Abstract
Background:
Type 2 diabetes (T2D) disproportionately affects youth with public insurance of minority and lower socioeconomic status backgrounds. We aimed to determine feasibility of CGM use in this understudied population.
Methods:
We enrolled youth <20 years old with T2D, provided or prescribed intermittent scanned CGM, and followed established clinic workflows with six data collection visits over 12-months. CGM use was measured by % wear time per two-week period (>75% wear-time as goal) from downloaded report prior to clinic visit. Exploratory outcomes included: 14-day CGM wear time in range (TIR: % time spent between 70 and 180 mg/dl), HbA1c, and patient-reported outcomes (PROs) collected from youth and parents.
Results:
We enrolled 30 youth (age 15.1 years [SD 2.48]; HbA1c 10.2%, range: 6.5%-15.5%), 46.7% female, 90% Hispanic. At baseline, 37% previously used CGM and 53% lacked glucometer data. CGM use was 50% at three months and 23% at 12 months. CGM wear time decreased by 6.4 days per two weeks by 12 months. Mean HbA1c was 9.8% at 12 months and median TIR decreased from 71% to 42%. Parents and youth had moderate-to-positive attitudes about diabetes technology. Youth endorsed fair levels of global health; and youth and parents endorsed fair general and diabetes-related health-related quality of life.
Conclusions:
Strategies for sustained CGM use in youth with T2D may differ from adults with T2D or youth with type 1 diabetes. Additional studies are needed to evaluate facilitators and barriers of sustained CGM use to optimize CGM use in youth with T2D.
Clinicaltrials.gov registration:
NCT05074667.
Introduction
Type 2 diabetes (T2D) disproportionately affects children of racial and minoritized ethnic groups and lower socioeconomic status (SES), with higher rates of cardiovascular and renal co-morbidities in youth with T2D compared with youth with type 1 diabetes (T1D) and similar duration of disease.1-5 The RISE Consortium and the Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) Study reported more rapid beta-cell damage, disease progression, and diabetes-related complications in youth compared with adults, highlighting the need for improved clinical management and treatment for these patients.4,6,7 One year post-diagnosis, a majority of youth with T2D do not meet HbA1c targets8-10 and over 60% of young adults with childhood-onset T2D develop at least one macrovascular or microvascular complication by their mid-20s. 4
Treatment of T2D in youth includes lifestyle, diet management, and medications, including insulin, frequently in a complex psychosocial and cultural environment that challenges optimal diabetes management. Data for CGM in adults with T2D demonstrates efficacy and cost-effectiveness.11-13 Data from the SEARCH for Diabetes in Youth study found that over 50% of youth with T2D had <3 self-monitoring blood glucose (SMBG) checks per day, and almost 25% had <1 daily. 14 Diabetes distress is well recognized in T2D and impacts both glycemic outcomes and diabetes self-care behaviors. 15
In contrast, CGM is the standard of care for youth with T1D, improves glycemic control and quality of life,16,17 and initiation is feasible and well-accepted. 18 Significant improvements in HbA1c have been shown in youth with T1D with public insurance with uninterrupted CGM use.19,20 Studies evaluating use of CGM in youth with T2D demonstrate acceptability and changes in behavior; however, these are studies of short duration.21,22 Evaluation of CGM use in youth with T2D, with specific attention to factors that influence uptake and sustained use of diabetes technology, is needed for this population to benefit from CGM.
This pilot expands our existing program of equitable CGM access to youth with T1D, which has led to an improvement in HbA1c of 1.1% over one year, positive feedback from patients and families, and similar benefits across insurance types and ethnic and racial groups.23,24 Our 12-month pilot study aimed to evaluate the feasibility of CGM start and continuation in youth with T2D and public insurance and describe glucose metrics and patient-reported outcomes (PROs). We hypothesized that CGM wear time would increase over time. Based on the completion of PRO surveys, we expected that diabetes-specific distress would decline and overall well-being/quality of life would improve. In addition, we aimed to assess the clinical outcomes (HbA1c, time-in-range [70-180 mg/dL], time in hypoglycemia [<70 mg/dL]) of study participants and hypothesized that the use of CGM would improve HbA1c and other CGM metrics. The goal is to provide data to inform future studies using CGM in youth with T2D.
