Abstract
Objectives:
There is limited evidence regarding the cost-effectiveness of real-time continuous glucose monitoring (rt-CGM) in the management of type 2 diabetes in patients receiving basal insulin regimens in South Korea. This study aimed to address this gap by analyzing the cost-effectiveness of rt-CGM when compared with self-monitoring of blood glucose.
Research design and methods:
The cost-effectiveness of rt-CGM was modeled using the IQVIA Core Diabetes Model from a South Korean payer perspective in a simulated cohort based on the Korea National Health and Nutrition Examination Survey. A remaining lifetime horizon (up to 50 years), and a discount rate of 4.5% per annum were adopted. The willingness-to-pay threshold was KRW 47 million per quality-adjusted life year (QALY).
Results:
The use of rt-CGM resulted in an increase in quality-adjusted life expectancy of 0.490 QALYs and a KRW 13.3 million increase in costs compared with self-monitoring of blood glucose. The incremental cost-utility ratio in the base case analysis was KRW 27.1 million per QALY, below the willingness-to-pay threshold of KRW 47 million.
Conclusions:
rt-CGM is likely to be cost-effective versus self-monitoring of blood glucose in the management of patients with type 2 diabetes receiving basal insulin regimens in South Korea.
Introduction
Type 2 diabetes mellitus (T2D) is associated with a substantial burden in South Korea, with 15.5% of adults >30 years of age reported as having T2D in 2021–2022. 1 Furthermore, the prevalence is reported to have increased over the last two decades in a nationwide cross-sectional study 2 and an analysis of claims data from young adults and pediatric patients.3,4 Such high prevalences are notable due to the substantial economic and societal impacts of T2D, 5 with an economic study estimating the total cost of diabetes in South Korea in 2017 to be KRW 3.2 trillion. 6
Maintaining glycemic control is critically important for the prevention of complications of diabetes.7 -10 In South Korea, a glycated hemoglobin (HbA1c) target of <6.5% is considered optimal for T2D and insulin therapy is recommended for patients with T2D with severe hyperglycemia and hyperglycemic symptoms. 11 However, a cross-sectional study found that only 32.4% of adults with diabetes achieve glycemic control (HbA1c <6.5%). 1
The management of diabetes can be facilitated with real-time continuous glucose monitoring (rt-CGM) devices, which can provide information on trends in blood glucose and alerts for both hyperglycemia and hypoglycemia. 12 Such devices have been associated with reductions in HbA1c and improvements in glycemic control.13 -19 The use of rt-CGM is currently recommended by the Korean Diabetes Association to achieve better glycemic control and reduce the risk of hypoglycemia in adults with type 1 diabetes or T2DM treated with insulin injections. 11 Such systems have recently been shown to be cost-effective for the management of patients with T2D receiving intensive insulin regimens in South Korea 20 and have previously been shown to be cost-effective for the management of T2D in other countries. 21 However, evidence regarding the cost-effectiveness of rt-CGM in the management of patients with T2D receiving treatment with basal insulin regimens in South Korea is currently limited.
The objective of this analysis was to evaluate the cost-effectiveness of rt-CGM when compared with self-monitoring of blood glucose (SMBG) in patients with T2D receiving basal insulin therapy in South Korea.
Methods
Model Structure
The cost-effectiveness of rt-CGM compared with SMBG for people with T2D on basal insulin regimens was assessed using the IQVIA Core Diabetes Model (CDM, v9.5).22 -24 The model structure and its use have previously been described elsewhere.22 -24
The CDM is composed of 17 interdependent Markov sub-models that interact to predict the long-term health outcomes and costs of diabetes interventions. It predicts the progression of risk factors using equations derived from studies including the Framingham Heart Study and the United Kingdom Prospective Diabetes Study (UKPDS). Outputs of the CDM include life expectancy, quality-adjusted life years (QALYs), direct costs, indirect costs, cumulative incidence and time to onset of complications, and incremental cost-utility ratios (ICURs) per additional life years or QALYs gained.
Baseline Cohort Characteristics and Treatment Effects
The baseline characteristics for the study population were obtained from a previous cost-effectiveness analysis from a South Korean payer perspective using the CDM, with cohort characteristics estimated from the Korea National Health and Nutrition Examination Survey. 25 Further risk factors were added from a cohort study from the Kaiser Permanente health care delivery system diabetes registry 13 and the CDM default values based on the ACCORD study. 26 The cohort had a mean (standard deviation) age of 64.4 (11.4) years, duration of diabetes of 18 (9.5) years, baseline HbA1c of 8.6% (1.3%), and all patients were Asian (Supplemental Table S1). 25
Clinical Effectiveness
In the base case analysis, the efficacy of rt-CGM and SMBG was based on results of the Steno2Tech study and the MOBILE study. Steno2Tech was a 12-month single center trial comparing rt-CGM and SMBG including inadequately controlled adult patients with T2D with HbA1c ≥7.5% treated with daily insulin. 27 Patients who used rt-CGM experienced a 0.9% reduction in HbA1c and a 1.1 kg/m2 reduction in body mass index at 12-month follow-up. 27 The MOBILE study compared rt-CGM and SMBG in adult patients with T2D treated with insulin at 15 centers in the United States. 28 The cohort had HbA1c values between 7.8% and 11.5% and had been on stable regimens of other antidiabetic medications for greater than or equal to three months. 28 Patients in the rt-CGM arm experienced a 0.4% reduction in HbA1c compared with the SMBG arm at eight months. 28
The reduction in HbA1c in this analysis was sustained for an additional two years in the rt-CGM arm after Year 1 based on evidence from longitudinal studies.29 -31 The increases in HbA1c after Year 1 in the SMBG arm and after Year 3 in the rt-CGM arm were both 0.15 units per year based on the CDM clinical table value.
