Abstract
Hybrid closed-loop (HCL) systems have transformed outpatient diabetes management, yet their application in complex inpatient and hospital-based scenarios remains underexplored. This article examines the utilization of HCL systems in three challenging clinical contexts: the perioperative period, dialysis (hemodialysis and peritoneal dialysis), and during glucocorticoid therapy. Our article and case series examples provide an overview of the current available literature, preliminary data, and practical guidance for clinicians on HCL systems in these settings. Further research is urgently needed to establish the evidence base in this high-risk cohort.
Introduction
Commercially available advanced hybrid closed-loop (HCL) systems are able to modify insulin delivery via an insulin pump based on continuous glucose monitoring (CGM) and its own algorithm. They are widely used in the outpatient setting and are associated with improved glycemia and clinical outcomes. 1 The hospital setting, however, provides unique challenges with rapid and unpredictable glucose fluctuations. As a result, its adoption and impact in hospital settings remain limited.2-4
Several barriers hinder the full potential of these devices in hospital settings, particularly in the perioperative setting, on dialysis and while on glucocorticoids. These barriers include concerns about the accuracy of inpatient CGM, insulin pump, and HCL use, lack of integration of data into electronic health record, inadequate staff training on the management of these technologies in specialist settings, and the absence of standardized protocols in this complex cohort.5-11 Furthermore, there are limited research and guidance on real-world use and safety. 12
Hybrid closed-loop systems show promise in surgical settings, with early trials indicating better glucose control and less hypoglycemia than standard care.13-15 Key challenges include device interference during procedures and the need for trained monitoring. Glucocorticoid use increases insulin resistance and glucose levels. Hybrid closed-loop systems can adapt insulin delivery accordingly but still require close monitoring and manual adjustments. In people on dialysis, HCL systems may improve glycemia despite complex insulin dynamics, offering a safer, more tailored approach.
We have highlighted five real-life clinical scenarios (see cases 1-5) in which the use of diabetes technology was supported in each of these complex settings. For each situation, we highlight the multidisciplinary team (MDT) recommendations within this setting.
HCL Use in Perioperative Settings
Current perioperative strategies for diabetes management emphasize individualized care to maintain safe and optimal blood glucose levels during the surgical period. National guidelines in the United Kingdom from the Joint British Diabetes Societies for Inpatient Care (JBDS-IP) recommend maintaining intraoperative glucose levels between 6.0 and 10 mmol/L (108-180 mg/dL) for most individuals to minimize complications, such as infection or delayed healing.16,17
Pre-operative assessments should include a detailed review of diabetes and medical history, current medications and personal use of diabetes devices technologies, such as CGMs, insulin pumps, and HCL systems, while the post-operative phase focuses on resuming appropriate diabetes management plans and ensuring adequate glycemia before discharge. The use of HCL systems during the perioperative period is an emerging area in perioperative diabetes care. While these systems offer promise for maintaining stable glucose control in the perioperative settings, their application in these settings remains under investigation. Preliminary evidence from small, randomized trials suggests that such systems can provide superior glucose control compared with conventional methods, particularly in preventing hypoglycemia, making them a compelling option for selected patients undergoing surgery.
Herzig et al conducted a single-center study involving 45 adult surgical patients with diabetes, excluding those with type 1 diabetes (T1D). Participants undergoing surgeries lasting more than two hours were assigned to either fully closed-loop CamAPS HX (Camdiab, Cambridge, UK) system or to standard diabetes management. The study reported no adverse events and demonstrated improved time in target glucose range (76.7 ± 10.1% in the closed loop vs 54.7 ± 20.8% in the control group; P < .001). 15 In a case series of seven children undergoing eight brief surgical procedures, during which activity or exercise modes were enabled on the HCL systems, no hypoglycemic or hyperglycemic interventions from clinicians were required. 14 Similarly, Seget et al 13 reported on two children with T1D undergoing endoscopy and umbilical herniorrhaphy, where HCL was maintained in a temporary higher glucose target range. Neither individual experienced adverse events or required discontinuation. 13
Potential challenges and risks in the perioperative use of HCL systems, however, include device compatibility with surgical equipment (eg, interference during magnetic resonance imaging [MRI] or diathermy), potential accuracy issues during rapid physiological changes and CGM compression, as well as the need for intensive monitoring by trained staff. 18
Several perioperative diabetes guidelines including perioperative management of conventional (non-HCL) insulin pump therapy have been published by international professional societies, but universally established guidelines specifically for the use of commercial HCL systems during surgery remain unavailable. Local guidance, such as those implemented at the authors’ own institution and which is adapted from published perioperative diabetes management guidelines16,17 are outlined in Table 1. This could serve as pragmatic clinical care models for promoting safer and more standardized use of HCL systems in selected surgical patients. Future developments, including studies in broader perioperative inpatient cohorts and regulatory approvals, are likely to shape more definitive guidelines for HCL use in the perioperative period.