Methods
Participants and Procedure
We enrolled 30 youth with T2D in a 12-month pilot study. The study enrolled youth (1) on public insurance; (2) between ages four and 19.99 years; (3) with a diagnosis of T2D (diagnosed by pediatric endocrinologist per American Diabetes Association (ADA) standards and diabetes autoantibody negativity) on insulin and noninsulin medications followed in the pediatric endocrinology clinic at Stanford Children’s Health; and (4) HbA1c >6.5% at enrollment. In our clinic population, youth with T2D have a mean HbA1c of 8.6% with 35% at goal HbA1c of <7% (n = 311 HbA1c values), indicating that most youth with T2D were not meeting this therapy goal. 9 Reflective of the national data, a significant portion of our clinic T2D patient population (n = 131) was publicly insured (77%), of a racial minoritized group (73%, specifically 66% are Latinx), and had Spanish-speaking parents or guardians (56%), all groups which traditionally have lower rates of diabetes technology use. 25 Additional inclusion criteria were (5) interest in starting CGM and (6) access to a mobile device compatible with the CGM application or willingness to use the provided CGM receiver. Informed consent and assent (in the participant and guardian’s preferred language) was approved by Stanford University’s Institutional Review Board. Intermittent scanned CGM (Freestyle Libre 2, Abbott) was provided to all participants, if not covered by insurance, for the duration of the study to minimize gaps in supply access. The type of CGM was selected to align with insurance coverage and ensure consistent device use throughout the study.
Study participants were followed during routine diabetes clinic visits. Following CGM initiation by a Certified Diabetes Care and Education Specialist, youth were seen two to four weeks later for CGM follow-up, and then per clinic standard of care, every three months thereafter with their diabetes provider for the 12 months of study duration. The study team contacted the participant every six weeks by phone or during a clinic visit to assess barriers to CGM use and visit attendance. At each visit, data on 14-day CGM wear time and glucose metrics were collected from LibreView, and additional CGM support was provided per routine clinical care. CGM data from LibreView were collected for each participant regardless of attendance at clinic visits during the specified clinic visit window.
Review of study visit documentation through the electronic health record was performed. Patient-reported outcomes and quality of life (QoL) measures were administered to participants and guardians at baseline, three months, and 12 months, either during a study visit or participants were contacted directly to complete PROs. Study participant data and PROs were collected in REDCap. PROs were completed in their preferred language (English or Spanish). At each study visit and phone calls between study visits, study and clinic staff noted participant experiences with CGM and reasons for CGM nonuse at the time of the visit, if applicable. A coding team (NS and MT) used content analysis to categorize this qualitative content and identify most common reasons for CGM nonuse at each visit. 26
PRO and QoL Measures
Diabetes-related quality of life
Participants completed the 33-item PedsQL Diabetes Module 3.2, which assesses five domains of diabetes-related quality of life (QoL): diabetes symptoms, worry, treatment I, treatment II, and communication. Responses are on a 1-to-5 point Likert scale and converted to 0, 25, 50, 75, and 100. The total score is a mean of all items (range 0-100). 27
Problem areas in diabetes—teen and parent versions
To assess diabetes distress in youth in their parents, adolescents completed the 14-item PAID-T; and parents of adolescents completed the 15-item P-PAID-T. For all PAID measures, items are on a 6-point Likert scale with higher scores indicating higher diabetes-specific emotional distress.28-30 For the 14-item PAID-T, a score greater than or equal to 44 indicates high distress, while a score of 54 or greater indicates high distress for the P-PAID-T.
Diabetes technology attitudes
Attitudes toward diabetes technology were assessed using the five-item Diabetes Technology Attitudes Scale (DTAS). 31 Each item is on a 5-point Likert scale. An example item is: “Diabetes technology has made my life better.” Higher scores indicate more positive attitudes toward diabetes technology.
Global health
Participants completed the seven-item PROMIS Pediatric Global Health Scale (PGH-7), which measures physical, mental, and social health.32,33 Items are on a 5-point Likert scale. The total score is converted to a T score with higher scores indicating better global health and a score of 50 reflecting an average score.
Based on the evidence in the PROs literature and our clinical experiences, we have developed an algorithm for responding to PROs screen scores that were outside of the normative range, leading to a referral to a social worker and/or diabetes psychologist. 34 Treatment followed ADA Position Statement on Psychosocial Care of People with Diabetes guidelines. 35
Data Analysis
We generated descriptive statistics for all participants included in the study at baseline and participants having CGM data at follow-up time points. The descriptive statistics for continuous variables were presented as mean and standard deviation or median and interquartile range depending on the distribution of variables, while for categorical variables, frequency and percentages were reported. To address the feasibility of CGM use in T2D, we estimated the proportion of participants with sustained CGM use. Our feasibility analysis included all participants. We assessed sustained CGM use in the last 14 days at each follow-up time point and across all points via linear mixed effects model with a patient-specific random intercept to account for patient-level correlation. This model used an indicator of CGM wear time in 14 days at each time point as the dependent variable. Independent variables in the model are follow-up time points, duration of T2D diagnosis, and T2D treatments (insulin vs others). All independent variables are mean-centered. The time point variable included in the model allowed us to evaluate whether CGM wear time in 14 days varied over time, and the significance of the timepoint variable on the CGM wear time in 14 days was conducted using a likelihood ratio test. The models were analyzed using follow-up time points alone, and then the duration of T2D diagnosis and T2D treatments were added individually in separate models. Additionally, box plots were used to visualize the distribution of CGM wear time in 14 days at each follow-up time point. For youth and parent PRO measures, medians and interquartile ranges were calculated.
