Abstract
The goal of human-centered insulin pen design is to relieve the treatment burden of a chronic condition and help affected individuals to feel free of disease. The patient as well as their entire ecosystem should be considered. At Novo Nordisk A/S, we believe that embedding human-centered design at the heart of our development processes is best achieved with multidisciplinary experts in-house to work alongside product development teams and, importantly, the end user. Novo Nordisk introduced the first commercially available insulin pen in 1985 and has continued to develop reusable/durable and prefilled insulin pens to meet different patient needs, through to the latest NovoPen 6 and NovoPen Echo Plus with SMART technology. Human-centered design is essential for delivering meaningful and practical solutions for individuals with diabetes.
Human-Centered Design to Serve People Living With Diabetes: Philosophy and Practice
Human-centered design has made significant contributions to developing injection devices for insulin. The goal of this approach is to alleviate the treatment burden of a chronic condition and help affected individuals to feel as free as possible of disease, rather than consider themselves or have others perceive them as “patients.” The aspiration for delivery of injectable glucose-lowering therapy (insulin and/or glucagon-like peptide-1 receptor agonists) is that it should be as effortless and painless as possible, and not take away from everyday life.
With this in mind, human-centered design should adopt a holistic approach in which the patient, medication, existing drug delivery device, and entire ecosystem of the patient are considered (including family members or carers who may be managing medication for children, the elderly or those with other impediments to self-management). Insulin pens are designed for “users,” but individuals have so many other facets to their life. Consequently, the patient and their everyday environment and needs must be at the center of the design of insulin administration devices, not the technology or the disease. Usability is another important aspect of health care devices, but one that is easily misconstrued. While the usability of a product can be enhanced, health care companies must think beyond usability requirements to ensure that they are designing the “right” product to address the needs of patients. It is also critical to stress that technology alone should not lead development when designing products for people with chronic illnesses. However, it can act as a tool to help realize meaningful design solutions.
Successful products balance human desirability, technical feasibility, and business viability. Human-centered design processes identify solutions that are desirable from a human point of view, first and foremost (Figure 1). In order to understand the variety of users, field anthropology studies can provide helpful insights into different patient groups in their own environments. It is important for engineers and device designers to understand that patient priorities and barriers to therapy can vary greatly according to socioeconomic and educational status. 1 In a descriptive study of patients with type 1 diabetes (T1D), those in lower socioeconomic groups faced difficulties accessing care if they were in low-paid work, and displayed poor health literacy. 1 Some of these patients reported low motivation for diabetes management and did not always follow clinical recommendations. 1 For some, putting a “simple” written instruction for use into practice might represent a huge hurdle. To help ensure that new devices will successfully address issues important to patients and carers, patient/carer input on product concepts and designs should be sought at an early stage of the device development. End users of all educational levels should be represented, and their involvement should continue throughout the development process, including the testing of early prototypes and new products with providers. Once products have reached the market, post-marketing surveillance and other types of user data, such as patients’ perceptions and preferences, 2 offer insights from their real-world use that can be used by developers to improve the next product versions or generations.