Guidance for HCL Use in Perioperative Settings.

Here, we present two clinical cases to illustrate perioperative HCL application following the guidance used.
Case 1
History
Seventy-year-old man with a long-standing history of T1D managed on a hybrid closed-loop therapy (Medtronic 780G; MiniMed Medtronic, Northridge, CA, USA) by his outpatient diabetes team. He was referred to the surgical team for open repair incisional hernia. He had a preoperative assessment and his glycated hemoglobin (HbA1c) prior to surgery was 47 mmol/mol. The rate of his two-week time in range (3.9-10.0 mmol/L) and below range (3.9 mmol/L) was 67% and 0%, respectively. Hybrid closed-loop target glucose was set at 6.7 mmol/L. His operation was performed under general anesthesia as a day case. He was advised to fast for six hours for food and two hours for clear fluids, and was placed first on the operating list.
Management
He was advised to ensure his insulin pump and CGM sensor were sited distant to the surgical site and diathermy earthing plate.
Advice given to theater and clinical staff:
Measure capillary blood glucose hourly while in theater and during recovery.
If the difference between the sensor and capillary blood glucose > 3 mmol/L in recovery, recalibrate the sensor if feasible. If recalibration is not feasible, remove the insulin pump and revert to conventional blood glucose monitoring and insulin therapy management as per usual hospital guidance.
A new glucose sensor and insulin pump to be re-sited by the patient, once able to do so independently.
Outcome
Glucose levels were within normal range throughout preoperative, operative, and post-operative periods (Figure 1).
No hypoglycemia treatment or insulin correction doses were needed.
The individual recovered well in the recovery room, and was able to use an insulin pump/HCL without interruption while in hospital.
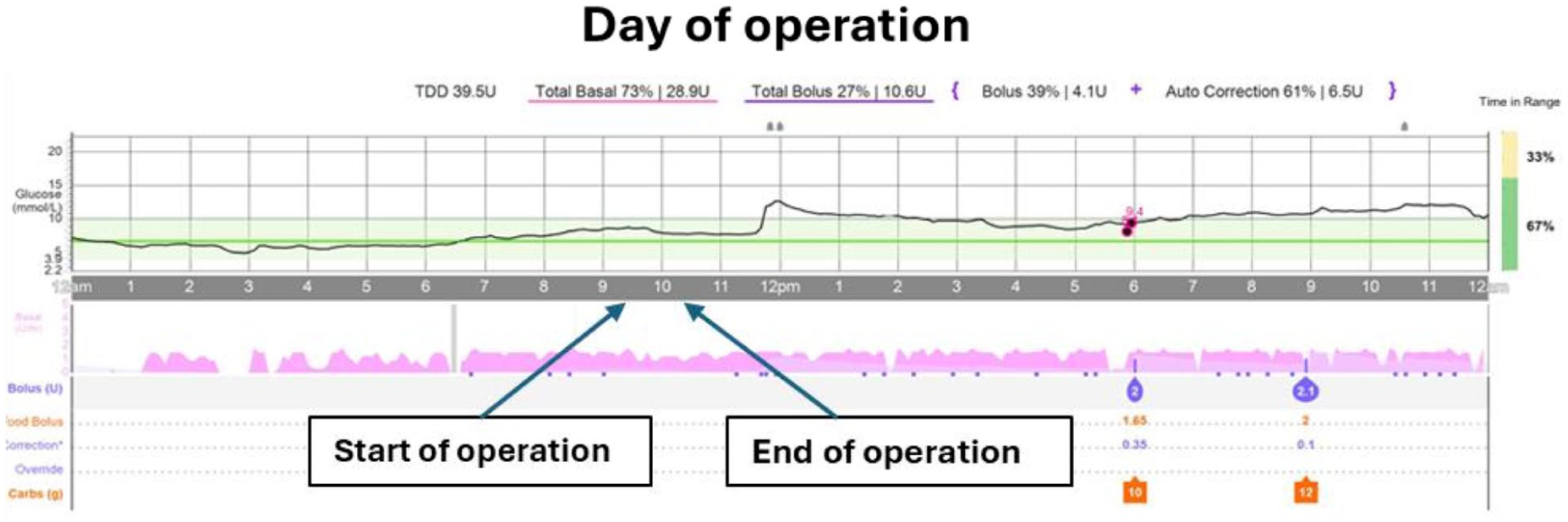
Case 1: Glycemia using HCL system on day of open repair incisional hernia. HCL system and settings: Medtronic MiniMed 780G with Guardian 4 sensor; insulin Novorapid, active insulin time, four hours; insulin sensitivity factor, 4 mmol/L/unit; HCL target glucose, 6.7 mmol/L.