Clinical notes on reasons for CGM nonuse were analyzed using content analysis. All statistical analysis was performed using R v4.3.2 (R Foundation for Statistical Computing, Vienna, Austria). Qualitative analysis of participant free-text survey comments was conducted using NVivo 14 (Lumivero, 2023) qualitative data software.
Results
Participants
We approached 61 youth meeting inclusion criteria and enrolled 30 youth with T2D (Figure 1). 36
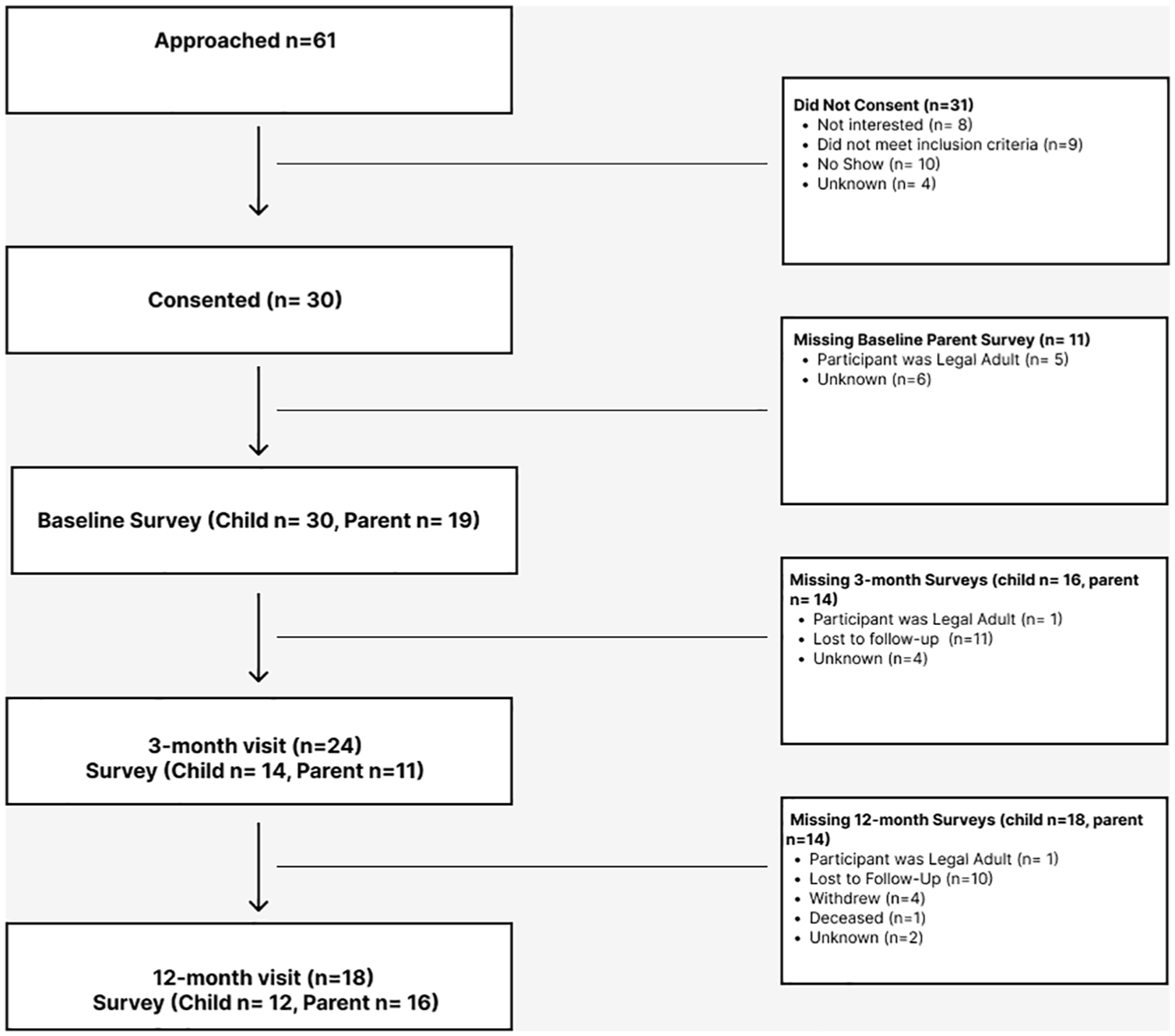
Consort diagram of participants screened, recruited, and followed throughout the study: consolidated standards of reporting trials for the pilot study and completion of youth and parent PRO and QoL measures.
At baseline, mean age was 15.2 years, mean HbA1c was 10.2% (range 6.5%-15.5%), 46.7% identified as female, 90% as Hispanic and 63.3% identified Spanish as their guardian’s primary language. At study enrollment, 86.7% of participants used insulin. Prior to study enrollment 37% of participants had intermittent CGM use. At study enrollment, 53% did not have SMBG data within two weeks of the visit. At three months, 24 out of 30 youth attended their visits; however, only 18 out of 26 youth attended their 12-month visit (Table 1).