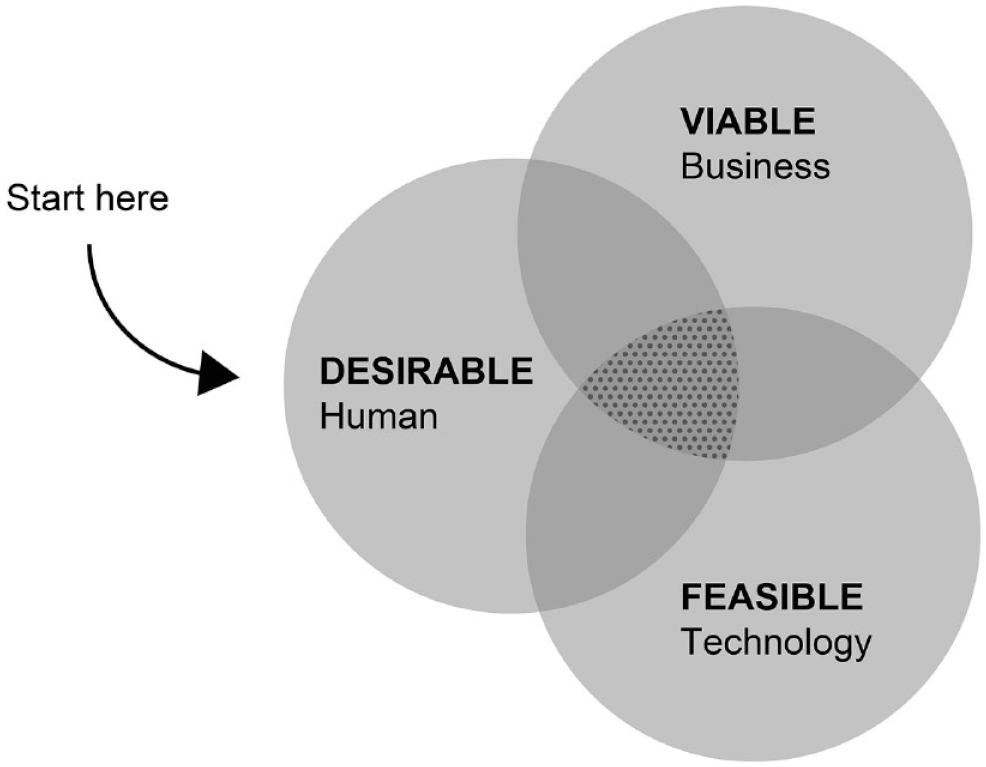
Human-centered design and where to start.
Watterson and colleagues describe the collaboration between the design company 3 IDEO and Eli Lilly in the human-centered design of insulin pens. At Novo Nordisk, we believe that embedding human-centered design at the heart of our development processes is best achieved with multidisciplinary experts in-house to work alongside product development teams. We have a dedicated design and user experience team that includes anthropologists, user experience researchers, industrial and digital designers, usability engineers, design-thinking specialists, medical doctors, and others. Design-thinking specialists work to apply human-centered design approaches and methods to teams and projects around the organization. Novo Nordisk also hosts events at which employees are given the opportunity to gain insights from meeting and/or hearing from patients from around the globe. We gain further insights by constantly listening to spontaneous feedback from our customers, as well as evaluating market feedback on an ongoing basis. The aforementioned investments and activities ensure that human-centered processes are an integral part of the way we work. One advantage of this strategy is to facilitate relationships among providers, patients, and designers, something that has previously been identified by designers as a challenge. 4 Overall, a human-centered design-thinking approach is essential for modern companies in the health care sector to improve their products for the benefit of patients. Part of this approach involves taking social responsibility into account, by making affordable and environmentally friendly products, for example.
Insulin Pens Based on Human-Centered Design
Novo Nordisk introduced the first commercially available insulin pen in 1985 and has continued to develop families of reusable/durable and prefilled insulin pens to meet different patient needs, through to the latest NovoPen 6 and NovoPen Echo Plus with SMART technology (Table 1).11,23,24 Interestingly, the fundamental pen design has remained broadly similar since the 1980s, demonstrating that certain designs have longevity and can be improved incrementally over time. The design process for each of these products has always started with people’s needs. Indeed, the inspiration for the first insulin pen came from a mother and doctor, Dr Reith, who experienced challenges with using an insulin vial, steel needles, and a glass syringe for her young daughter with T1D, particularly while traveling.
25
Research by Dr Reith and colleagues on the original pen device, which was designed to simplify insulin injections, was published in
Evolution of Novo Nordisk Insulin Pens.