Severe hypoglycemic and hyperglycemic event rates were sourced from the Kaiser Permanente retrospective cohort study. 13 As the Kaiser Permanente study inclusion criteria favored high-risk patients, a 50% lower severe hypoglycemic event (SHE) rate than the Kaiser Permanente study was assumed for the current analysis. For hypoglycemic events, the rates were 0 events per 100 patient years for rt-CGM and 2 events per 100 patient years for SMBG. For hyperglycemic events, the corresponding rates were 0 events and 1.25 events per 100 patient years, respectively.
Costs
The analysis included direct medical costs and was conducted from a South Korean payer perspective. Costs of concomitant medications, screening, and diabetes-related complications were taken from published sources and inflated to 2023 costs using the consumer price index (health) from the Korean Statistical Information Service. Medication and screening costs were obtained from the cost-effectiveness analysis reported by Park et al. 25 Complication costs (Supplemental Table S2) were also obtained from Park et al. 25 Data on the utilization of aspirin, statins, and angiotensin-converting enzyme (ACE) inhibitors for the prevention of cardiovascular and microvascular disease were obtained from Jung and Lee, 32 and the Kaiser Permanente retrospective study. 13 The screening rates for eye and renal disease reported in the study reported by Byun et al 33 were used.
Annual treatment costs for rt-CGM were based on data from the manufacturer, accounted for the use of 36 sensors, and assumed a 50% reimbursement rate from the payer (Supplemental Table S3). The SMBG price assumed two tests per day, as observed in the MOBILE trial, 28 and was based on the current price in South Korea.
Utilities
All utilities used in the analysis are listed in Supplemental Table S4. A utility value of 0.785 was used for T2D without complications based on the meta-analysis reported by Beaudet et al. 34 Further utilities and disutilities for diabetes-related complications and treatment-related adverse events were obtained from Beaudet et al 34 and Zhao et al. 35
A utility benefit of 0.03 was applied to patients in the rt-CGM group due to the avoidance of daily and frequent fingerstick testing (AFS) based on a study reported by Matza et al. 36 An additional benefit due to reductions in fear of hypoglycemia (FoH) of 0.008 was applied to the rt-CGM group based on results reported by Soriano and Polonsky. 37
Discounting, Time Horizon, and Willingness-To-Pay
A remaining lifetime horizon, with a maximum of 50 years, and a discount rate of 4.5% per year for future costs and clinical effects were adopted as specified in guidelines issued by Health Insurance Review and Assessment service. Due to the lack of an official willingness-to-pay (WTP) threshold in South Korea, a threshold of three times the per capita annual GDP (KRW 47 million in 2023) was used as recommended by the World Health Organization (WHO). 38 A WTP threshold of KRW 32 million was also taken into account, reflecting the median incremental cost-effectiveness ratio (ICER) of novel drugs for rare diseases in South Korea between 2007 and 2021. 20
Sensitivity Analyses
A total of 42 sensitivity analyses were performed. All probabilities were initially sampled using a first-order Monte Carlo simulation to provide point estimates for use in a second-order Monte Carlo simulation. The resampling method was then applied to each parameter from the first-order results with costs and outcomes accumulated for 1000 theoretical patients in both arms, with each going through the model 1000 times. This resulted in a joint distribution of mean and incremental costs and effectiveness gains. Percentages of joint distributions falling within a cost-effective range were calculated and used to produce a scatter plot.
One-way sensitivity analyses examining the effects of varying the intervention-related utilities associated with FoH and avoidance of fingerstick testing were conducted. For FoH, the two additional analyses included removing the utility, or increasing it by 50%. For AFS, the utility was changed by ±50%. The intervention effect on HbA1c was varied across five values in a range of −0.9% to −0.35%, based on those reported in the Steno2Tech trial, 27 MOBILE study, 28 and subgroup analyses of the MOBILE study. The rates of severe hypoglycemic and hyperglycemic events were varied by ±50% to 100% or removed entirely. Additionally, the number of SMBG tests per day was reduced from 2 to 1.1 to match the frequency reported by the Steno2Tech study 27 and 1.5 to match the MOBILE study. 28 To determine the impact of the cardiovascular risk prediction equation used, several alternatives were tested including UKPDS68, UKPDS82, 39 China Prediction for Atherosclerotic Cardiovascular Disease Risk (PAR), 40 Japan, 41 and East Asia (meta-regression). 42
Other alterations of parameters investigated in the sensitivity analyses included reducing the 50-year time horizon to one, five, 10, or 30 years, reducing the mean age at initiation from 64.4 years to 35 or 45 years, and varying the duration of diabetes between one, five, and 10 years. Finally, six scenarios were used to test the effect of changing the reimbursement price of rt-CGM, varying it by ±10%, ±15%, ±20%.