Characteristics of All Youth at Baseline and Youth With CGM Data at Each Study Time Point.
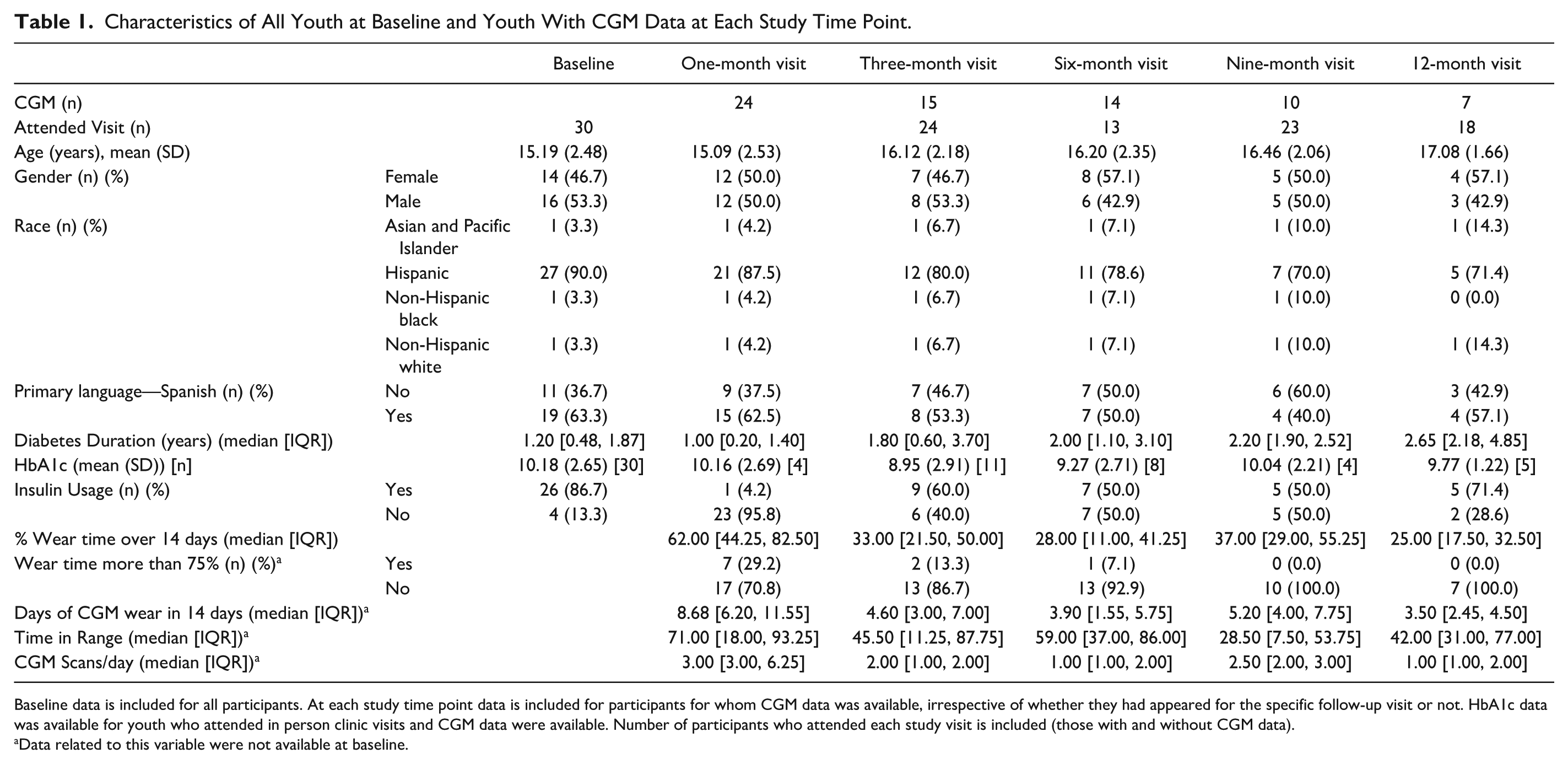
Baseline data is included for all participants. At each study time point data is included for participants for whom CGM data was available, irrespective of whether they had appeared for the specific follow-up visit or not. HbA1c data was available for youth who attended in person clinic visits and CGM data were available. Number of participants who attended each study visit is included (those with and without CGM data).
Data related to this variable were not available at baseline.
CGM Use and TIR
For youth using CGM (n = 24/30), median percent wear time over 14 days at the one-month visit was 62% (interquartile range [IQR] 44.3-82.5), with 29.2% of participants having a 14-day wear time over 75%. At the three-month visit, CGM data were available for 15 out of 30 participants, with a median 14-day wear time of 33% (IQR 21.5%-50%) and 13.3% with wear time >75%. At 12-months, seven out of 30 had CGM data available with a median wear time of 25% (IQR 17.5-32.5) and no participants had > 75% wear time. Continuous glucose monitor wear time over 14 days for each participant throughout the study is shown in Figure 2. Participant median CGM scans per day decreased from three to one scan per day and median TIR decreased from 71% to 42% throughout the study for youth with data at each time point. Mean HbA1c was lower at three months (8.95%) compared with baseline (10.16%) in participants who wore CGM; however, this was not sustained.
