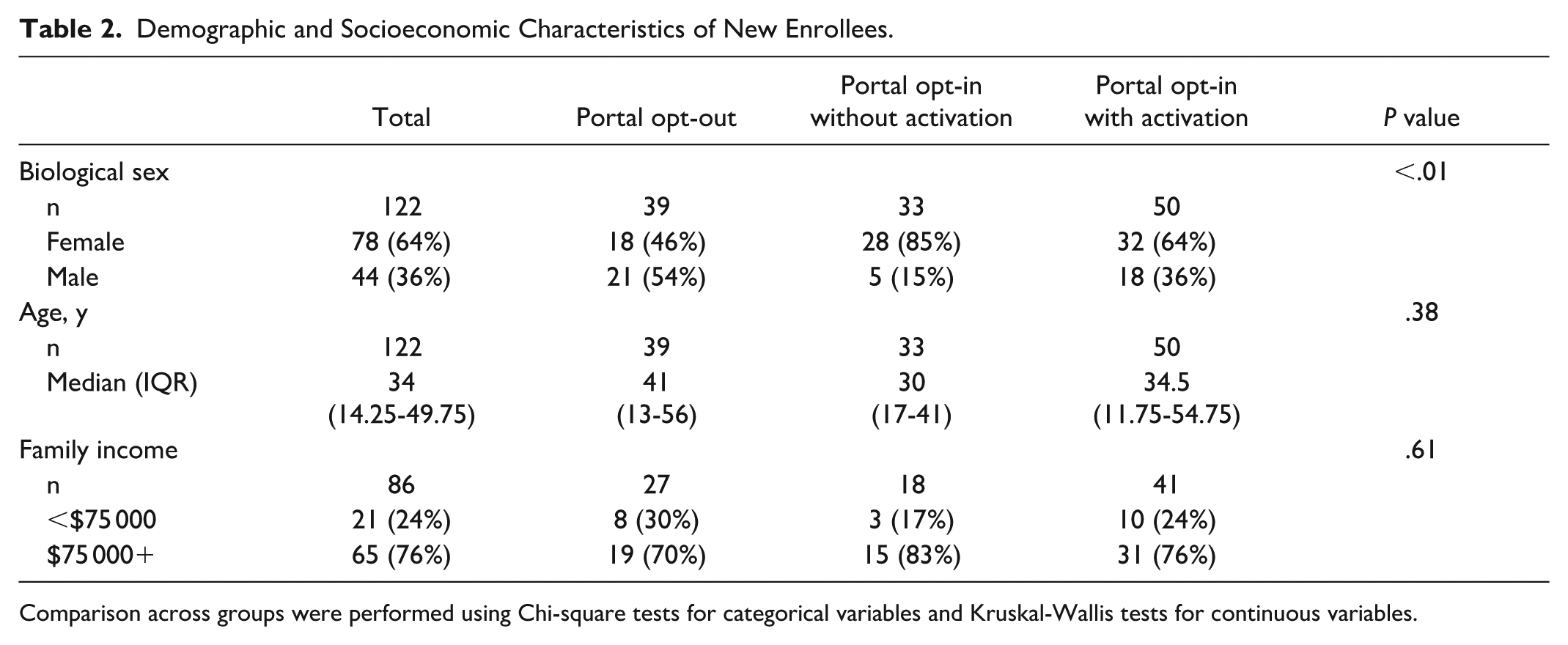
Comparison across groups were performed using Chi-square tests for categorical variables and Kruskal-Wallis tests for continuous variables.
Portal Feedback Survey
A survey asking for feedback about the portal was completed by 116 participants. Seventy-six percent found the platform user-friendly, and 64% reported it was helpful for viewing the consent form. A majority also agreed it was helpful for viewing previously completed tasks (62%) and improving task organization (52%). Fewer than 8% of participants disagreed with any item (Figure 2).
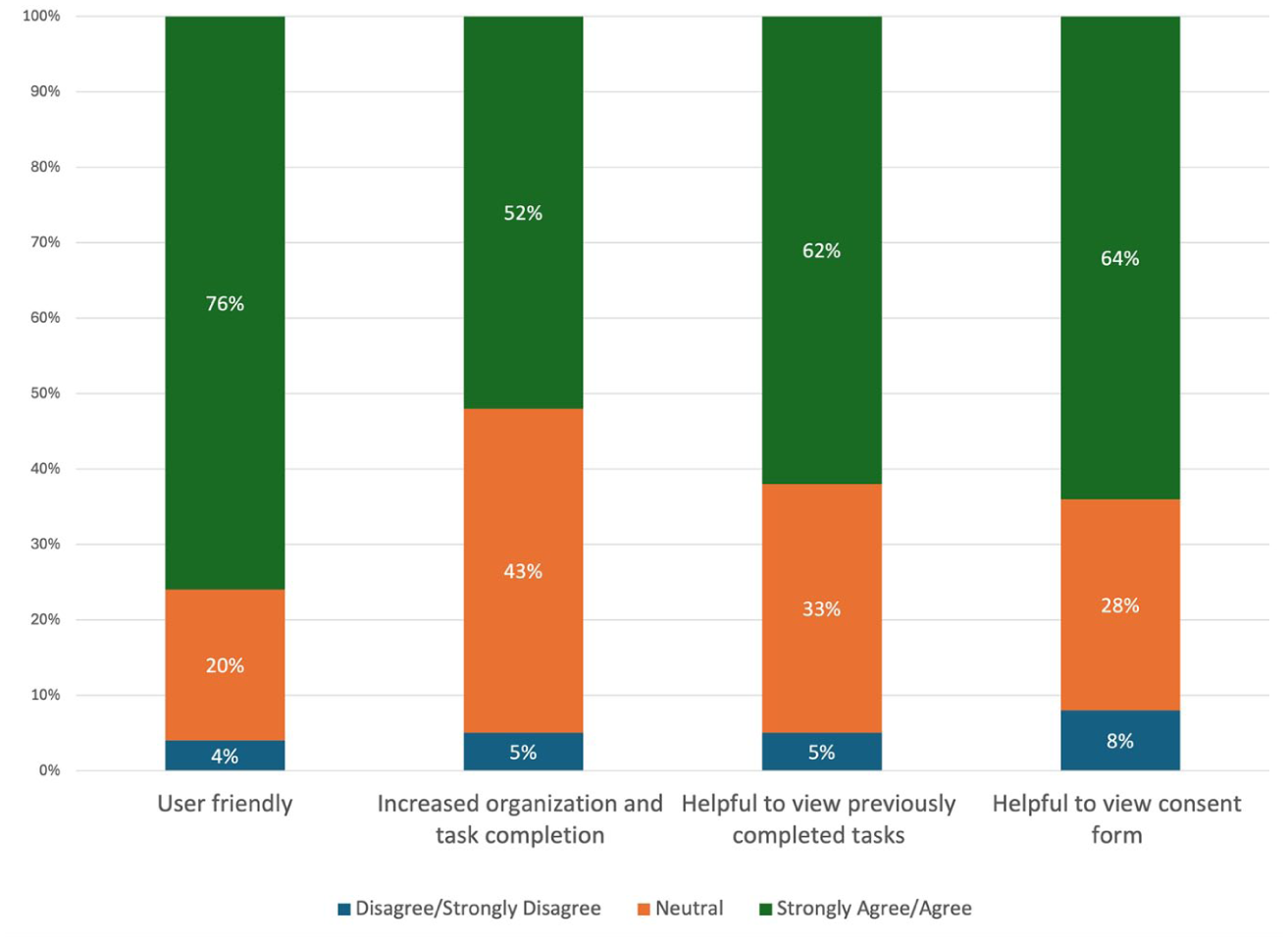
Participant user experience feedback (n = 116).
Discussion
The participant portal described here represents an important milestone in the digital infrastructure of our UCMDR research program, offering participants a streamlined and secure interface to engage with study activities. We successfully developed and implemented the portal, achieving a 40% account activation rate among all participants who received invitations, and a 41% activation rate among newly enrolled participants in the past year. This demonstrates feasibility and interest in portal use within our population.
Notably, baseline survey completion among portal users who were newly enrolled increased to 88%, compared with a historical rate near 60% prior to portal implementation. This improvement suggests that the portal not only facilitates communication and data access but may also enhance early participant engagement and data completeness. Such increases in timely survey completion can have significant downstream benefits for study efficiency and data quality.
Despite this success, actual utilization of the portal’s features was more limited than anticipated. Fewer than one-third of all activated users accessed surveys, and even fewer downloaded prior submissions or uploaded new documents. These usage patterns suggest that while participants were willing to activate their accounts, the current feature set may not have offered sufficient incentive or utility to drive consistent engagement. It is also possible that this lower functional engagement partly reflects the large number of historical participants among portal users, who may have been accustomed to traditional pre-portal study processes and perceived less need to adopt new digital tools. Future outreach and training efforts targeting this group may help increase adoption and highlight the added value of portal participation.