Case 2
History
Thirty-two-year-old woman with long-standing history of T1D managed on hybrid closed-loop (Tandem T:slim X2 with Control IQ technology; Tandem Diabetes Care, San Diego, CA, USA) by her outpatient diabetes team. She was referred to the oral and maxillofacial surgery team for bilateral osteotomy of her mandible due to deformity of facial bone. She had a pre-operative assessment and her HbA1c prior to surgery was 48 mmol/mol. The rate of her two-week time in range (3.9-10.0 mmol/L) and below range (3.9 mmol/L) was 81% and 0%, respectively. Her operation was planned to be performed under general anesthesia. She was admitted to hospital the day before the operation (for reasons other than diabetes), advised to fast for six hours for food and two hours for clear fluids, and was placed first on the operating list.
Management
She was advised to ensure her insulin pump and CGM sensor were sited distant to the surgical site and diathermy earthing plate. She was advised to place her pump in “Activity Mode” on the day of the procedure, to reduce the risk of hypoglycemia in the operating theater.
Advice given to theater and clinical staff:
Measure capillary blood glucose hourly while in theater and during recovery.
If the difference between the sensor and capillary blood glucose > 3 mmol/L in recovery, recalibrate the sensor if feasible. If recalibration is not feasible, remove the insulin pump and revert to conventional blood glucose monitoring and insulin therapy management as per usual hospital guidance.
A new glucose sensor and insulin pump to be re-sited by the patient, once able to do so independently.
Outcome
Glucose levels were with normal range throughout pre-operative, operative, and post-operative periods (Figure 2).
No hypoglycemia treatment or insulin correction doses were needed.
The individual recovered well in the post-recovery room, and was able to use insulin pump/HCL without interruption while in hospital.
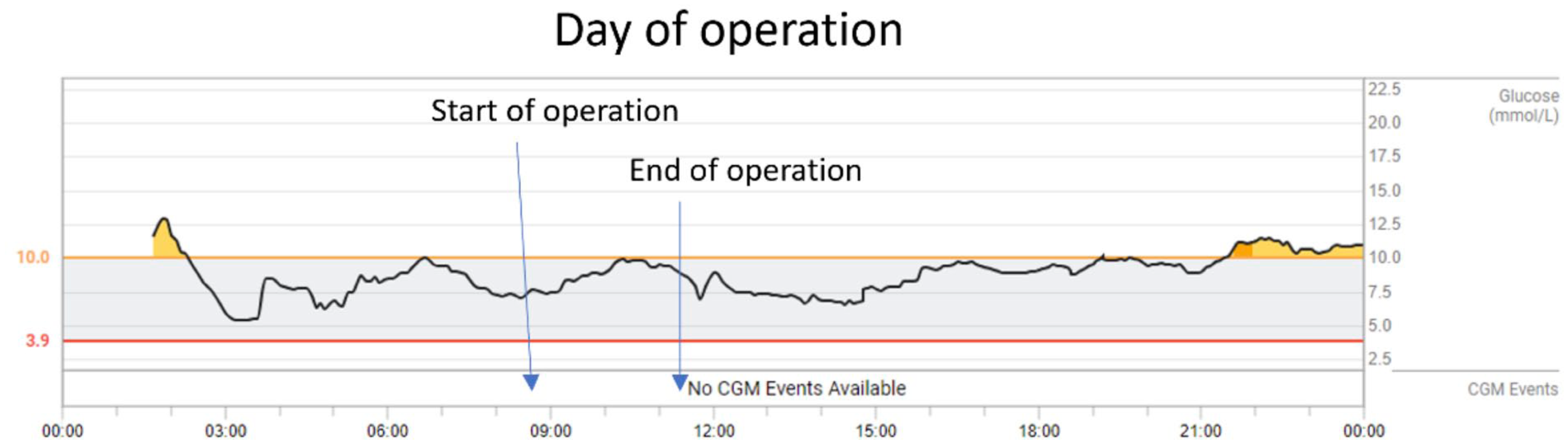
Case 2: Glycemia using HCL system on day of oral and maxillofacial surgery. HCL system and settings: Tandem T:slim X2 with Dexcom G6 CGM; insulin Novorapid; active insulin time, five hours; basal rates ranged from 0.2 to 0.5 units/h; insulin sensitivity factor, 3 mmol/L/unit; and HCL target glucose range, 6.25 to 8.9 mmol/L.
HCL Use and Corticosteroids
Corticosteroids (glucocorticoids) pose another challenging scenario for HCL systems in hospital, causing severe insulin resistance and uncontrolled hyperglycemia. There are very limited data of HCL use in this area.