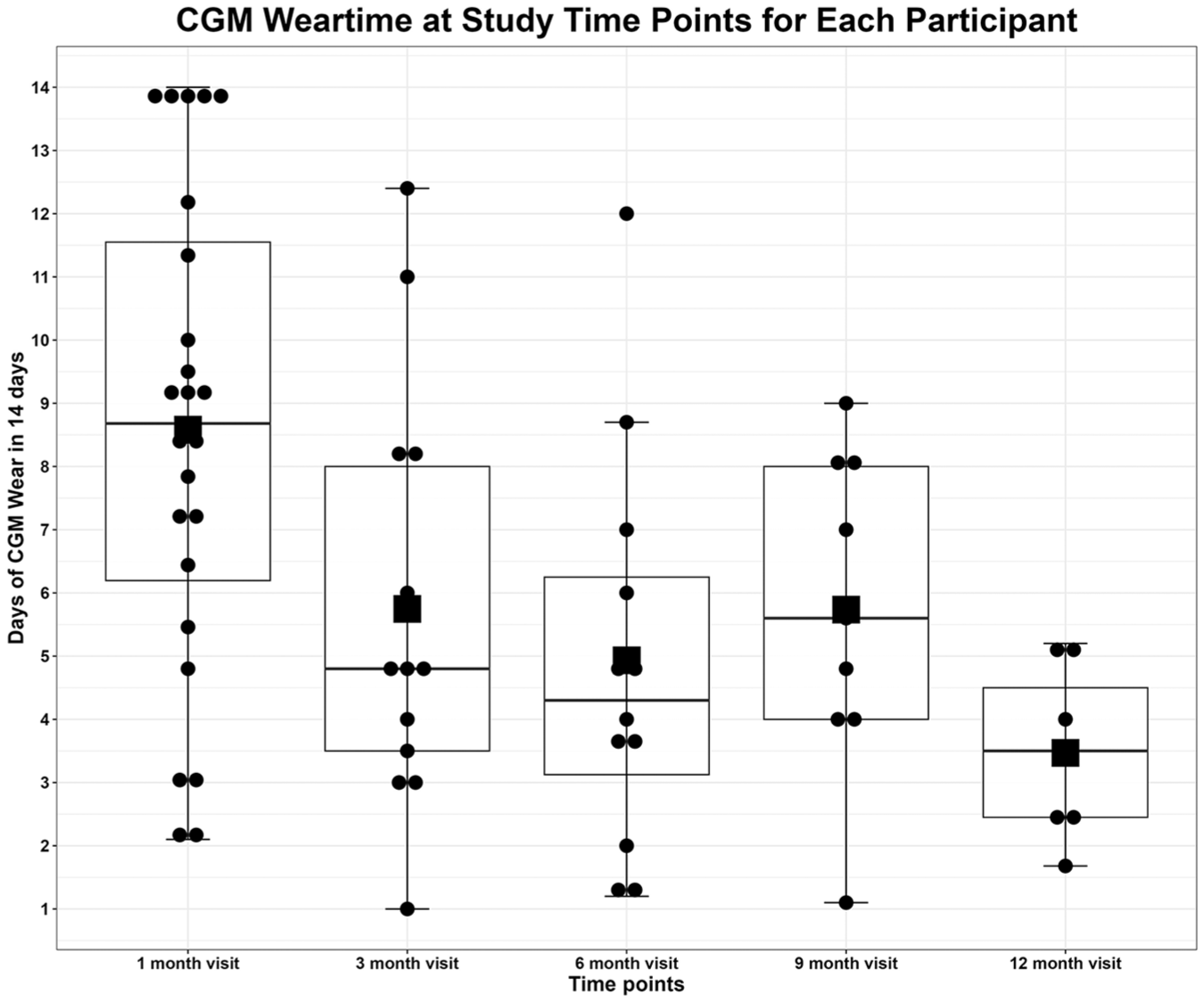
CGM 14-day wear time at each study time point. Black dot: study participant; Black line = median, Upper line of box = Upper quartile Q3, Lower line of box = Lower quartile Q1, Upper line = Maximum value, Lower line = Minimum value, Black Square = mean Note: at 12-month visit there are seven data points, with one data point beneath the black square indicating the mean.
CGM wear time over 14 days was 3.72 days lower (95% CI –5.77 to –1.66) in month 3 compared with month 1, accounting for within-subject correlation. CGM wear time was not associated with diabetes duration or insulin use. CGM 14-day wear time was 2.13 days lower (95% CI [−4.88, 0.62]) in the month 6 visit compared with month 1 visit after controlling for diabetes duration, accounting for within-subject correlation. CGM 14-day wear time was 6.4 days lower (95% CI [−9.36, –3.44]) in month 12 visit compared with month 1 visit after controlling for insulin use, accounting for within-subject correlation. Over the 12-month duration of the study there was a statistically significant decrease in 14-day CGM wear time based on fixed effects analysis (Table 2).
