Abstract
Background:
The increasing incidence and prevalence of diabetes have led to a strain on health care systems, necessitating innovation in diabetes care delivery and substantial resource allocation for disease management and treatment of complications. Recently, Saudi Arabia has made significant strides in digital transformation, implementing cutting-edge technologies across various sectors, including the health sector.
Aims:
To develop a report that examines the current status, identify challenges and opportunities in implementing diabetes technology solutions for diabetes management, and provide a strategic roadmap for enhancing the accessibility to diabetes technology solutions.
Methods:
The Saudi National Diabetes Center (SNDC) convened a group of experts to develop a comprehensive report that serves a dual purpose. First, it provides a narrative review on insulin pumps and CGMs that are approved by the SFDA and discusses their clinical effectiveness based on local studies. Second, it presents a comprehensive report on the current status and future directions of diabetes technology in Saudi Arabia.
Results:
Our experts identified six major barriers to the adoption of diabetes technology solutions in Saudi Arabia and proposed 14 strategic initiatives that can help overcome these barriers and transform the current overwhelmed diabetes care model into a more innovative, effective, efficient, safe, accessible, and cost-effective model of care.
Conclusion:
While Saudi Arabia offers one of the most diverse ranges of diabetes technology solutions globally, major accessibility barriers persist. The SNDC Diabetes Technology Report is the first report developed primarily for policymakers, regulatory bodies, clinicians, academicians, and industry partners to address these accessibility barriers.
Background
Diabetes poses a significant health and economic burden in Saudi Arabia and the Middle East and North Africa (MENA) region, with the region experiencing the highest prevalence rates globally. 1 Saudi Arabia is among the top five countries in the MENA region, and top 10 countries worldwide, in diabetes prevalence among adults with an estimated age-standardized prevalence of 23.1%. 1 The increasing incidence and prevalence of diabetes have led to a strain on the region health care systems, necessitating substantial resource allocation for disease management and treatment of complications. The health burden of diabetes in Saudi Arabia is evident in the rising rates of cardiovascular diseases, renal failure, non-traumatic amputation, and blindness associated with poorly controlled diabetes. 2 It is estimated that one in two individuals with diabetes who visit diabetes clinics in Saudi Arabia have at least one microvascular complication. 2 Moreover, the economic impact of diabetes and its complications is substantial, encompassing direct medical costs for the management of diabetes and its complications, and indirect costs due to worsened quality of life, absenteeism and reduced productivity at work, and premature mortality.3,4 People with diabetes in Saudi Arabia have ten times the health care expenditures compared to those without diabetes. 3 Such high prevalence and economic burden of diabetes presents a considerable challenge to the nation’s health care infrastructure and economic development. This underscores the urgent need for developing and implementing more accessible, efficient, and cost-effective strategies for both preventing new cases of diabetes and improving the management of existing ones to prevent diabetes complications.
Over the recent years, Saudi Arabia has made significant strides in digital transformation, implementing advanced technologies across various sectors and launching initiatives like the Ministry of Health (MOH) Vision for E-Health, 5 highlighting its commitment to becoming a technologically advanced nation. In diabetes, the utilization of continuous glucose monitors (CGMs) and insulin pumps, coupled with smartphone apps and cloud-based platforms, can significantly enhance the quality, accessibility, efficiency, and cost-effectiveness of diabetes management. 6 These technologies enable real-time glucose monitoring, automated insulin delivery, and data-driven decision making, allowing for more precise and personalized treatment regimens. By providing patients with continuous feedback and health care providers with comprehensive data, diabetes digital health solutions can improve glycemic control, minimize risk of complications, and enhance the overall quality of life among people living with diabetes. Such innovative management strategy is expected to yield significant reductions in health care expenditure associated with diabetes and its complications. Moreover, diabetes technology solutions can bridge geographical barriers, extending specialized diabetes care to remote areas and alleviating the strain on health care facilities. The potential for remote monitoring and virtual consultations can increase the efficiency and safety of health care delivery and reduce the need for frequent in-person visits and potentially lowering the overall health care costs.
In response to the rapidly evolving landscape of diabetes technologies and their transformative potential in diabetes care, this comprehensive report examines the current status, challenges, and opportunities in implementing diabetes technology solutions for diabetes management in Saudi Arabia and provides an in-depth analysis of emerging technologies, implementation frameworks, and evidence-based recommendations. Through this analysis, we aim to bridge the gap between technological innovation and practical implementation while addressing critical aspects of accessibility and clinical outcomes in diabetes care in Saudi Arabia.
Materials and Methods
Setting
As of 2024, the total population of Saudi Arabia exceeds 35 million individuals with approximately 19.6 million Saudi citizens and 15.7 million non-Saudi residents. 7 Most Saudi citizens with diabetes, including governmental-sector employees, receive care through the public health care sector, where health care services are provided free-of-charge. In contrast, all non-Saudi residents, and some of the private-sector Saudi employees, rely on health insurance coverage provided by their employers, to access health care services. The Saudi health insurance system is relatively young and continues to evolve rapidly. The Council of Health Insurance (CHI) is the governmental body that oversees and regulates the health insurance sector in Saudi Arabia to ensure compliance with policies, quality of services, and dispute resolutions. The Saudi Food and Drug Authority (SFDA) is another key player in the health care system in Saudi Arabia. The SFDA oversees and regulates medical devices to ensure that they meet the safety, quality, and performance standards.8,9
Methods
This article serves a dual purpose. First, it provides a narrative review on insulin pumps and CGMs that are approved by the SFDA and discusses their clinical effectiveness based on local studies. Second, it presents a comprehensive report on the current status and future directions of diabetes digital health in Saudi Arabia. The two parts were developed primarily for health care policymakers who seek to integrate digital health into national strategies, health care professionals (HCPs) who may utilize these technologies in their clinical practice, academic researchers who advance the field through research and training, and industry partners who develop innovative solutions.