In addition to the one-way analyses, two-way sensitivity analyses were used to examine the effect of varying both age and duration of diabetes. This included assessing a mean age of 45 years and duration of diabetes of one year, in addition to a mean age of 55 years and duration of 10 years.
Projected Clinical Outcomes
Absolute risk reductions (ARR) and relative risks (RR) were calculated for all diabetes-related calculations included in the modeling. For cardiovascular risks, ARR and RR calculations were performed with all risk equations used in the sensitivity analysis in addition to that used in the base case analysis.
Results
The use of rt-CGM resulted an increase in QALYs of 0.490 compared with SMBG in the base case analysis (7.344 vs 6.855) (Table 1). Although total costs were KRW 13.3 million higher with rt-CGM than with SMBG, the ICUR was KRW 27.1 million per QALY, below the WHO-recommended WTP threshold of KRW 47 million (Table 1). In a probabilistic base case analysis (Figure 1), rt-CGM had a 63.5% likelihood of being cost-effective at a WTP threshold of KRW 47 million and a 53% likelihood at a threshold of KRW 32 million (Figure 2).
Summary of Base Case Findings.
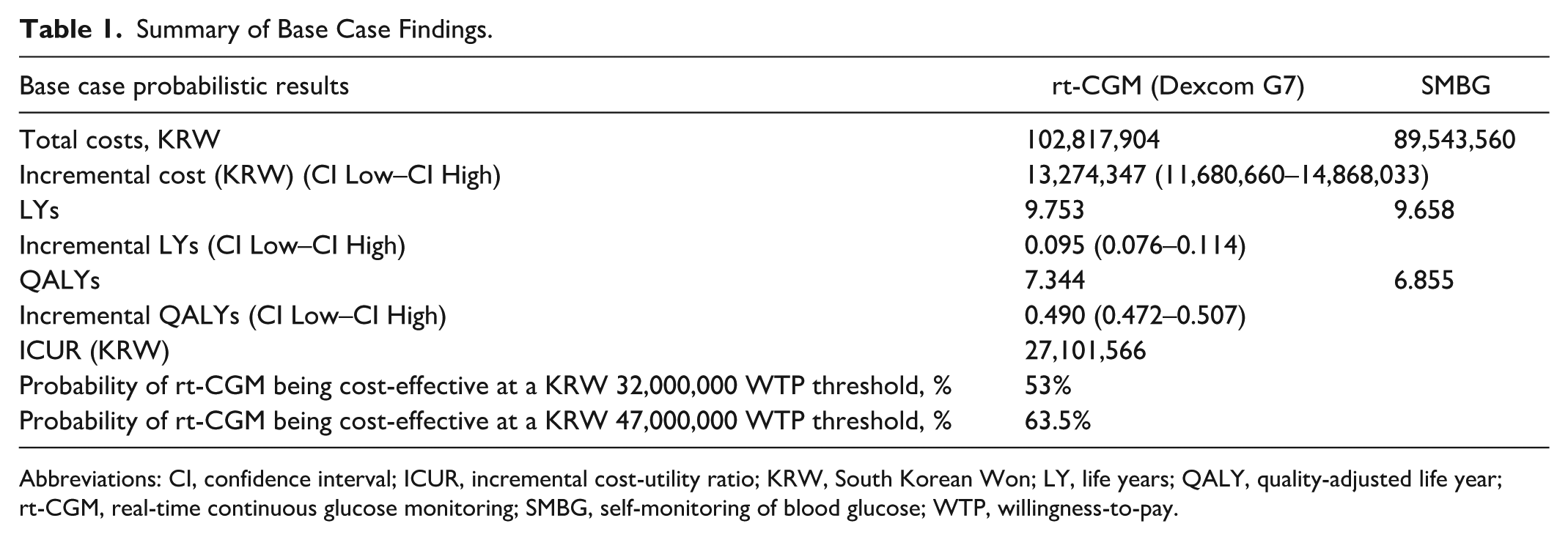
Abbreviations: CI, confidence interval; ICUR, incremental cost-utility ratio; KRW, South Korean Won; LY, life years; QALY, quality-adjusted life year; rt-CGM, real-time continuous glucose monitoring; SMBG, self-monitoring of blood glucose; WTP, willingness-to-pay.