Changes in the CGM Wear Time (in 14 Days—Measured Continuously) at Different Follow-Up Visits.
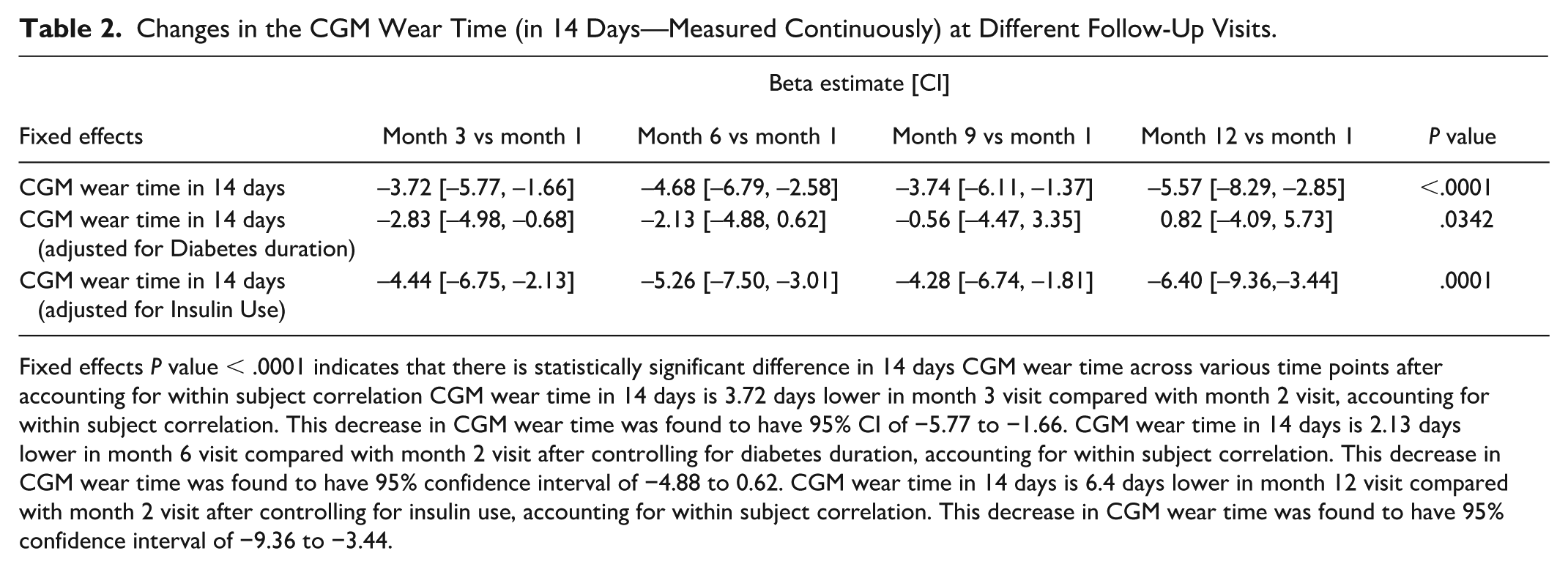
Fixed effects P value < .0001 indicates that there is statistically significant difference in 14 days CGM wear time across various time points after accounting for within subject correlation CGM wear time in 14 days is 3.72 days lower in month 3 visit compared with month 2 visit, accounting for within subject correlation. This decrease in CGM wear time was found to have 95% CI of −5.77 to −1.66. CGM wear time in 14 days is 2.13 days lower in month 6 visit compared with month 2 visit after controlling for diabetes duration, accounting for within subject correlation. This decrease in CGM wear time was found to have 95% confidence interval of −4.88 to 0.62. CGM wear time in 14 days is 6.4 days lower in month 12 visit compared with month 2 visit after controlling for insulin use, accounting for within subject correlation. This decrease in CGM wear time was found to have 95% confidence interval of −9.36 to −3.44.
Patient-Reported Outcomes
Median values and IQRs for youth and parent PROs at baseline, month 3, and month 12 are presented in Table 3. Teens endorsed elevated diabetes distress at all time points, while parent distress was elevated at baseline and three-month time points. Parents and youth had moderate-to-positive attitudes about diabetes technology. Youth endorsed fair levels of global health (>1 standard deviation below the average score of 50). Youth and their parents endorsed fair diabetes-related QoL.
PROs for Youth and Parents at Different Time Points.