In the real-world implementation study of a fully closed-loop CamAPS HX system for inpatients with diabetes (type 2 and/or pancreatogenic diabetes), corticosteroids were used by 11 people (34%). Although outcomes were not subanalyzed for this cohort, there were no episodes of severe hypoglycemia or diabetic ketoacidosis during closed-loop use. 19 Valenzano et al reported two cases of adults with T1D on oral dexamethasone and HCL. In both cases, post-prandial glucose improved with only a slightly higher rate of hypoglycemia. 20
Another case series of six people post-transplant (simultaneous kidney/pancreas or pancreas only) has highlighted the potential use of HCL systems in post-transplant settings, where steroids and other immunosuppressive treatments can make diabetes management particularly challenging. 21
Here, we present a clinical case to illustrate HCL use during intravenous glucocorticoids.
Case 3
History
Twenty-two-year-old man with T1D managed on hybrid closed loop Omnipod 5 (Insulet Corporation, Billerica, MA, USA) with FreeStyle Libre 2 Plus (Abbott Diabetes Care, Alameda, CA, USA) by his outpatient diabetes team. He was admitted for an elective Sistrunk’s procedure (excision of thyroglossal duct and cyst). His pre-operative HbA1c was not documented, but was known to be managing his diabetes effectively with the HCL system with baseline time in range 70%. The HCL system was continued intraoperatively, with target glucose set at 6.7 mmol/L. Post-operatively, he received intravenous dexamethasone 6 mg for three consecutive days.
Management
The HCL system was continued post-operatively while on high-dose intravenous steroids. He was reviewed by the diabetes team and an adjustment of the lunchtime insulin-to-carbohydrate ratio (ICR) was made from 1:12 to 1:10 g between 12:00 and 19:00 hours to mitigate steroid-induced hyperglycemia. Advice given to the individual and clinical staff:
The ICR was strengthened in the afternoon and the individual was advised to make frequent corrections during the time.
Management plan included monitoring for elevated blood glucose and ketones, and switching from automated to manual mode if ketones > 1.5 mmol/L. If ketone levels exceeded 3 mmol/L or if the individual was unable to self-manage, advised to stop the pump and providing basal insulin via subcutaneous injection.
The inpatient diabetes team reviewed glycemia daily.
Outcome
Glucose levels remained elevated in the afternoon despite the use of HCL in automated mode (Figure 3). However, there were no episodes of ketonemia observed. In view of the short duration of steroid therapy, HCL was continued in this instance.
The individual recovered well post-operatively. He was advised to adjust his ICR/insulin sensitivity factor (ISF) back to baseline settings approximately 48 to 72 hours of the following discharge.
In retrospect, the individual would have also benefited from a stronger ISF during the afternoon to mitigate against steroid-induced hyperglycemia, with target settings adjusted back to baseline. Despite multiple correction attempts on day 2, the system only delivered minimal boluses (1 unit on each of six occasions) likely due to the presence of substantial insulin on board.
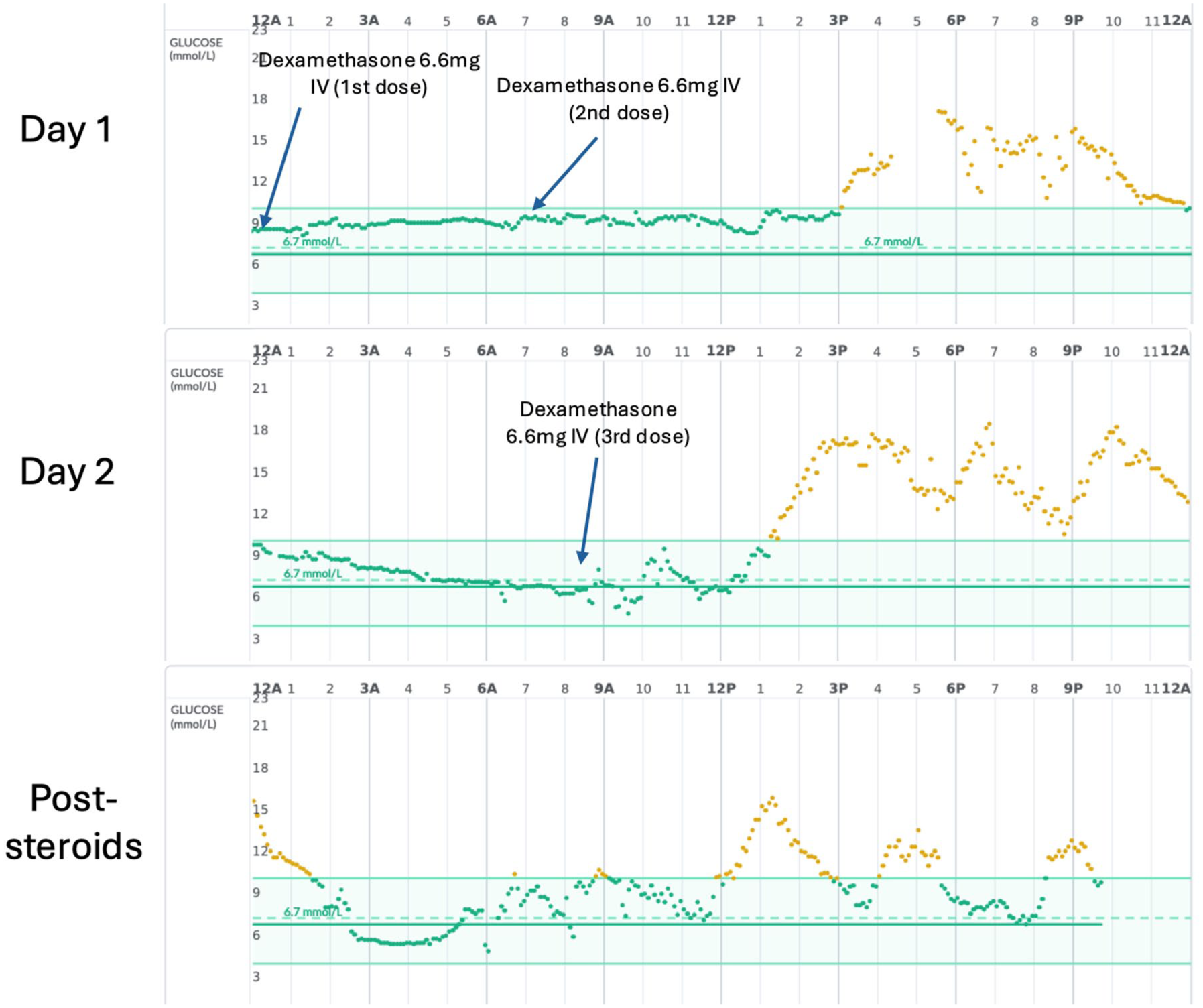