Part 1: Narrative review on insulin pumps and CGMs in Saudi Arabia
A narrative review on the insulin pumps and CGMs available in Saudi Arabia was performed using the following keywords “insulin pump,” “continuous glucose monitoring,” and “diabetes technology” and “Saudi Arabia” alone and in combination to retrieve available local literature from PubMed from January 2019 until January 2024. We limited the search to studies that examined the effectiveness and safety of insulin pumps and CGMs in patients with type 1 and type 2 diabetes in Saud Arabia.
Part 2: Diabetes technology report
To develop this report, the Saudi National Diabetes Center (SNDC) formed a working group of five national experts in diabetes digital health in Saudi Arabia. The working group included endocrinologists, a diabetologist, and representatives from the Saudi Food and Drug Authority (SFDA) and Council of Health Insurance (CHI) who identified the topics that need to be covered in the report. Subsequently, the group collaboratively drafted the report based on the evidence found in the narrative review of literature, clinical experience, and published local and international policies and regulatory documents.
Following this, the drafted report underwent a comprehensive review by the SNDC Scientific Committee composed of adult and pediatric endocrinologists and representatives from 13 health sectors in Saudi Arabia (Ministry of Health, Ministry of Defense, Ministry of Interior, National Guard Health Affairs, King Faisal Specialist Hospital & Research Center, Saudi Red Crescent Authority, Ministry of Education, Saudi Commission for Health Specialties, The Public Health Authority, Presidency of State Security, Federation of Saudi Chambers, SFDA, and CHI). This manuscript does not involve human subjects or primary data collection; therefore, IRB approval was not required.
Results
Part 1: Narrative Review on Insulin Pumps and CGMs in Saudi Arabia
The SFDA-approved insulin pumps and CGMs
Continuous glucose monitors
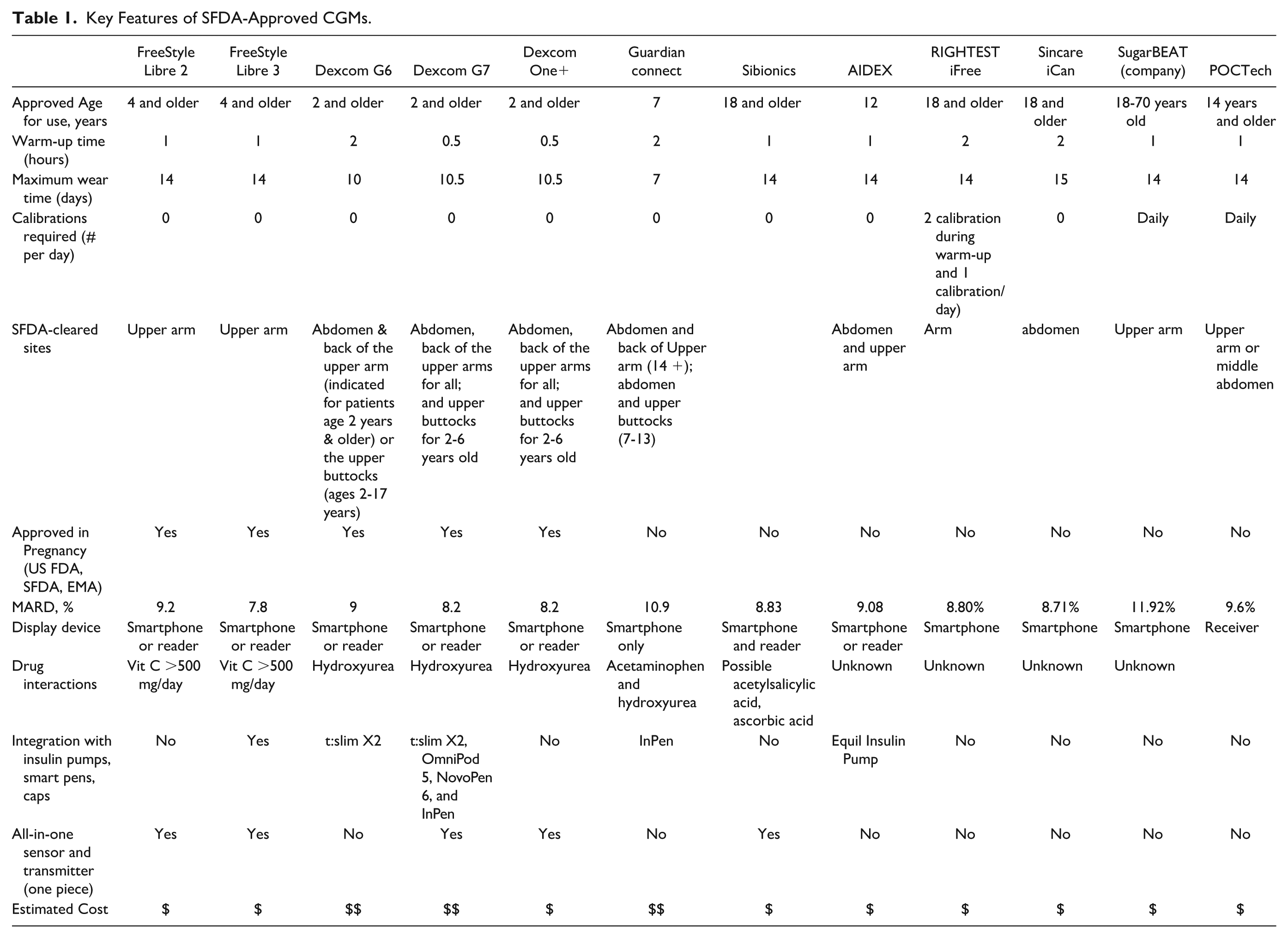
A continuous glucose monitor (CGM) is a wearable device that continuously monitors interstitial glucose, provides real-time glucose readings, and displays those data on a receiver or smartphone app. In addition, CGMs offer an option to transmit glucose data, through the cloud, to remote caregivers including HCPs. The latter feature empowers people living with diabetes and their HCPs as they practice personalized medicine, makes the clinic visits more efficient and safer, and facilitates telemedicine visits whenever and wherever needed.10,11 As of September 2024, the date of writing this report, there are 12 CGMs approved by the SFDA as highlighted in Table 1.