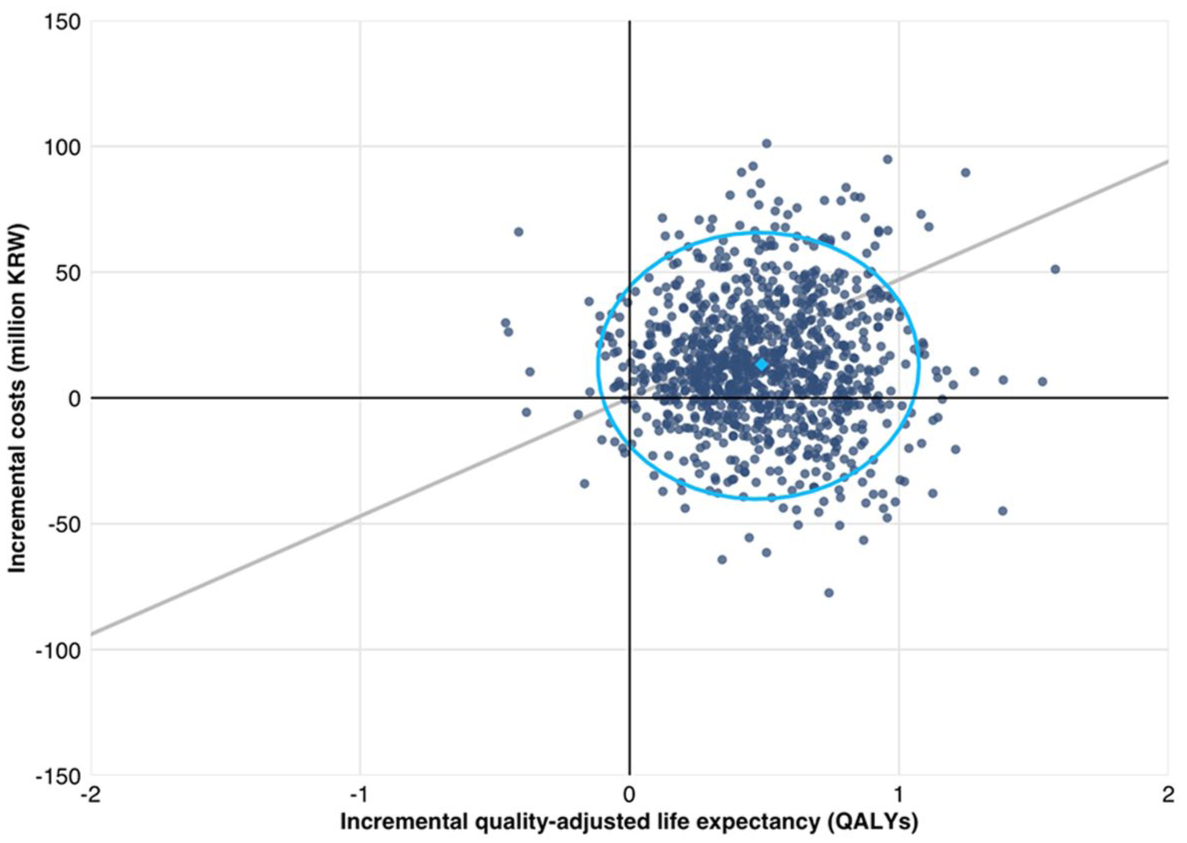
Cost-effectiveness scatterplot from the probabilistic base case analysis. Light blue diamond depicts mean outcome over all 1000 model iterations. Light blue ellipse is a 95% data ellipse based on a multivariate T-distribution. Gray line represents a willingness-to-pay threshold of KRW 47 million per QALY.
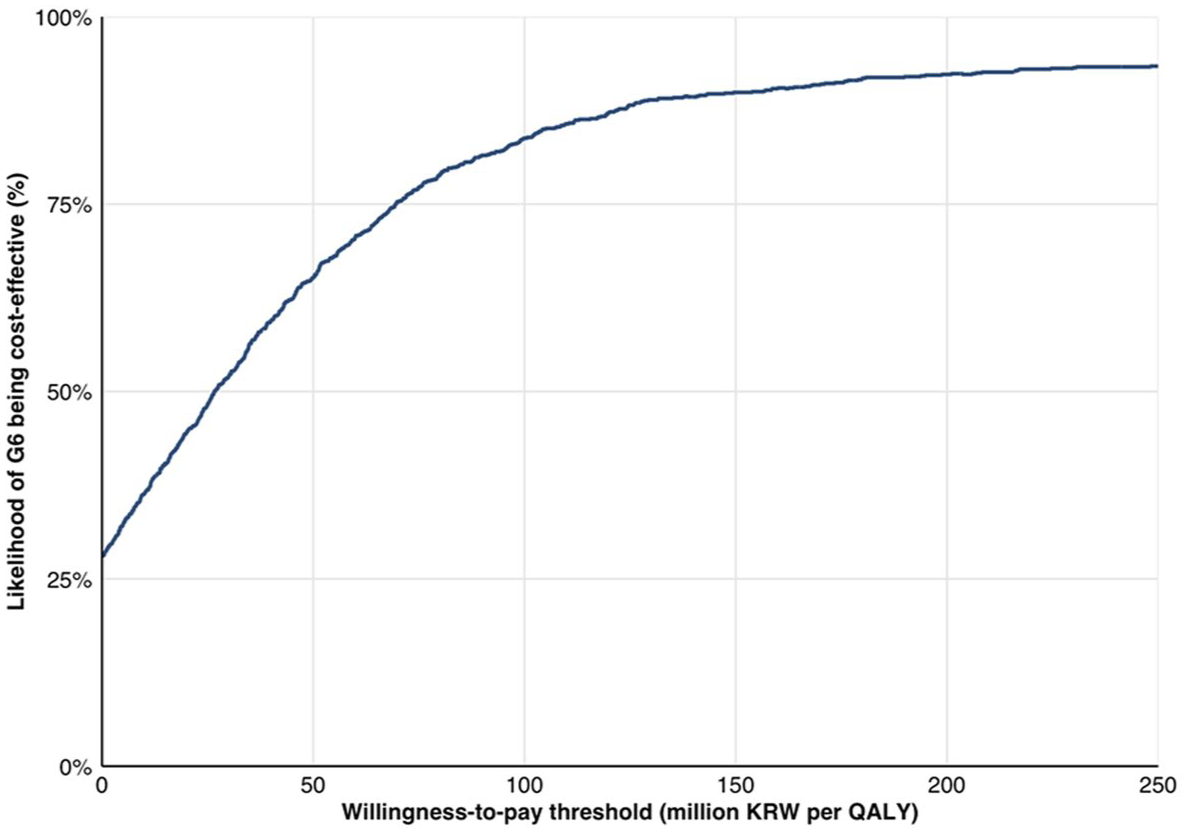
Cost-effectiveness acceptability curve from the probabilistic base case analysis.