Case 3: Glycemia in hospital using HCL system after three doses of intravenous dexamethasone and the following day. HCL system and settings: Omnipod 5 with FreeStyle Libre 2 Plus; insulin Novorapid; insulin active time, 5.5 hours; insulin sensitivity factor, 5.5 mmol/L/unit; and HCL target glucose, 6.7 mmol/L. Full pump downloads are available in the Supplementary Material.
HCL Use and Dialysis
Diabetes management in people with end-stage kidney disease (ESKD) is particularly complex due to disruptions in glucose homeostasis. People with diabetes and advanced kidney disease have highest rates of admissions of hospitalization with hypoglycemic and hyperglycemic emergencies, 22 wide glucose fluctuations, high mortality, and low quality of life. 23
End-stage kidney disease is linked to insulin resistance, altered insulin pharmacokinetics, and marked glycemic variability. 23 Dialysis further disrupts glycemia through various mechanisms. Insulin may be lost via diffusion or adsorption to the dialysis membrane, contributing to post-dialysis hyperglycemia. 24 Conversely, correcting uremia during hemodialysis can improve insulin sensitivity and lower glucose levels. 25 Dialysis-related hypoglycemia may also result from glucose loss into the dialysate 26 and increased erythrocyte glucose uptake, possibly due to intracellular pH shifts during dialysis. 27 For individuals on peritoneal dialysis, hypertonic glucose solutions are often used to improve ultrafiltration and may contribute to dysglycemia. While modern osmotic agents such as icodextrin practically do not enter the bloodstream and provide higher efficiency than glucose, glucose remains preferable in certain cases.28,29
The efficacy and safety of HCL systems have yet to be established in people with diabetes and advanced kidney disease, particularly those undergoing hemodialysis and peritoneal dialysis. While CGM accuracy has been established for certain sensors,30-32 it has not been consistently demonstrated across all CGM devices integrated into HCL systems.
In a study with 26 adults with type 2 diabetes on dialysis, individuals with the fully closed-loop CamAPS HX system demonstrated increased time in target range (5.6-10.0 mmol/L) by 15.1% with lower mean glucose than standard insulin therapy. Hypoglycemia was reduced with no severe events occurring during its use. 33
In a single-center case series of four individuals with long-standing T1D and ESKD on thrice-weekly hemodialysis, significant improvement in time in range was observed (pre-HCL 43.5% vs post-HCL 64.8%; P = .015), and a reduction in time above range (pre-HCL 55.5% vs post-HCL 34.8%; P = .03) over a mean follow-up of 4.5 months. 34 A further case series of nine people with T1D on hemodialysis demonstrated that HCL use led to significant improvements in time in range (pre-HCL 39.7% vs post-HCL 59.8%; P = .001), and reduced time below range (pre-HCL 4.0% vs post-HCL 1.4%), although this did not reach statistical significance (P = .10). 35
Due to the changes in insulin pharmacokinetics and pharmacodynamics that occur in advanced kidney disease, further research is needed to understand the optimal HCL system settings for this cohort. Table 2 provides pragmatic guidance, such as those implemented at the authors’ own institution.
Guidance for HCL Use in Hemodialysis.