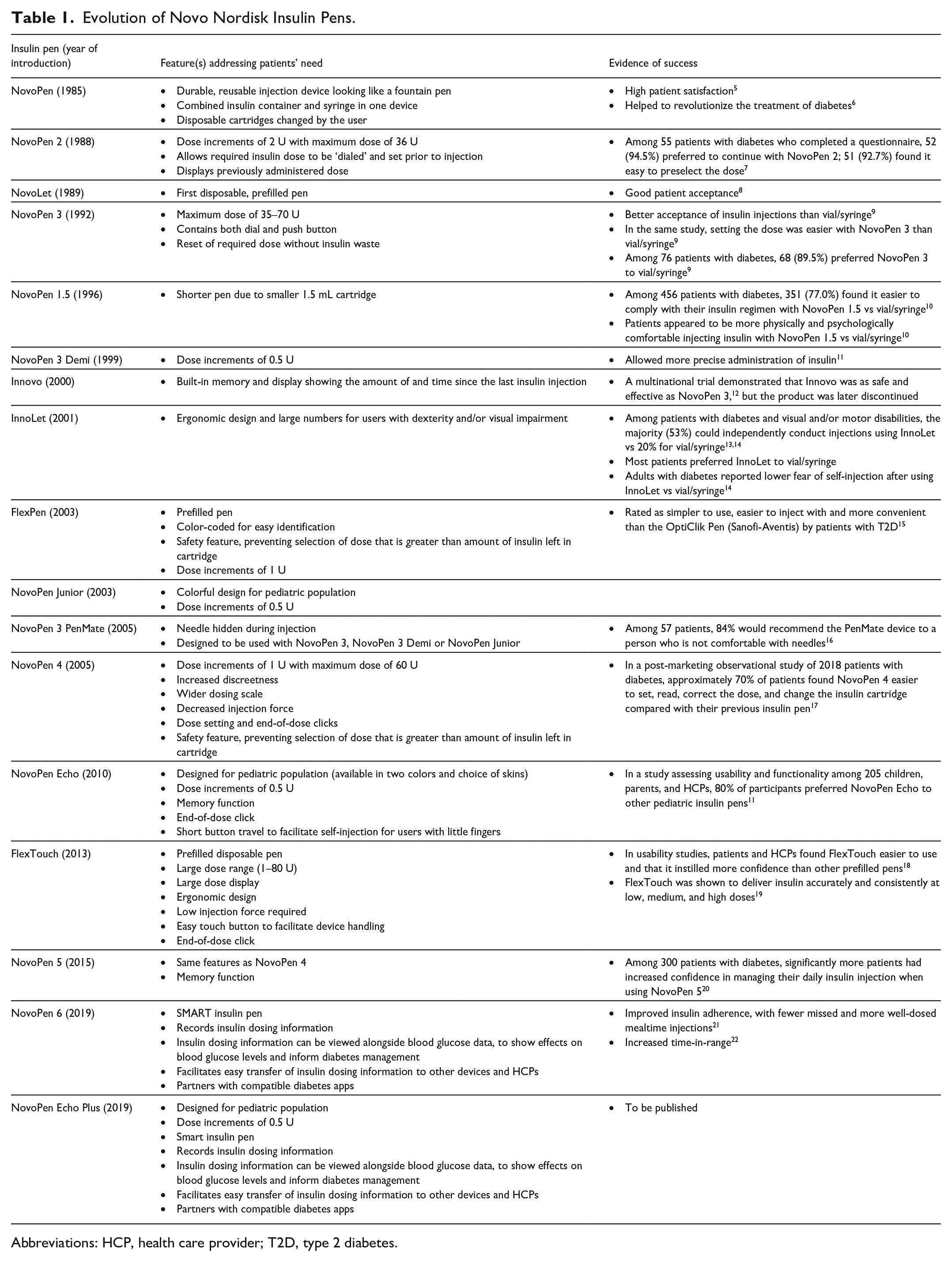
Abbreviations: HCP, health care provider; T2D, type 2 diabetes.
With respect to insulin delivery, there may be different ways to solve similar needs in distinct populations, and conversely the same “solution” may address more than one need within or across patient groups. For example, children’s requirements include accurate dosage of half-units of insulin, injection devices that are easy to use with small hands, safety features to avoid mistakes in doses, and education to motivate self-management. These requirements may not always be recognized or articulated by the children themselves, but the NovoPen Echo was designed to address them.11,27 Child-friendly features of the NovoPen Echo have been reviewed previously and include its half-unit increment dosing, easy-to-use dial, and memory function. 11 Elderly patients may also have limited manual dexterity and less strength than younger adults, not because of small hands but perhaps owing to arthritis or peripheral neuropathy. This may be combined with diabetic retinopathy, and cognitive and memory impairment. These were considerations in the design of InnoLet, FlexTouch, NovoPen 4, and subsequent NovoPen devices, which include features such as short button travel, reduced injection force, and/or larger dosage scale displays than other devices.11,13,23,28 -30 In addition, simplicity is critical for some patients and a driver for prefilled insulin pens such as the FlexTouch. 31 Minimal training and maintenance are needed to use prefilled, disposable pens, which do not require the installation of new cartridges. 32 Other patients may prioritize the environmental credentials of being able to use refillable, reusable cartridge pens. The industry makes a toolbox of options available for health care professionals and patients to find the best-suited injection device solution for individual users.