Sensitivity Analyses
Sensitivity analyses were carried out to examine the effects of a variety of parameters on the ICUR, including treatment effect, treatment-specific utility, severe hypoglycemia/hyperglycemia rates, time horizon, duration of diabetes, age, cardiovascular risk prediction equation used, and the price of rt-CGM. The results of these analyses are presented in Table 2. Changes in the effectiveness of rt-CGM in reducing HbA1c resulted in small changes in ICUR, with the largest being a decrease to KRW 23.1 million per QALY with a 0.9% decrease in HbA1c and an increase to KRW 29.9 million per QALY with a 0.35% HbA1c reduction. The largest changes in ICUR were observed when changing treatment-specific utility values associated with avoidance of frequent AFS and FoH. Increasing the utility associated with AFS by 50% increased the difference in QALYs to 0.644, decreasing the ICUR to KRW 20.6 million per QALY while decreasing the utility by 50% reduced the difference in QALYs to 0.336 and increased the ICUR to KRW 39.6 million per QALY. For FoH, a 50% increase in the utility gain resulted in a decrease in the ICUR to KRW 23.3 million per QALY while reducing the utility change to zero increased the ICUR to KRW 32.6 million per QALY.
Results of Sensitivity Analyses Comparing rt-CGM and SMBG.
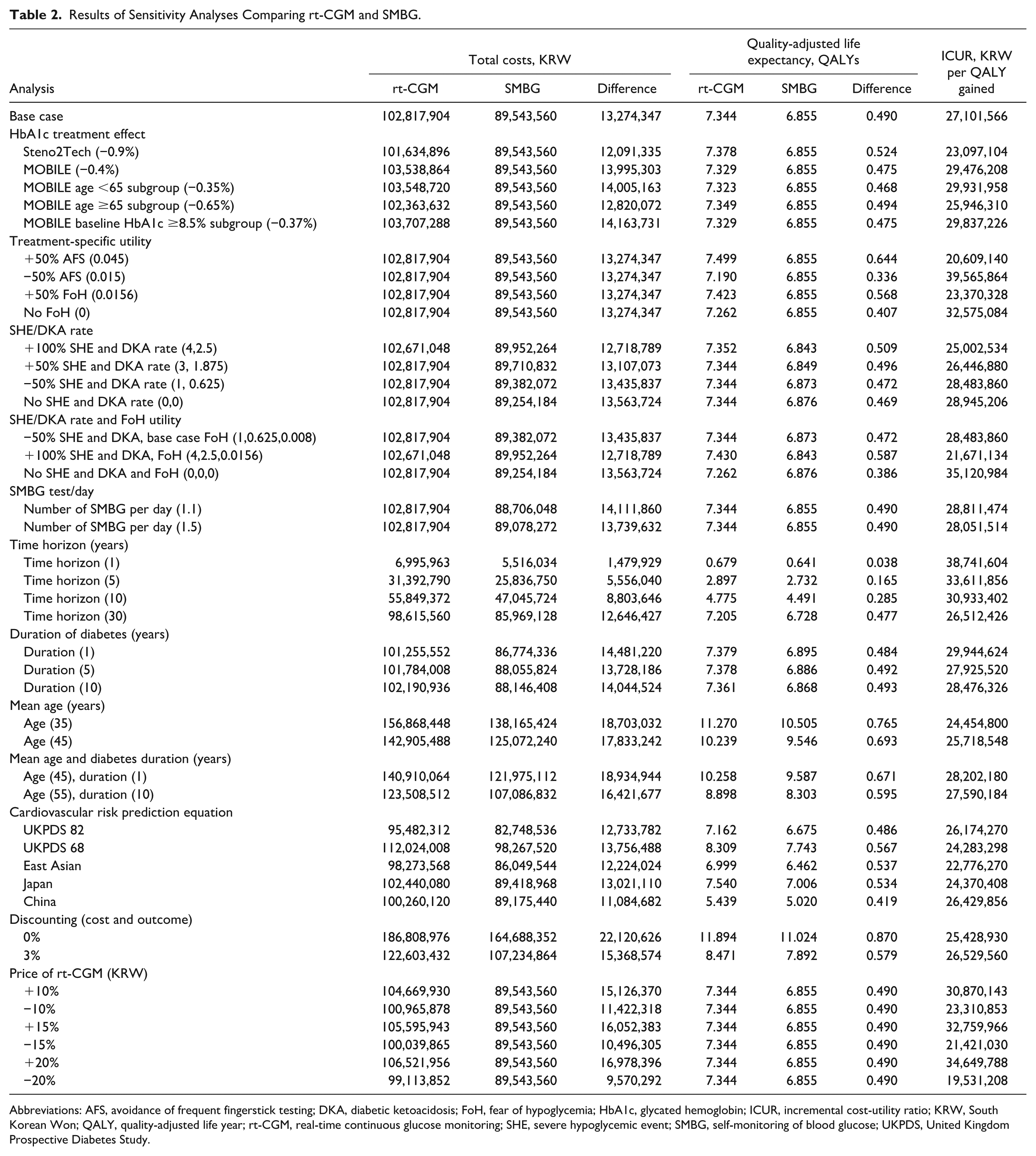
Abbreviations: AFS, avoidance of frequent fingerstick testing; DKA, diabetic ketoacidosis; FoH, fear of hypoglycemia; HbA1c, glycated hemoglobin; ICUR, incremental cost-utility ratio; KRW, South Korean Won; QALY, quality-adjusted life year; rt-CGM, real-time continuous glucose monitoring; SHE, severe hypoglycemic event; SMBG, self-monitoring of blood glucose; UKPDS, United Kingdom Prospective Diabetes Study.