Case 4
Thirty-year-year-old man with long-standing T1D and established diabetes-related complications, including diabetic retinopathy, diabetic nephropathy, peripheral neuropathy with previous neuropathic ulceration, and possible diabetic gastroparesis.
He was established on an HCL Tandem T:slim X2 pump with Control-IQ technology via Dexcom G6 (Dexcom Inc., San Diego, CA, USA). After a few months, he progressed on to requiring continuous ambulatory peritoneal dialysis (five days/week). The regimen consisted of three daily exchanges: two liters of 1.36% glucose solution for two exchanges, and one exchange with icodextrin.
Outcome
Although he was flagged for diabetes review, this did not happen at the time he was transitioned on to dialysis. Soon after starting dialysis, he experienced two episodes of severe hypoglycemia requiring third-party assistance requiring A&E admission (Figure 4), prior to review by the diabetes team. It was identified that hypoglycemia was precipitated by two key factors:
Failure to replace the Dexcom G6 sensor after it expired overnight and promptly reactivating automated mode, meaning Control-IQ technology was unable to suspend insulin delivery in response to falling glucose levels.
Pre-programmed basal insulin settings through the pump had not been adjusted (reduced) at time of initiation of dialysis, resulting in relative insulin overdose.
These issues were addressed by reinforcing education around timely sensor replacement, reviewing alarm settings, and reprogramming basal rates based on his insulin requirements on dialysis. The Dexcom G6 CGM sensor was upgraded to the Dexcom G7, which has demonstrated improved accuracy in people on dialysis.30,31 Backup safety measures were discussed, including use of temporary basal reductions. This case highlights the importance of close monitoring and active involvement of the diabetes team, particularly during initiation or transition to dialysis.
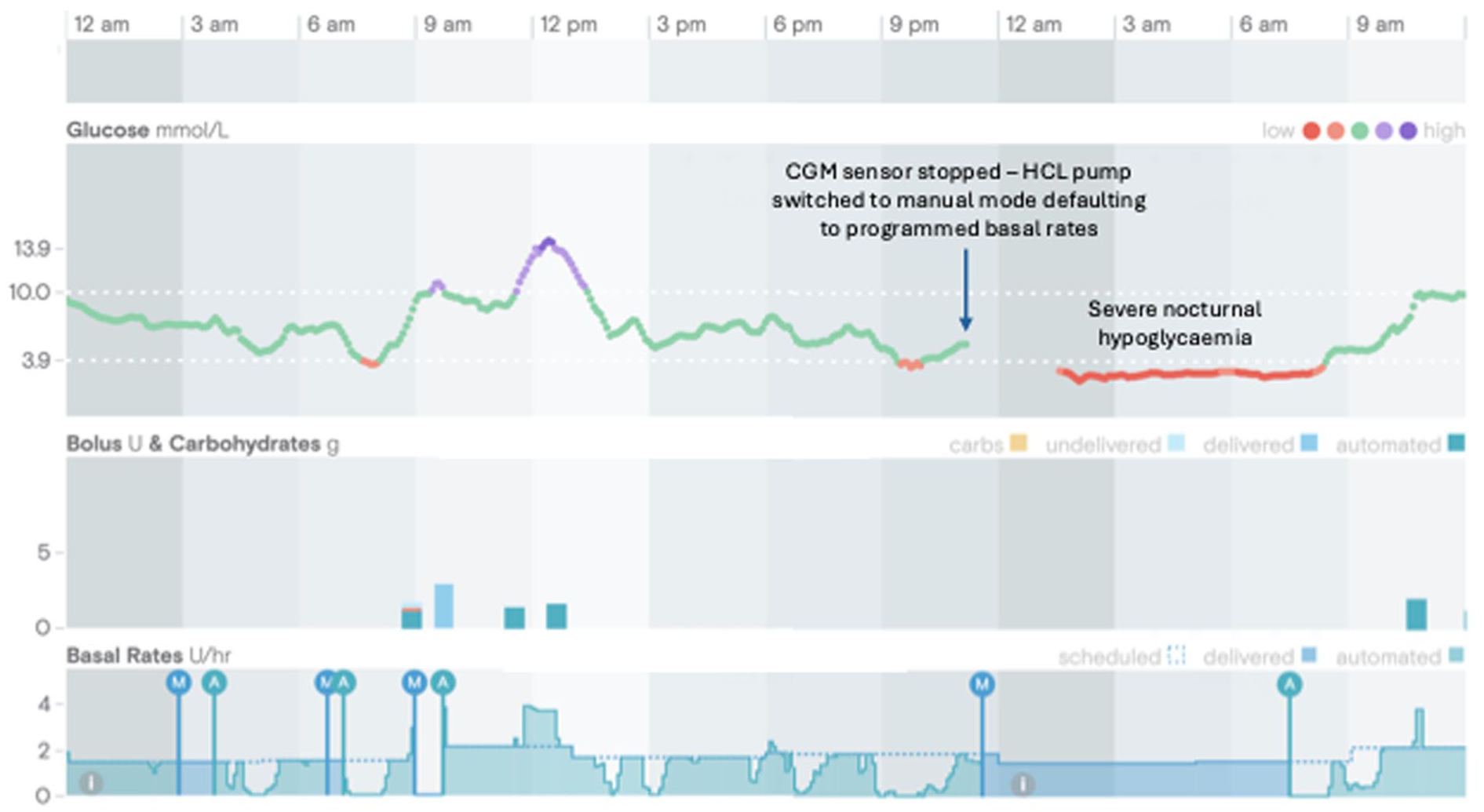
Case 4: Episode of severe hypoglycemia in individual on peritoneal dialysis. HCL system and settings: Tandem T:slim X2 with Dexcom G6 CGM; insulin Novorapid; active insulin time, five hours; basal rates, 1.4 to 2.1 units/h; insulin sensitivity factor 1.5 mmol/L/unit; HCL target glucose, 6.25 to 8.9 mmol/L. Settings after episodes of hypoglycemia and diabetes team review adjusted basal rates to 0.72 units/h and insulin sensitivity factor to 3.7 mmol/L/unit.