Key Features of SFDA-Approved CGMs.
Insulin pumps
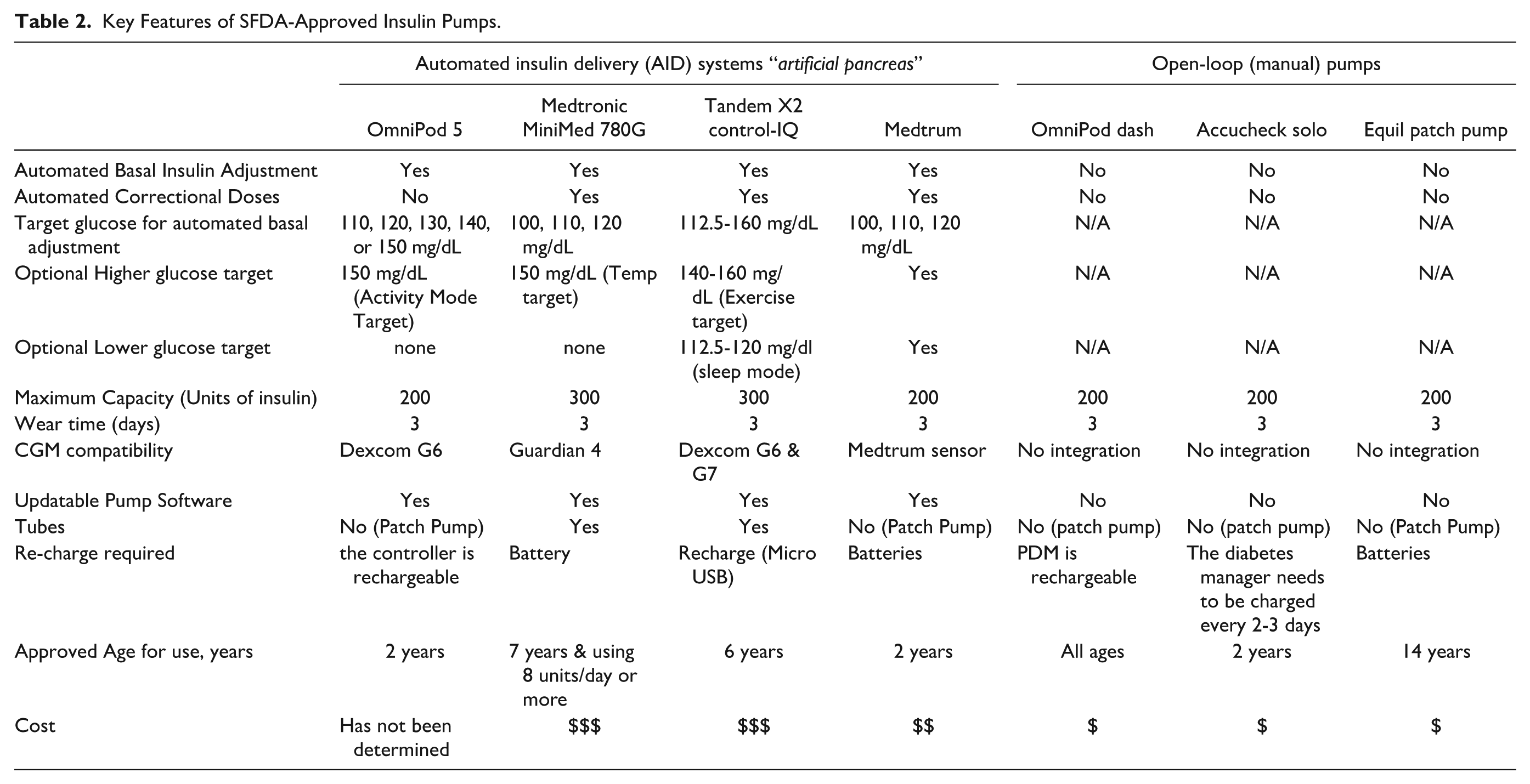
Insulin pumps have revolutionized the management of diabetes, particularly type 1 diabetes, offering improved glucose control and quality of life for patients. These devices come in various forms, including open-loop pumps and automated insulin delivery (AID) systems. Open-loop pumps require manual input for basal insulin dosing; while AID systems, also known as closed-loop systems, represent the latest advancement, integrating CGMs with insulin pumps to automatically adjust insulin delivery based on real-time glucose readings. Studies have shown that these technological advancements have significantly improved glycemic control (by >10% improvement in time in range), decreased HbA1c levels (by 0.1%-0.9%) and time spent in hyperglycemia, and maintained or decreased time below range.12 -14 While initial costs of these devices may be higher, the long-term benefits in terms of health outcomes and quality of life suggest that insulin pump therapy, particularly AID systems, could be a cost-effective solution for managing type 1 diabetes.15 -17
Another way to classify insulin pumps is by whether the pump is tubed or tubeless. Tubeless pumps offer increased convenience with a patch-like design. Some tubeless pumps are open loop and others are automated (ie, AID). Table 2 summarizes the key features of the SFDA-approved insulin pumps as of September 2024, the date of writing this report.
Key Features of SFDA-Approved Insulin Pumps.