When examining the influence of hypoglycemic and hyperglycemic events, the ICUR tended to decrease with increases in the rates of severe events, dropping to KRW 25.0 million per QALY when they were increased by 50% and increasing to KRW 28.9 million per QALY when they were removed. When the time horizon was varied between one, five, 10, and 30 years, the ICUR was found to decrease with an increasing horizon, ranging from KRW 38.7 million per QALY at one year to KRW 26.5 million per QALY at 30 years. For cardiovascular risks, five scenarios assessed alternative prediction equations, with all five resulting in ICURs lower than the base case. Other variables found to affect the ICUR included the mean age of the population, their average duration of diabetes, and the price of rt-CGM (Table 2).
Projected Clinical Outcomes
The projected RRs of diabetes-related complications in addition to ARRs and cumulative incidence for patients with T2D treated with non-intensive insulin regimens are displayed in Table 3. For all complications other than two, the RRs comparing rt-CGM and SMBG were numerically less than 1.0 suggesting lower rates of complications with rt-CGM, while for the remaining two (congestive heart failure fatalities and stroke fatalities) it was equivalent to 1.0. In particular, rt-CGM was significantly associated with a lower risk of ophthalmic complications, renal complications, peripheral vascular disease, and neuropathy. The largest reductions in risk were observed for end-stage renal disease (RR 0.68), gross proteinuria (0.76), proliferative diabetic retinopathy (0.76), and microalbuminuria (0.81). Although