Statistical analysis of demographic and socioeconomic variables associated with portal uptake revealed significant differences by biological sex, with female participants more likely to opt in and activate accounts. This finding aligns with prior literature showing higher digital health engagement among women. 10 By contrast, age and family income were not significantly associated with portal engagement. These results suggest that while portal adoption may be broadly feasible across participant demographics, engagement barriers could differ by subgroup and are not solely technical in nature.
Efforts to enhance portal uptake should therefore include tailored outreach and usability improvements that address these differences, including streamlined account activation procedures, targeted engagement of long-standing participants who may be less accustomed to new digital interfaces, and strategies to promote equitable access and participation across diverse demographic and socioeconomic groups. Looking forward, there are clear opportunities to enhance the portal’s functionality to drive greater engagement. Potential improvements could include real-time study progression tracking, personalized gene-specific surveys, expanded educational content, mobile optimization, and centralizing survey prompts within the portal. Expanding the scope of portal functionality could increase both the frequency and depth of participant interaction. Next steps include engaging with UCMDR participants (both users and nonusers of the portal) to understand which potential feature enhancements they value most.
Ultimately, these findings highlight both the feasibility of implementing a participant-facing research portal and the importance of considering demographic and behavioral factors that influence engagement. The presence of statistically significant differences by sex underscores the need to monitor equity in digital engagement as the portal evolves. A long-term goal for the UCMDR participant portal is to implement continuous platform improvements that both facilitate and enhance research study participation and further advance the understanding and optimal management of monogenic forms of diabetes.
Footnotes
Acknowledgements
Abbreviations
UCMDR, University of Chicago Monogenic Diabetes Registry; CRI, Center for Research Informatics.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the National Institutes of Health National Institute of Diabetes and Digestive and Kidney Diseases (R01DK104942) and gifts from the Kovler Family Foundation. REDCap development was originally funded by the Biological Sciences Division and by the Institute for Translational Medicine, CTSA grant number UL1 TR000430 from the National Institutes of Health. The Center for Research Informatics is funded by the Biological Sciences Division at the University of Chicago with additional funding provided by the Institute for Translational Medicine, CTSA grant number 2U54TR002389-06 from the National Institutes of Health.