Local scientific evidence for the effectiveness, safety, and cost-effectiveness of diabetes technology solutions in Saudi Arabia
Continuous glucose monitors
Type 1 Diabetes
(a) Glycemic Control:
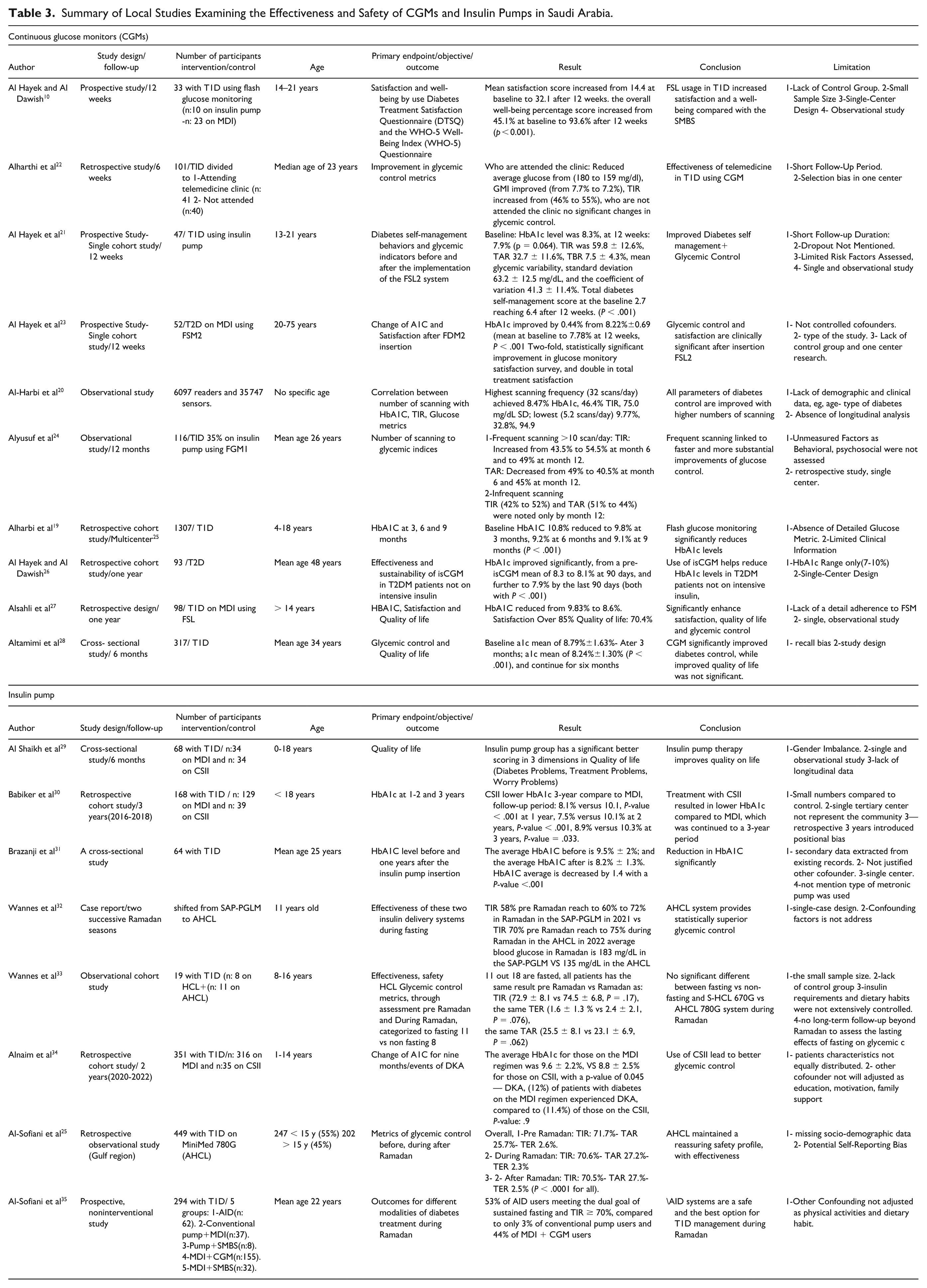
Numerous studies showed that CGMs improve glycemic control in people with T1D.18 -21 In pediatrics, the use of CGM resulted in a notable reduction in hemoglobin A1c levels from 10.8 to 9.1% over nine months, particularly benefiting those with poor baseline glycemic control. 18 Likewise, adults using CGM for one-year experienced a decrease in hemoglobin A1c from 9.8% to 8.6%, 19 demonstrating the effectiveness of CGM in improving glucose levels across different ages (Table 3).
Summary of Local Studies Examining the Effectiveness and Safety of CGMs and Insulin Pumps in Saudi Arabia.
(b) Quality of Life.
For younger individuals with T1D (ages 14-21), the transition from the conventional fingerprick method to CGM has significantly improved the mental well-being and treatment satisfaction of these individuals. The WHO-5 Well-Being Index in one study increased from 45.1% to 93.6%, and the Diabetes Treatment Satisfaction Questionnaire (DTSQ) score increased from 14.4 to 31.7, indicating improved psychological health and satisfaction. 27 Likewise, during the COVID-19 lockdown, patients with T1D who had access to telemedicine and CGM were more likely to maintain good glycemic control, highlighting the key role of CGM and telemedicine in providing remote health care services that are essential for continuity of care and quality of life 22 (Table 3).
Type 2 Diabetes on Multiple Daily Injections (MDI)
(a) Glycemic Control:
A 12-week study revealed that patients with T2D on MDI who transitioned to CGM experienced a significant improvement in hemoglobin A1c, with levels dropping from 8.2% to 7.8%. 23 Furthermore, the frequency of hypoglycemic events decreased from an average of 4.4 to 1.2 per month, indicating the role of CGM in stabilizing glucose levels and minimizing adverse events in insulin-dependent T2D patients 23 (Table 3).
(b) Quality of Life:
Patients with T2D on MDI reported increased satisfaction with glucose monitoring using CGM, as reflected in improved DTSQ and Glucose Monitoring Satisfaction Survey scores. These findings highlight the role of CGM in enhancing self-management confidence and improving overall quality of life in people with T2D on MDI 27 (Table 3).
Type 2 Diabetes Not on Insulin Therapy
A longitudinal one-year study demonstrated that people with T2D who are on oral glucose-lowering medications and utilize CGM experienced a sustained reduction in Hemoglobin A1C levels from an average of 8.3% to 7.9%. 26 These findings suggest that CGM is a valuable tool for glycemic management even in patients with T2D who are not requiring intensive insulin therapy (Table 3).