the risks of certain cardiovascular complications, such as stroke or myocardial infarction, were not significantly reduced with rt-CGM when using the equation by Park et al, 43 alternative risk equations demonstrated significant reductions in cardiovascular complications (Table 4).
Projected Diabetes Complications for Non-Intensive Insulin-Treated Adult Patients With T2D in South Korea.
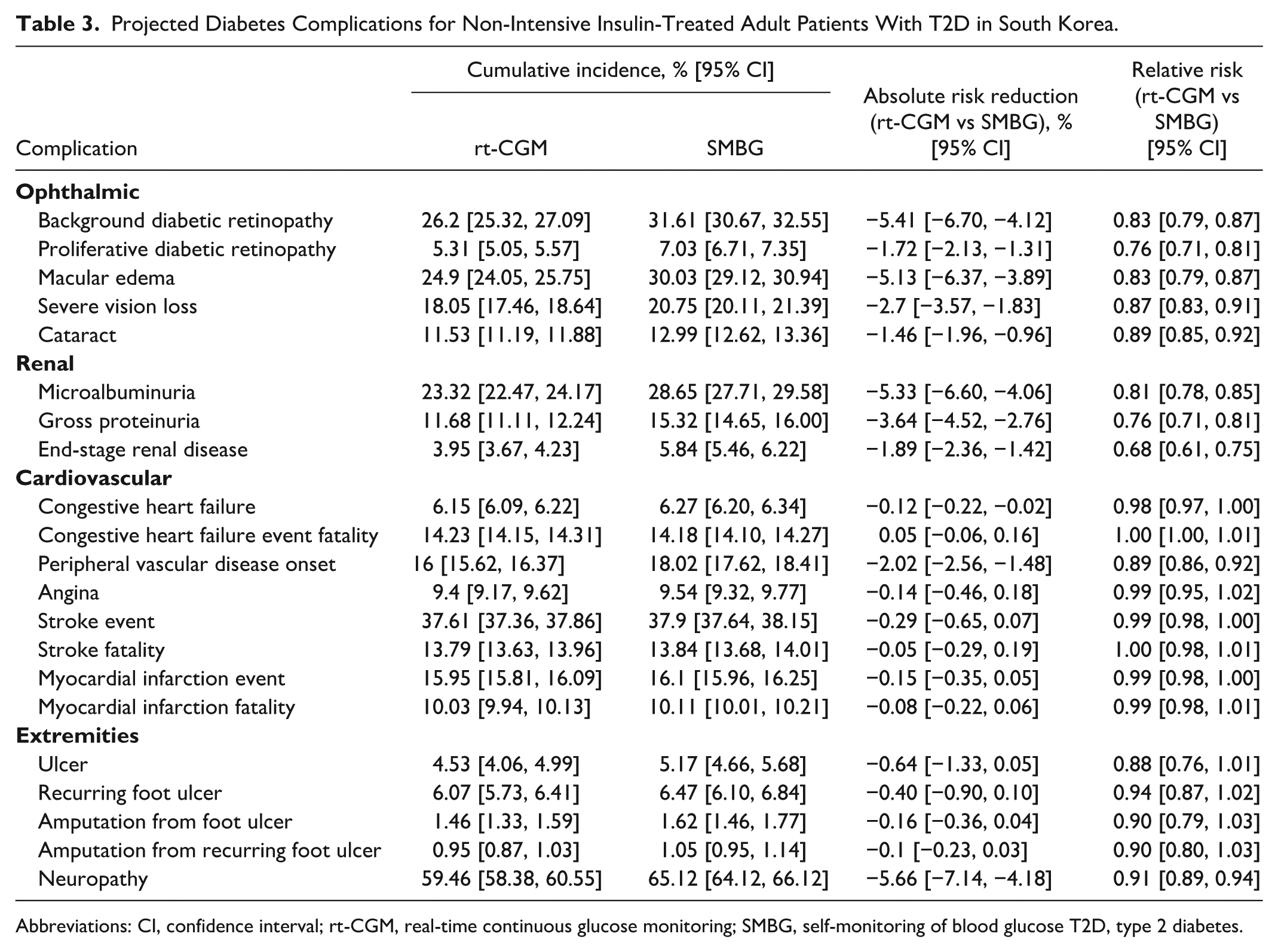
Abbreviations: CI, confidence interval; rt-CGM, real-time continuous glucose monitoring; SMBG, self-monitoring of blood glucose T2D, type 2 diabetes.
Comparison of Projected Cardiovascular Complications Between rt-CGM and SMBG Between Different Equations.
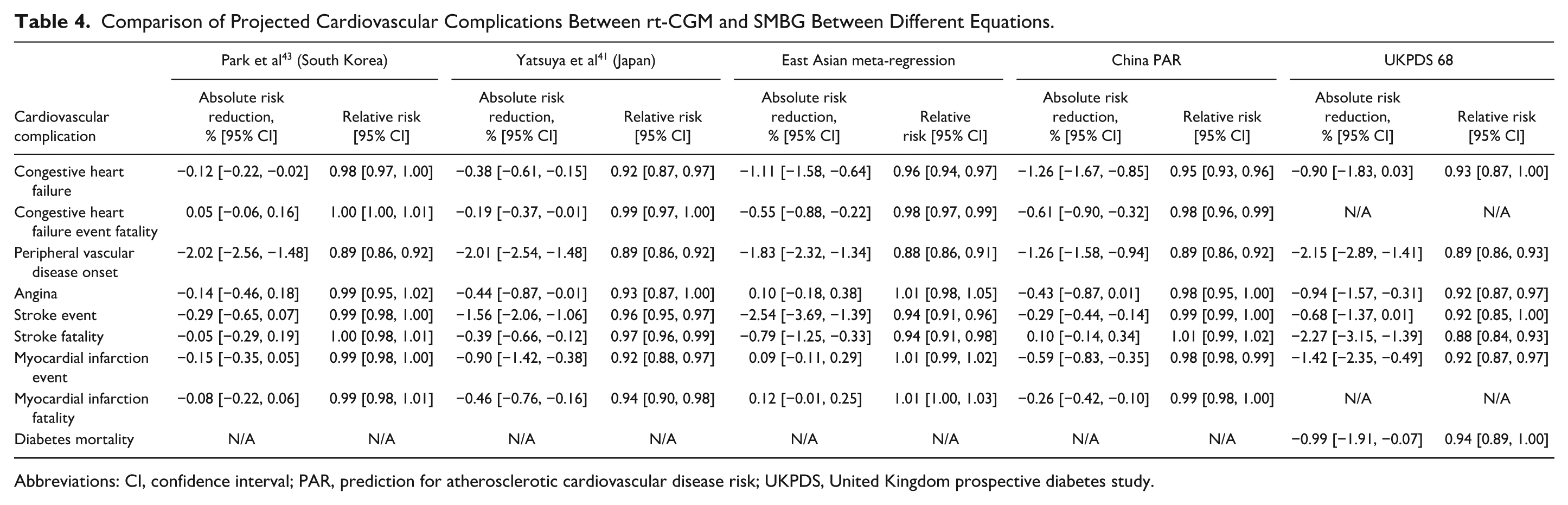
Abbreviations: CI, confidence interval; PAR, prediction for atherosclerotic cardiovascular disease risk; UKPDS, United Kingdom prospective diabetes study.
Discussion
The results presented here show that rt-CGM is likely to be cost-effective for the management of patients with T2D treated with basal insulin regimens with an ICUR in the baseline case of KRW 27.1 million per QALY, below the WHO-recommended WTP threshold of KRW 47 million per QALY. This was driven by an incremental QALY gain of 0.490 compared with SMBG over a lifetime horizon. Although sensitivity analyses showed the ICUR to be sensitive to a number of factors, the overall findings were robust with the intervention remaining cost-effective regardless of changes in treatment efficacy, rates of severe hyperglycemic and hypoglycemic events, time horizon, population age, duration of diabetes, and cardiovascular risk prediction equation used.