Innovo, which launched over two decades ago, contained a built-in memory and display to show the time since the last insulin injection and dose size, 33 to help to answer the common question “did I take my last insulin dose?” Later, the HumaPen Memoir digital insulin pen (Eli Lilly and Company) was introduced, which had a multiple-dose memory feature allowing patients to record and review their last 16 insulin doses. 34 Recording and analysis of glycemic parameters and medication doses are key elements of diabetes management, to determine whether glycemic targets have been met, and to inform personal, evidence-based decisions on dosing. To this end, all newer-generation SMART insulin pens, including NovoPen 6 and NovoPen Echo Plus, have real-time wireless connectivity to record injection logs on SMART phones, which among others can be shared with the patient’s clinician.24,35,36 One drawback of SMART technology is that it can focus overly on the ability to provide extensive data that enable the patient to “do more” in terms of monitoring, when actually they want to “do less.” Indeed, technology should not overshadow the goal of making lives easier for people living with diabetes, which may not necessarily be reached by simply providing them with more data. One potential solution involves integration of data from SMART pens and glucose monitors in the same app, with customized settings that automatically calculate the right dose for the patient. 36 Partner systems that are compatible with glucose-lowering products from different companies and provide one unifying solution (e.g. by integrating the data into a blood glucose meter app or a continuous glucose monitoring app) could also make the devices and apps more user friendly. However, this technology will only be helpful and adopted if people perceive its value as higher than the burden it involves. The best solution will be one that meets the user where they are, rather than one forced upon them. Given that reusable SMART insulin pens are relatively new technologies, 35 enabled by the broader availability of SMART phones in recent years, 37 they will probably improve and become easier to use over time, with the advent and availability of further technological innovations. Newer-generation SMART insulin pens may also be refined and become more widely used as early adopters offer their feedback and demonstrate the value of these devices.
What New Challenges for Insulin Delivery Can Human-Centered Design Address?
Despite the remarkable longevity of pen injection devices, future delivery devices may take a different form depending on the challenge they aim to address. The specific needs and circumstances of people requiring insulin continues to change over time, and administration devices need to evolve accordingly. The increasing incidence of T1D and type 2 diabetes (T2D) in children and adolescents is particularly alarming, and is especially high in some ethnicities and higher in female than in male adolescents.38 -40 Early-onset T2D has a more aggressive disease course and a greater risk of complications than the profile seen with more classic T2D. 39 New forms of insulin, eg, once-weekly basal insulin and the more distant possibilities of oral insulin and glucose-sensitive “smart” insulin, 41 could modify the treatment landscape and the experience being designed for. In addition, knowledge around factors affecting pain associated with injection, such as injection formulation and volume, continues to accumulate and could influence the development of new drug formulations and injection devices.42 -44 Future design solutions may be needleless or time-release implants. Sensor-augmented insulin pumps and hybrid closed-loop systems are already available for use in T1D. 45 These address some unmet needs, but do they create new and different challenges for patients, particularly with respect to technological and mechanical complexities, and access? Stem cell therapies that would deliver insulin-producing islet cells for cell transplantation could represent a new treatment avenue for patients with diabetes. 46 Whatever the future holds in terms of new delivery possibilities, the impact of changes should not be assumed. The perspectives of patients must always be sought and critically analyzed by developers and designers, ideally throughout the process. Given that one model does not fit all patients, many different treatment delivery options may be needed. Ultimately, a cure for diabetes needs to be found.
In conclusion, human-centered design is essential for delivering meaningful solutions for individuals living with diabetes who need or will need insulin therapy. The challenges are complex, but the best solutions will provide positive clinical outcomes while seamlessly fitting into a user’s life.
Footnotes
Acknowledgements
Medical writing support was provided by Nicolas Bertheleme, PhD, an employee of Oxford PharmaGenesis.
Abbreviations
HCP, health care professional; T1D, type 1 diabetes; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.S., N.A.H., A.S.W., and M.G. are employees of and own shares in Novo Nordisk A/S.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for editorial assistance was provided by Novo Nordisk A/S.