Insulin pumps
People with type 1 diabetes during regular days
Comprehensive analyses comparing insulin pump therapy to MDI in both pediatric and adult populations with T1D in Saudi Arabia revealed the following findings (Table 3):
Patients using insulin pumps reported improved health-related quality of life, better scores in diabetes management satisfaction, fewer treatment-related challenges, and reduced anxiety about glucose control. 29
Studies revealed that insulin pump use offers better glycemic control, with consistently lower A1C levels and less glycemic variability compared to MDI.30,31,34
While insulin pump users had better adherence and overall well-being, they experienced increased concern about disease management. 29
Insulin pump users demonstrated sustained HbA1c improvement over extended period of time. 30
People with type 1 diabetes during Ramadan fasting
Studies on the Medtronic MiniMed 780G AID systems showed safe and effective glycemic control during Ramadan fasting. The use of this system was associated with better glucose control, higher number of days fasted, and reduced hypoglycemia risk compared to MDI and other modalities of treatment.32,33 Al-Sofiani et al 25 showed that 449 users of the AID system in the Gulf region were able to maintain stable glycemic control during Ramadan within the international consensus targets for TIR (average TIR of 70.7%) compared to the months before and after Ramadan. In another prospective Saudi study, users of AID fasted more days of Ramadan (median days the fast was broken because of diabetes was only two days) and had higher TIR during Ramadan (average TIR: 73%) compared to users of other modalities of T1D treatment. 35 These findings provide additional benefits of the AID system in Saudi Arabia and the MENA region.
Other diabetes technology solutions
Other diabetes technology solutions such as Mobile health (mHealth) and telemedicine have gained more traction lately, particularly during the COVID-19 pandemic and afterwards. However, there are very few local studies that examined the efficacy and safety of these digital solutions in people with diabetes.22,36,37 For instance, Al-Sofiani et al, 36 tested a simplified diabetes telemedicine protocol during the COVID-19 pandemic. The telemedicine clinic was well received by people with diabetes and HCPs and was associated with improved glycemic control in those with T1D and T2D, comparable to that seen in those who attended in-person visits. Several other local studies have shown consistent findings.38 -40 Most local studies regarding mHealth were limited to descriptive studies that explore patients’ perception and satisfaction with the use of mHealth.41 -43 For instance, Alanzi et al, 41 identified several barriers to the utilization of mHealth among people with diabetes in Saudi Arabia including limited mHealth expertise, regulatory barriers, and funding challenges.
The local evidence supporting the benefits of insulin pump therapy, CGM, and telemedicine is consistent with that from other countries. The use of CGM in people with T1D significantly reduced HbA1c and improved quality of life, particularly in those with poorly controlled T1D, as reported from the ALERT1, SWITCH, and COMISAIR studies.44 -46 Likewise, when CGM use is combined with telemedicine people with diabetes in Saudi and worldwide reported more satisfaction and better glycemic control over the course of the COVID-19 pandemic and beyond. 47 Consistent with the REPLACE and FLASH-T2DM studies, use of CGM substantially reduced hypoglycemia and improved glucose control in people with T2D who use insulin in Saudi.48,49 Similarly, and consistent with the MOBILE trial findings, a beneficial effect of CGM use was observed among people with T2D who do not use insulin. 50 Furthermore, and consistent with the findings from the STAR 3 and REPOSE studies, use of insulin pump therapy improved glycemic control and patients’ satisfaction in Saudi T1D cohorts.51 -57
There are no local studies that examined the cost-effectiveness of using CGMs, insulin pumps, mHealth, or telemedicine in PWD in Saudi Arabia.
Part 2: Diabetes Technology Report
Patients’ journey to diabetes technology solutions: Navigating the accessibility framework in Saudi Arabia
The journey of people living with diabetes to accessing diabetes technology in Saudi Arabia varies significantly depending on whether they are treated in the public or private health sector. Figure 1 demonstrates the typical pathway to insulin pump therapy for a person with diabetes in Saudi Arabia. As of now, the pathway to acquiring CGMs is not completely streamlined neither in the private nor in the public health sector. Most insurance companies and public clinics develop their own guidelines for who is eligible for CGM use. These guidelines vary significantly because of the lack of national guidance. Access to diabetes technology solutions in Saudi Arabia is generally more favorable among individuals with private insurance who live in urban areas, compared to those who have no insurance or are living in rural regions. In rural and underserved areas, access to diabetes technology remains extremely limited due to disparities in infrastructure, resource availability, and insurance coverage. Moreover, the proportion of people with poorly controlled glucose and complications is higher in those who reside in rural areas of Saudi Arabia compared to those living in urban areas.58,59 Although local data on the utilization of diabetes technology solutions remains limited, the ability to remotely transmit data through the cloud offers a promising avenue for digital health solutions and telehealth to address the current disparities linked to area of residence, limited resources, shortage of specialists, and patients’ social determinants of health. 60 Given the documented regional differences in diabetes and its complications in Saudi Arabia, particularly affecting remote and rural areas, diabetes digital tools to screen for diabetes and its complications present a valuable solution to improve early detection and care.61 -63 Screening for retinopathy remains below target nationwide (58%) due to various reasons including the limited access to ophthalmologists especially in remote areas.64 -66 A systematic review showed high prevalence of diabetic retinopathy in Saudi Arabia which varies widely depending on population characteristics and diagnostic methods and can reach up to 88.1%. 67 Vision-threatening diabetic retinopathy was found in 4% to 17% of patients.68,69 Utilizing telemedicine and diabetes digital tools provides an opportunity to expand the screening programs for diabetes and its complications beyond large cities and tertiary medical centers.