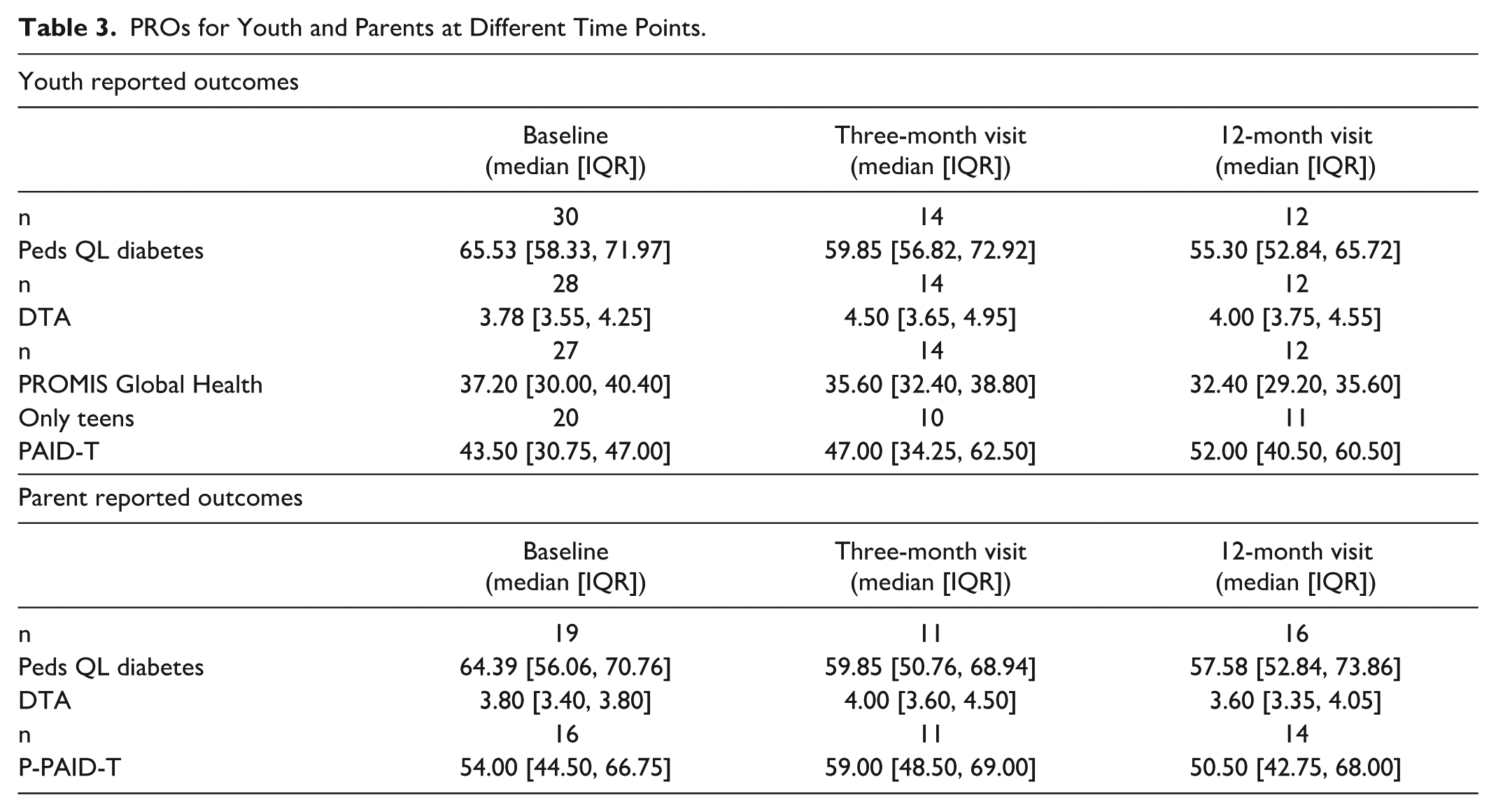
Correlations between PROs and CGM wear time at month 3 showed that the three-month PedsQL diabetes score was positively associated with CGM wear time (P = .0496), while other PROs did not demonstrate significant correlations with CGM wear time. 37
Qualitative Results: Documenting Reasons for CGM Nonuse
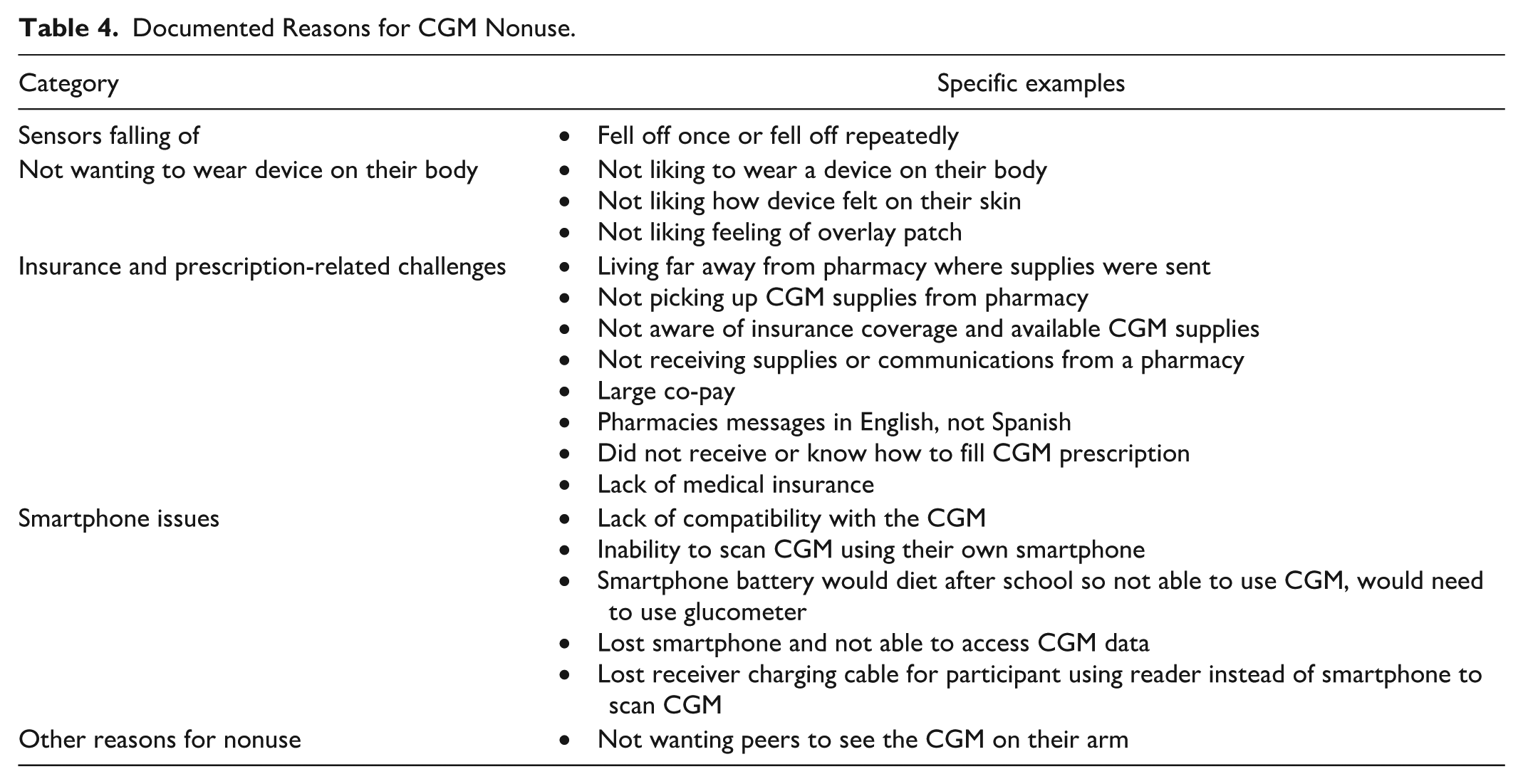
Reasons for CGM nonuse were grouped into the following categories: sensors falling off, insurance and prescription-related challenges, smartphone issues, not wanting to wear device on their body, and other (Table 4). The most frequently noted reason for not using CGM was that the sensor either fell off once or repeatedly, leading to nonuse and discontinuation for some. Participants who experienced sensors falling off were provided support, including receiving adhesive supplies from the clinic to assist with sensor adhesive issues and guidance on how to obtain additional adhesive supplies. Some participants/guardians received guidance on how to contact the manufacturer for replacement CGMs. While public insurance covered CGM for most of the study duration, several participants encountered logistical and insurance issues with obtaining supplies from the pharmacy. Several issues with using smartphones arose as reasons for nonuse or usability issues.
Documented Reasons for CGM Nonuse.
Discussion