Case 5
History
Thirty-five-year-old man with long-standing T1D and established hemodialysis (in-center three times/wk) following failed kidney transplantation. He had diabetes-related complications, including bilateral diabetic retinopathy and peripheral neuropathy. He was on sensor-augmented pump therapy (Medtronic MiniMed 640G and Dexcom G6).
Glucose data on pump therapy alone demonstrated time in range (3.9-10 mmol/L) 47%, mean glucose 10.4 mmol/L, glucose management indicator (GMI) 7.8% (62 mmol/mol), and coefficient of variation (CV) 41.4%.
Management
Given persistent glycemic variability and preparation toward transplant, he was transitioned to an HCL (Medtronic MiniMed 780G with Simplera Sync CGM). Device settings were initiated at a target glucose of 6.1 mmol/L (higher than the recommended threshold from the manufacturer).
Outcome
Post-HCL therapy, time in range increased from 47% to 75%, mean glucose reduced from 10.4 to 8.3 mmol/L, GMI improved from 7.8% (62 mmol/mol) to 6.9% (52 mmol/mol), and CV improved from 41.4% to 32.8% (Figure 5). Time below range (< 3.9 mmol/L) reduced to 1% (no time < 3.0 mmol/L).
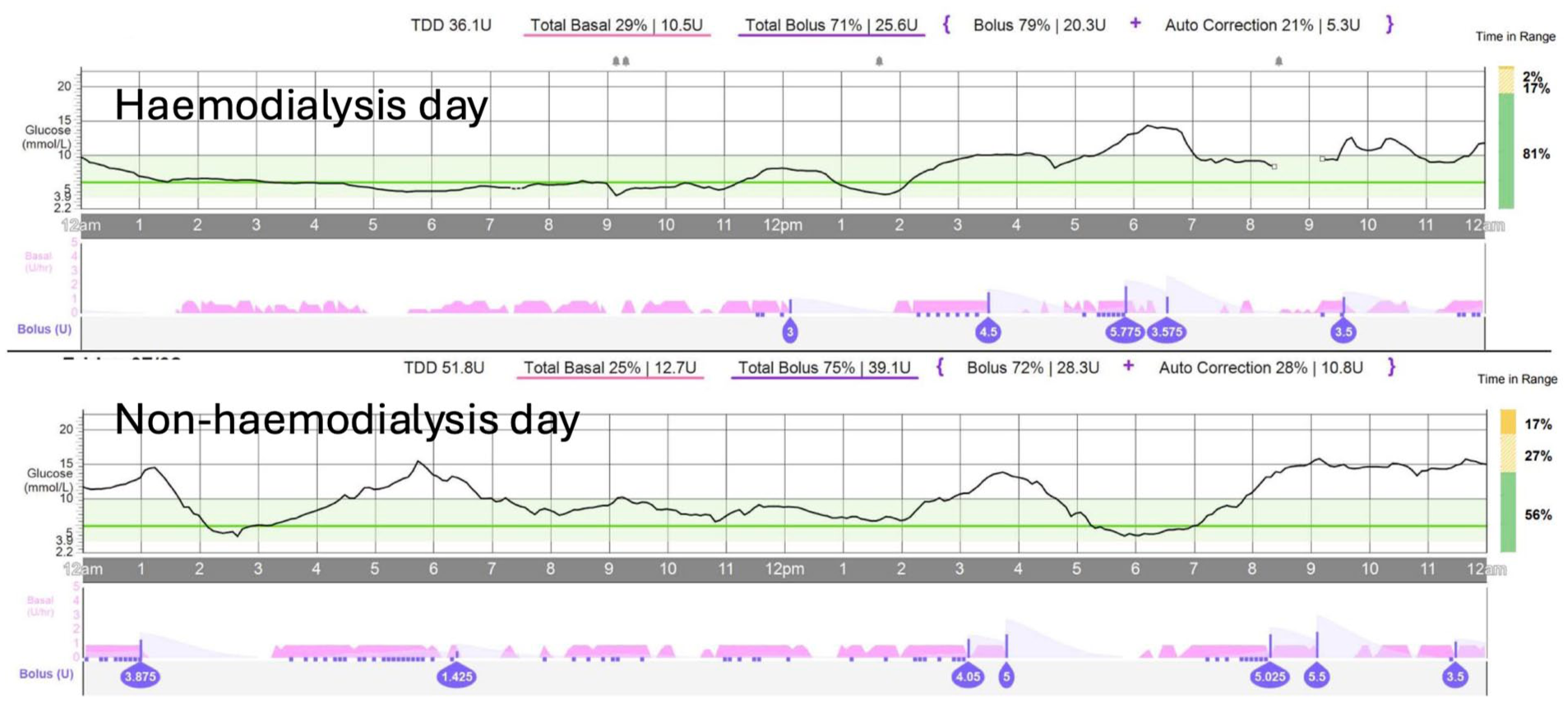
Case 5: Glycemia using HCL system while on hemodialysis. HCL system and settings: Medtronic MiniMed 780G with Guardian 4 sensor; insulin Humalog, active insulin time, two hours; insulin sensitivity factor 2.1 mmol/L/unit; target glucose 6.7 mmol/L.
He engaged well with the system, maintaining 96% sensor wear and 100% SmartGuard use. Education focused on system use around hemodialysis sessions.
Insulin requirements remained stable, with 67% delivered as bolus insulin, supported by automated corrections. The user reported increased confidence in managing diabetes around dialysis sessions.
Conclusion
This article, along with the cases presented in this series, highlight the expanding role of HCL systems in complex clinical scenarios amongst people with T1D. Overall, HCL use in these scenarios appears promising and highlights the adaptability and potential benefits of HCL systems in diverse and challenging settings. However, their successful implementation requires comprehensive education and understanding of the system, frequent monitoring, and close collaboration among MDTs.
We recommend adopting a multidisciplinary approach involving diabetologists and diabetes specialist nurses with expertise in HCL systems, in conjunction with other relevant specialists, such as anesthesiologists and surgeons. Such collaboration is essential to ensure the safe and effective use of HCL systems within complex inpatient and hospital-based settings. Close working relationships between these specialist teams and inpatient diabetes services allow for better planning and guidance on adjusting the system’s settings (eg, using higher target glucose to mitigate hypoglycemia)—and establish a clear and individualized management plan prior to discharge from hospital.