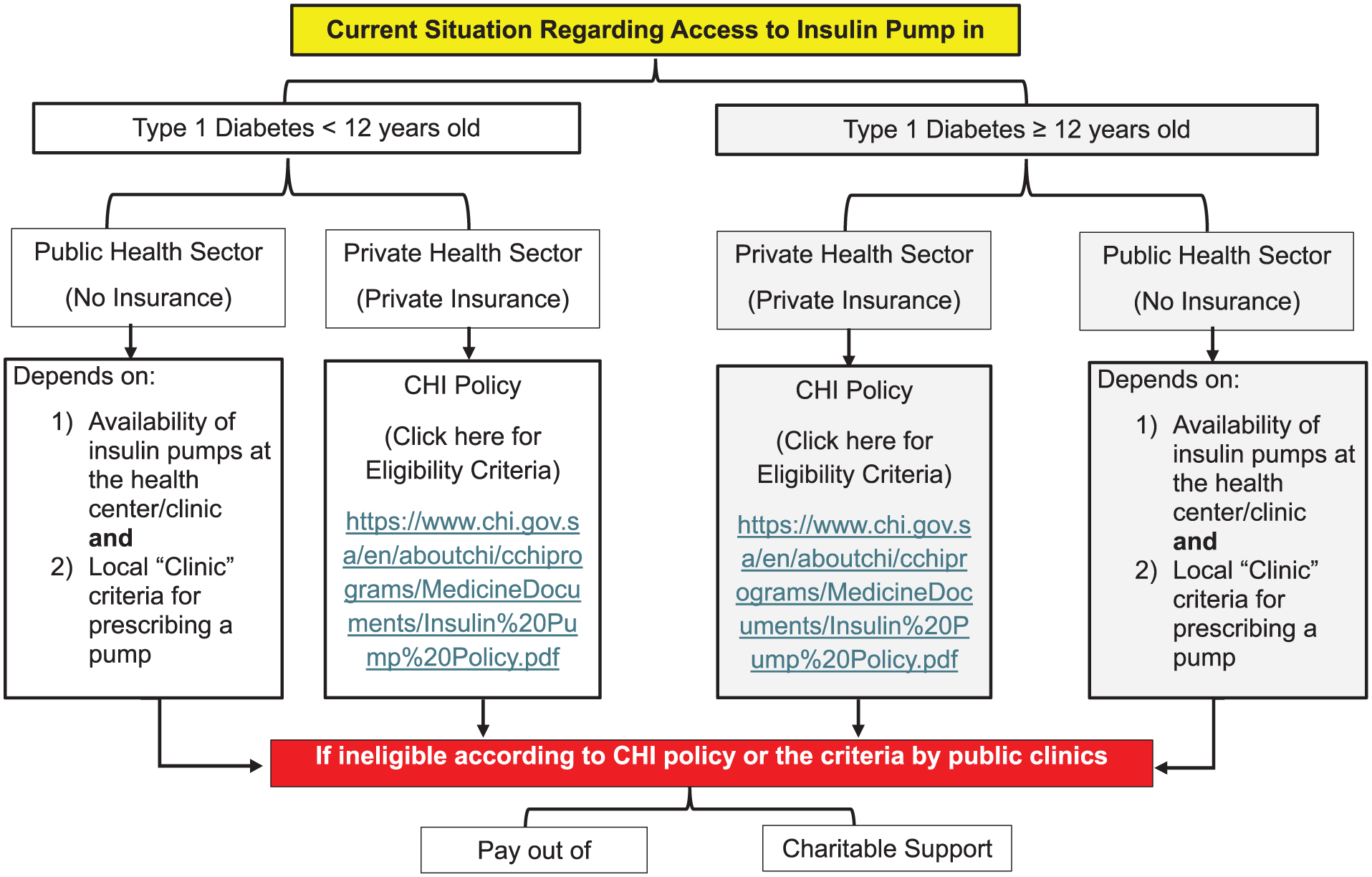
Patients’ journey to insulin pump therapy in Saudi Arabia at the moment.
In this section, we highlight the strengths and limitations of the public and private health sectors in Saudi Arabia and how this may affect the patients’ journey to diabetes technology.
Public sector
As of today, most people with diabetes in Saudi Arabia are cared for by the public health care sector, where they receive free-of-charge health care services, including diabetes medications and some basic digital health solutions. However, they often face longer waiting times, limited access to advanced diabetes technology solutions, and potential bureaucratic hurdles in obtaining advanced innovations such as automated insulin delivery (AID) systems and CGMs. Many diabetes centers in the public sector offer a limited quantity of insulin pumps. Many of these pumps are open-loop pumps (as opposed to AID systems), where the pump is not integrated with a CGM sensor. Local experts estimate the utilization rate of insulin pumps among people living with T1D in Saudi Arabia to be <1% to 3%. Due to the limited availability of insulin pumps in the public clinics and the long waiting list of patients, public clinics often develop very stringent criteria for people with T1D who they would consider eligible for insulin pumps. These criteria often include having a hemoglobin A1C of less than 7 or 7.5% for example, excluding patients with diabetes who live with poorly controlled glucose and may arguably be in more need for insulin pumps.
Likewise, CGMs are available in limited quantities in the public sector and is only dispensed by endocrinologists or family physicians/internists who have an additional training in diabetes. Interruption of CGM supply is a frequent issue that people with diabetes face in the public sector. As it is the case for insulin pumps, public clinics have their own stringent criteria for dispensing CGMs, albeit more relaxed criteria than those used for insulin pump therapy.
Private sector
The private sector, on the other hand, offers quicker access to diabetes medications and advanced diabetes technologies. While insurance coverage in the private sector is often perceived as advantageous, it can sometimes paradoxically become a barrier to accessing diabetes technology devices. Despite the potential for more comprehensive coverage, private insurers may implement stringent criteria and complex approval processes for diabetes digital solutions including CGMs and insulin pumps. These barriers can include restrictive eligibility requirements, caps on coverage for specific devices, or excluding diabetes technology devices from the insurance policy. To regulate this, the Council of Health Insurance (CHI) has issued a policy for insurance coverage of insulin pumps (Figure 2). This policy, however, has several limitations and is expected to be updated in the future.

CHI policy used by insurance companies to reimburse insulin pumps in the private sector.
Likewise, insurance coverage for CGMs is limited in many insurance companies and there is no official guidance by CHI on who among people with diabetes should be reimbursed for CGM. This gap in policy is being realized by CHI and is expected to be addressed in the near future.
For other digital health solutions, the Ministry of Health has published national regulations for the use of telehealth services. 70 Likewise, the National Health Information Center, Saudi Health Council has developed guidelines for telehealth applications in clinical practice. 71 These regulations are nationwide and applicable to the public and private sectors.
