Abstract

Electronic patient-reported outcomes (ePROs) are effective tools for collecting health information and managing chronic conditions such as type 2 diabetes mellitus (T2DM). 1 Hypoglycemic agents including sulfonylureas and insulin carry a significant risk for hypoglycemia.2,3 Hypoglycemia (blood glucose [BG] ≤70 mg/dl) increases the risk of cardiovascular and cerebrovascular events, and negatively impacts quality of life .4,5
We conducted a pilot in T2DM patients new to sulfonylureas and/or insulin to compare the reporting of hypoglycemia events between in-clinic paper surveys and the secure web-based EmpiraMed™ PRO Portal™ (Portal) Platform (EmpiraMed, Inc, Maynard, MA). This was a six-month, US prospective observational study, approved by the Institutional Review Board at Lahey Hospital and Medical Center (LHMC) in Burlington, Massachusetts.
The objectives were to
Compare patient-reported frequency of hypoglycemia events over six months using the Portal and in-clinic paper-based surveys
Evaluate the utility of the Portal to capture hypoglycemia events occurring between office visits
Participants completed an in-clinic paper survey at both baseline and six months, as well as an online survey at baseline. They made Portal entries during the study regarding symptoms, medications and BG readings. Portal hypoglycemia alerts were triggered by ≥2 BG readings ≤70 mg/dL in a week.
Number and proportion of participants with hypoglycemia events were obtained and assessed for agreement using kappa statistics (acceptable range kappa or ICC of at least 0.7). 6
A total of 100 participants consented, 90 registered on the Portal, and 82 completed the survey at 6 months. The means (SD) for age and HbA1c were 61 (11.1) years and 7.5% (1.2), respectively. The reporting of hypoglycemia symptoms by 89 participants approached agreement 37.1% (paper) and 39.3% (Portal) (κ = .667).
At follow-up, 37.8% (paper survey; N = 82) and 37.5% (Portal; N = 40) of participants reported hypoglycemia events. Among participants with low BG values, 87% reported symptoms on both Portal and paper surveys. Among those reporting no BG values, a larger percentage reported symptoms on the Portal (52%) compared to the paper survey (26%), indicating possible pseudo-hypoglycemia. There were 19 hypoglycemia alerts received for 12 participants. A lower rate of missing responses were noted on the Portal (0%) compared to paper surveys (7-14%). Portal-generated reports combined Portal entries along with end-of-study EMR data (Figure 1). Participant feedback on the Portal at the end of the study highlighted its user-friendliness.
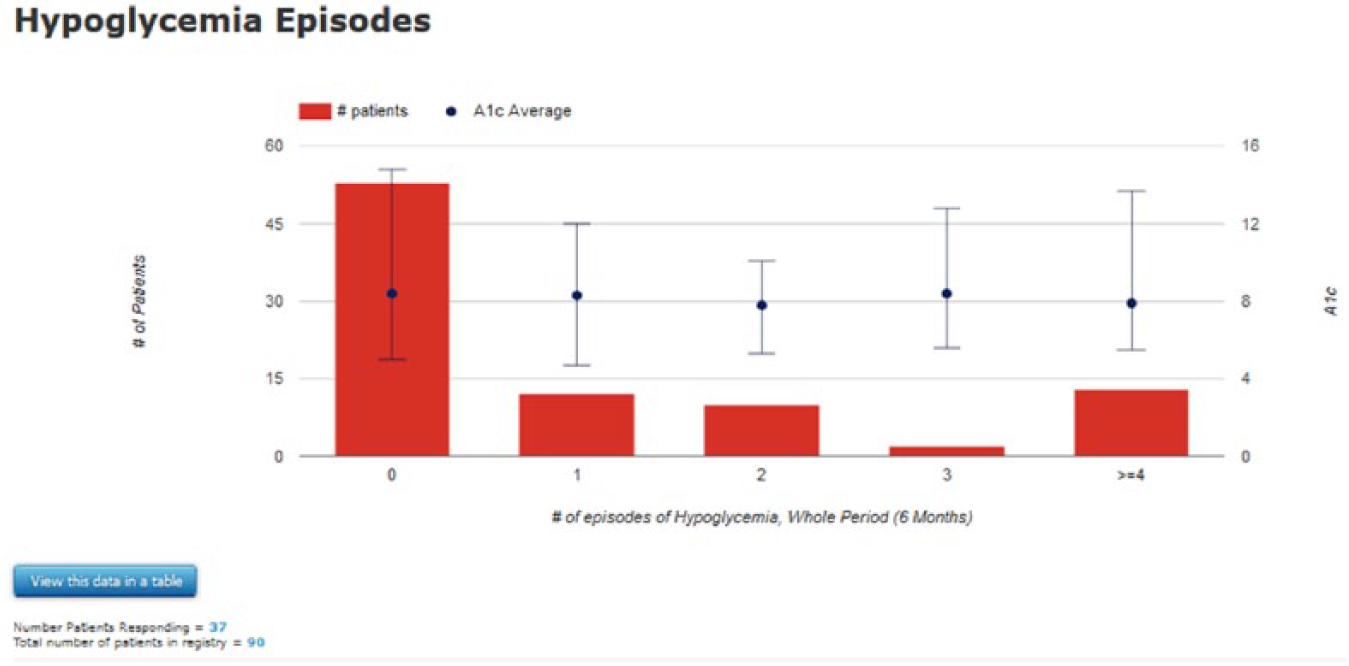
Portal sample report (screen capture): HbA1c values by number of hypoglycemia episodes.
The Portal was able to capture hypoglycemic alerts, potential pseudo-hypoglycemia, symptoms, medication changes, and impact on daily activities. Such data could help overcome drawbacks of relying on glucometers and patient recall. An additional utility of the Portal was its ability to integrate end-of-study EMR data.
Some limitations of the study include that (1) to date, the Portal is only available in English, and (2) this study required participants to have access to a personal computer.
In conclusion, future studies should investigate additional benefits of incorporating use of portals into routine outpatient settings and the ability to integrate EMR data in real-time, along with provisions for wider access through apps and additional languages.
Footnotes
Acknowledgements
We want to thank Dr Guy T. Napolitana, Dr Pallavi Heda, Dr Sarju Ganatra, Dr Jennifer C. Braimon, Jessica Speiser, Mary Anne Rutkowski, and Dr Kimberly Brodovicz for their support on this project.
Abbreviations
BG, blood glucose; EMR, electronic medical record; ePRO, electronic patient-reported outcome; LHMC, Lahey Hospital and Medical Center; PRO, patient-reported outcome; T2DM, type 2 diabetes mellitus.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Norquist, Rutkowski, Speiser, and Brodovicz are/were employees of Merck Sharp & Dohme Corp, a subsidiary of Merck & Co, Inc, Kenilworth, NJ, USA
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this research was provided by Merck & Co, Inc, Kenilworth, NJ, USA. Norquist is an employee of Merck Sharp & Dohme Corp, a subsidiary of Merck & Co, Inc, Kenilworth, NJ, USA