Most available studies on HCL use in perioperative care, on glucocorticoids and on dialysis focus on people with T2D. While these data provide useful insights, they may not fully reflect the unique challenges of T1D. Another important area with limited published evidence is the use of HCL systems in people with T1D receiving artificial nutrition. A recent case report has described successful management of blood glucose levels with HCL in this setting, but further studies are needed to establish feasibility and best practice. 38 Further research, including larger cohort studies and randomized controlled trials in people with T1D, are urgently required to establish outcomes and standardized protocols in these specialized settings.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251381282 – Supplemental material for Managing Hybrid Closed-Loop Insulin Delivery in Complex Clinical Scenarios: Focus on Perioperative, Dialysis, and Glucocorticoid Use
Supplemental material, sj-docx-1-dst-10.1177_19322968251381282 for Managing Hybrid Closed-Loop Insulin Delivery in Complex Clinical Scenarios: Focus on Perioperative, Dialysis, and Glucocorticoid Use by Parizad Avari, Catherine Robinson, Lalantha Leelarathna and Hood Thabit in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors wish to thank all members of the multidisciplinary team.
Abbreviations
CGM, continuous glucose monitoring; CV, coefficient of variation; ESKD, end-stage kidney disease; GMI, glucose management indicator; HCL, hybrid closed loop; ICR, insulin-to-carbohydrate ratio; ISF, insulin sensitivity factor; MRI, magnetic resonance imaging; T1D, type 1 diabetes; VRIII, variable rate intravenous insulin infusion.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PA is funded by NIHR Grant No. 132960. She has received equipment from Dexcom for investigator-initiated studies. LL has received research support from Abbott Diabetes Care, and Dexcom; participated in advisory groups for Abbott Diabetes Care, Insulet, Dexcom, Medtronic, and Roche Diabetes; and has received fees for speaking from Sanofi, Insulet, Medtronic, and Abbott Diabetes Care. HT has received speaker honoraria from Eli Lilly and Dexcom Inc, having received research support from Dexcom Inc and having served on advisory panels for Roche Diabetes.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