Insights from global strategies and best practices
Globally, the coverage and accessibility of diabetes digital solutions such as CGMs and insulin pumps remain a complex issue. While these technologies have proven to significantly improve diabetes management and quality of life for many patients, their adoption is often hindered by factors such as high costs, limited health care budgets, varying reimbursement policies, and limited specialized health care professionals and diabetes centers. Many countries are grappling with the challenge of balancing the intermediate- to long-term health benefits and cost savings of these technologies against their immediate financial impact on health care systems.72 -74 As a result, eligibility criteria for coverage often prioritize specific groups of patients, such as those with poorly controlled diabetes, recurrent acute or chronic complications, etc.75,76 The disparity in access to diabetes technology solutions not only exists between countries but also within countries, reflecting broader socioeconomic inequalities in health care access. As diabetes prevalence continues to rise globally, policymakers and health care systems in developed countries started to realize the need to adopt a strategy of “paying more upfront for future savings.” As a result, insurance coverage for these technologies and public sector support have been recently broaden in many countries to make these life-changing technologies more widely available to those who need them most. Supplemental Table 1 list the coverage and reimbursement criteria for CGMs and insulin pumps from select countries.77 -82
Conclusions (the Roadmap to Improving Accessibility to Diabetes Digital Health in Saudi Arabia)
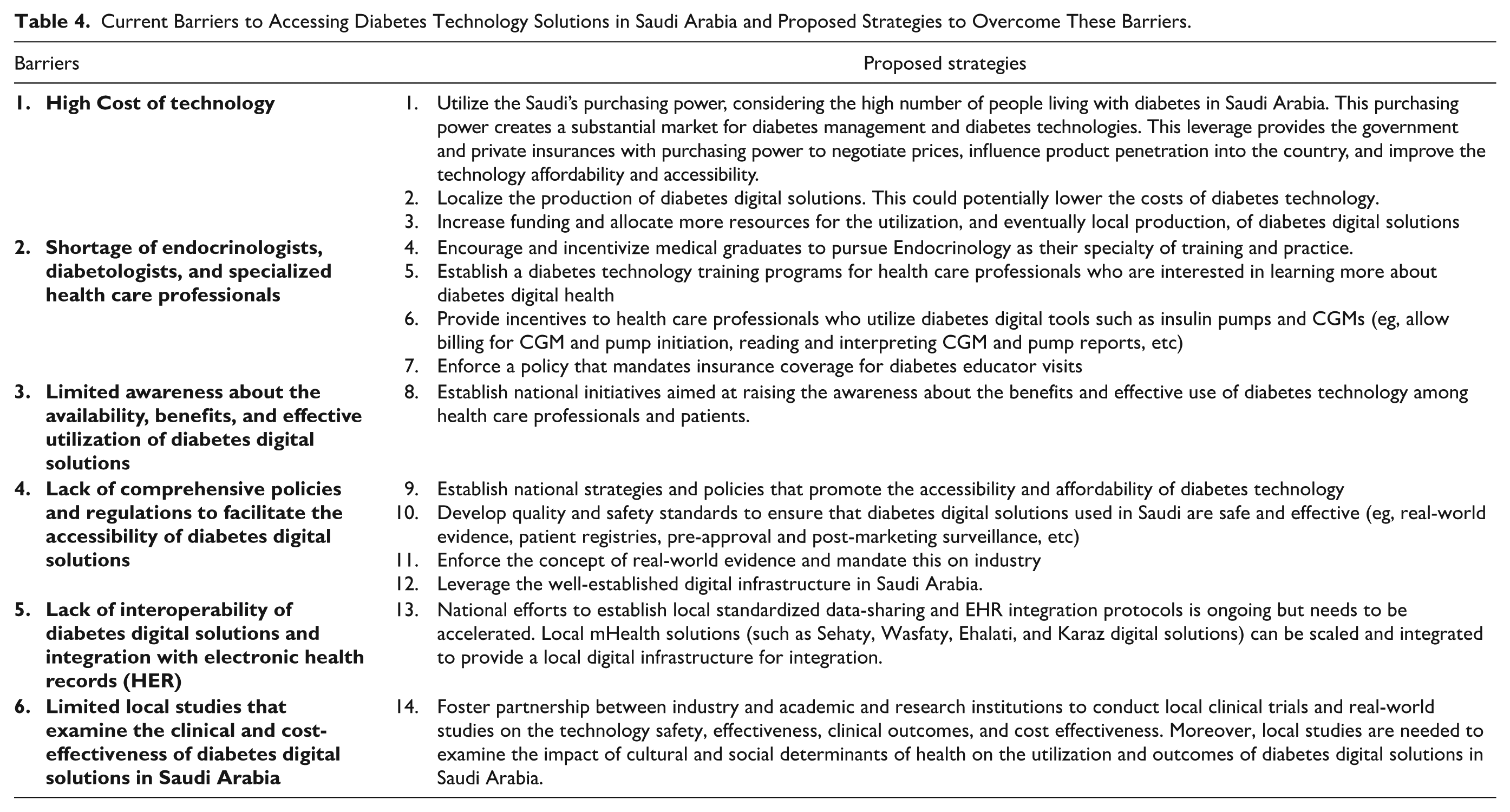
Both the public and private health care sectors in Saudi Arabia have made strides in improving diabetes care, but challenges remain in ensuring equitable access to diabetes technology across the country. The public sector’s strength lies in its broad reach and affordability, while the private sector excels in offering rapid access to innovations. Bridging the gap between these two sectors, improving coordination, and addressing barriers to access to diabetes technology solutions in both sectors is crucial in improving the overall quality of diabetes care and reducing the health and economic burden of diabetes on individuals and the overall health care system. The current disconnects between the two health sectors and gaps in their policies not only frustrate patients and health care professionals but also undermine the potential health benefits and long-term cost savings that these digital solutions can offer. We believe the Saudi health care system needs to address the six barriers impeding access to digital health solutions for diabetes management and implement the 14 initiatives proposed in Table 4 according to the roadmap and timeline highlighted in Figure 3.