Such reductions in FoH and diabetes distress have been common findings in the use of rt-CGM for type 1 diabetes and have also been established for insulin-using patients with T2D, 37 who also commonly experience FoH. 44 Fear of hypoglycemia not only has negative psychological impacts, but can also have further influences on self-management and health-related quality of life (QoL) by promoting behaviors that reduce the likelihood of hypoglycemia such as avoidance of exercise.45,46 The reduction of FoH is likely to be of particular relevance to the sensor considered in this analysis, the G7 (Dexcom, San Diego, CA), which provides a predictive alarm that warns of imminent hypoglycemic events and has been shown to reduce hypoglycemia. 47 Similarly, the avoidance of the pain and inconvenience of frequent fingerstick testing that may impact QoL or adherence is expected to be a key benefit of rt-CGM over SMBG. 48
An important strength of this analysis was that it was based on costs and other data relevant to the South Korean population. These data were obtained from a previous analysis conducted using a South Korean payer perspective with cohort characteristics estimated using the Korean National Health and Nutrition Examination Survey. 25 In addition, cardiovascular risks were predicted using an equation specific to South Korea as cardiovascular mortality in East Asian populations has been shown to differ from that in Western populations, with higher age-adjusted stroke mortality but lower coronary heart disease mortality.22,23,49 This is in addition to differences in risk factors, such as higher smoking rates, lower serum total cholesterol, higher salt intake, and lower hypertension awareness and treatment rates. 49 The Korean equation used was shown to be conservative when compared with other risk equations in the sensitivity analysis. However, the equation of Park et al 43 may underestimate the cardiovascular benefits of reductions in HbA1c due to it being based on an asymptomatic healthy population with an average HbA1c of 5.5%. Nevertheless, the ICUR using this equation (KRW 27.1 million per QALY) was below both the WHO-recommended WTP threshold (KRW 47 million per QALY) and the WTP threshold reflecting the median ICER of novel drugs for rare diseases in South Korea (KRW 32 million per QALY). 20
The limitations of this study include a focus on the direct costs associated with diabetes and not accounting for more complex interventions combining rt-CGM with diabetes education or nutritional advice. The exclusion of indirect societal costs from this analysis likely leads to substantial underestimations of the total costs of diabetes, with indirect costs often being associated with a substantial burden.6,50,51 A further limitation of the generalizability of this study was the focus on one monitoring system. Although, as discussed above, the predictive alarms of this system may be beneficial in avoiding hypoglycemia, they may not be present on other models of rt-CGM devices. Alternatives may instead provide threshold-based alarms that will not necessarily facilitate pre-emptive action to avoid a hypoglycemic event. In a comparison of the G7 device to a previous generation with a threshold-based alarm rather than a predictive one, patients who used the G7 experienced 33% to 40% reductions in hypoglycemia. 47
Overall, the long-term cost-effectiveness analysis reported here suggests that rt-CGM improves QoL and is cost-effective in the management of T2D when compared with SMBG. This was found to be a robust result when considering a WTP threshold of KRW 47 000 and driven largely by gains in utilities associated with reductions in FoH and AFS.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251382570 – Supplemental material for Cost-Effectiveness Analysis of Real-Time Continuous Glucose Monitoring Versus Self-Monitoring of Blood Glucose in People With Type 2 Diabetes Treated With Non-Intensive Insulin Therapy in South Korea
Supplemental material, sj-docx-1-dst-10.1177_19322968251382570 for Cost-Effectiveness Analysis of Real-Time Continuous Glucose Monitoring Versus Self-Monitoring of Blood Glucose in People With Type 2 Diabetes Treated With Non-Intensive Insulin Therapy in South Korea by Ji Yoon Kim, Sabrina Ilham, Hamza Alshannaq, Richard F. Pollock, Martin Field, Gregory J. Norman, Sang-Man Jin and Jae Hyeon Kim in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
None.
Abbreviations
ACE, angiotensin-converting enzyme; AFS, avoidance of frequent fingerstick testing; ARR, absolute risk reduction; CDM, Core Diabetes Model; FoH, fear of hypoglycemia; HbA1c, glycated hemoglobin; ICER, incremental cost-effectiveness ratio; ICURs, incremental cost-utility ratios; PAR, Prediction for Atherosclerotic Cardiovascular Disease Risk; QALY; quality-adjusted life year; QoL, quality of life; RR, relative risk; rt-CGM, real-time continuous glucose monitoring; SHE, severe hypoglycemic event; SMBG, self-monitoring of blood glucose; T2D, type 2 diabetes; UKPDS, United Kingdom Prospective Diabetes Study; WHO, World Health Organization; WTP, willingness-to-pay.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.I., H.A., and G.J.N. are current employees of Dexcom. G.J.N. and H.A. hold stock or stock options in Dexcom. R.F.P. and M.F. are current employees of Covalence Research Ltd, which received consultancy fees from Dexcom to prepare the manuscript. S.-M.J., J.Y.K., and J.H.K. have no competing interests, as they did not receive any compensation from Dexcom for this work nor did they receive any grants from Dexcom for other projects.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the analysis, manuscript preparation, and the journal’s article-processing fees were provided by Dexcom.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