Current Barriers to Accessing Diabetes Technology Solutions in Saudi Arabia and Proposed Strategies to Overcome These Barriers.
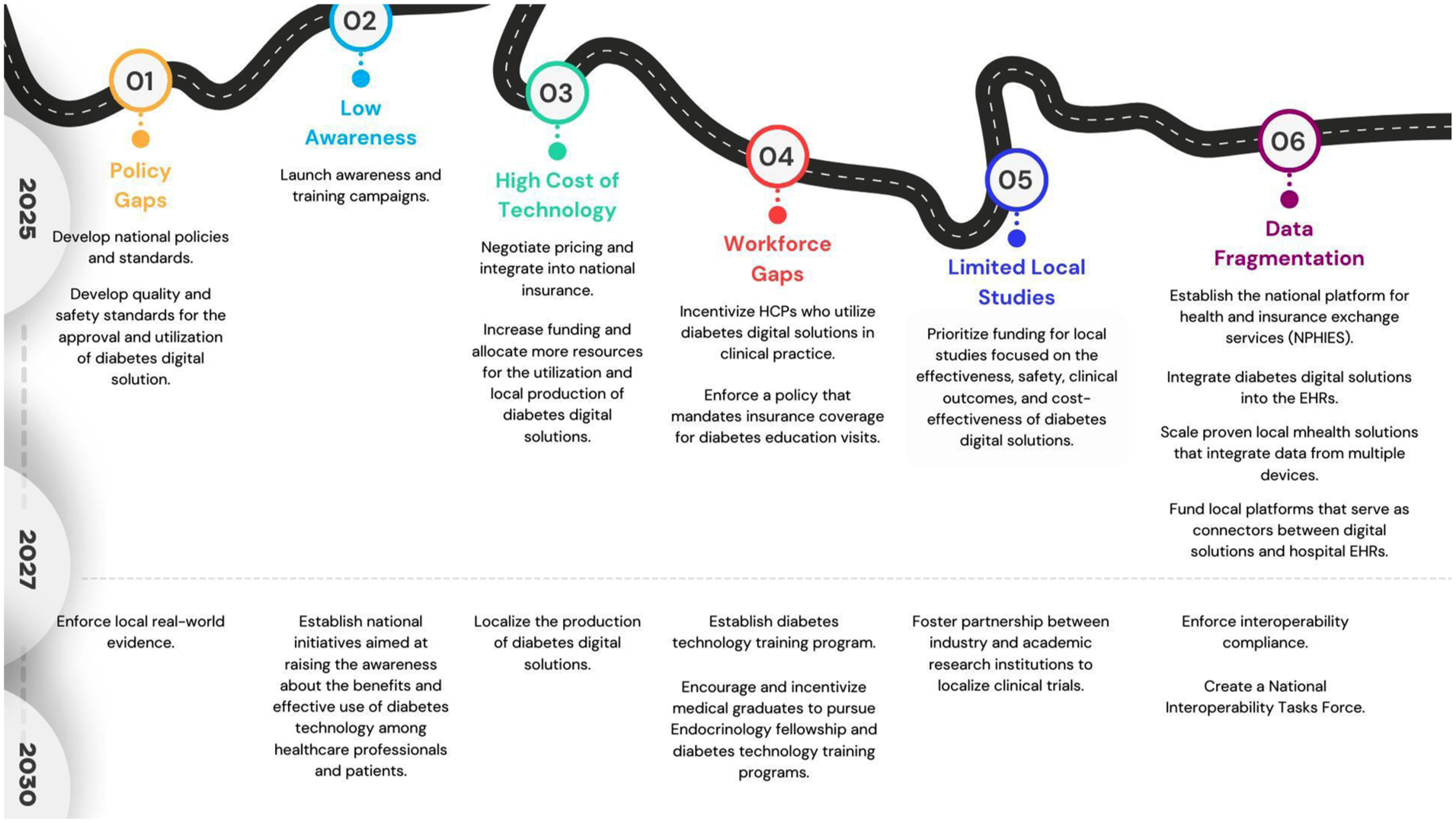
Roadmap to improving accessibility to diabetes technology solutions in Saudi Arabia.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251370756 – Supplemental material for Diabetes Technology in Saudi Arabia: Current Status and Future Directions
Supplemental material, sj-docx-1-dst-10.1177_19322968251370756 for Diabetes Technology in Saudi Arabia: Current Status and Future Directions by Mohammed E. Al-Sofiani, Abdulhalim M. Al-Murashi, Nada A. Al-Agil, Azzam O. Al-Othman, Shahad Y. Al-Faris and Ahmed M. Al-Enzi in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
We extend our sincere gratitude to the Saudi National Diabetes Center (SNDC) team for giving us the opportunity to contribute to advancing diabetes digital health in Saudi Arabia. Special acknowledgment goes to the dedicated health care professionals, researchers, and people living with diabetes in Saudi Arabia. We also appreciate the editorial support by Dr Sahar Alharthi and administrative support by the SNDC staff: Ms Malak Al-Ateeq and Ms Saja Alhosan who facilitated the smooth coordination between different team members, departments, and stakeholders.
Abbreviations
AID, automated insulin pump; CGM, continuous glucose monitoring; CHI, Council of Health Insurance; CSII, continuous subcutaneous insulin infusion; DTSQ, Diabetes Treatment Satisfaction Questionnaire; EBP, essential benefit package; GMI, glycemic management indicators; HbA1c, hemoglobin A1C; HCP, health care professional; HTA, health technology assessments; IQ, intelligence quotient; MDI, multiple daily injection; MOH, Ministry of Health; NHS, National Health Services; NICE, National Institute for Health and Care Excellence; SFDA, Saudi Food and Drug Authority; T1D, type 1 diabetes; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MEA has served on an advisory panel for Abbott, Medtronic, Insulet, VitalAire, and Sanofi; has received honoraria for speaking and consultancy from Abbott, Eli Lilly, Medtronic, Novo Nordisk, Sanofi, and VitalAire; and has received research fund from Medtronic and Sanofi. All other authors have no conflicts of interest to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