Our study demonstrates that while T2D youth initially consistently used CGM and had moderate-to-positive attitudes toward diabetes technology, wear time declined substantially over 12 months—from 62% at one month to 25% at 12 months. Sustained use was not achieved in over half of participants. HbA1c was lower at six months in CGM users, though not sustained. Multiple factors likely contributed to decreased wear time, warranting further study and indicating tailored approaches are required for this population.
Demographics of our study population reflected our clinic population, highlighting real-world applications of our findings. Participants had higher mean HbA1c than our general T2D clinic population and above target HbA1c for youth with T2D reflecting a subgroup of youth with T2D who need additional interventions to achieve glycemic targets. 9 Our clinic population had a high rate of insulin use, consistent with clinical practice at the time of the study. As GLP-1RA and SGLT-2 inhibitors are increasing in use for treatment of T2D in youth, the role of CGM may further evolve toward using glucose data to guide health behaviors including medication-taking, diet modifications, and lifestyle changes.
Strengths of the study include integration into established clinic workflows and clinical visits with primary diabetes providers to decrease participation burden, language-concordant support, and CGM supply provision if not covered by insurance as uninterrupted CGM access in youth is associated with decrease in HbA1c. 19 As with other studies, our study showed initial high rates of CGM use at one month (24/30, 80%); however, this high rate of CGM use was not sustained throughout the study.21,22 Lack of sustained CGM use, clinic visit attendance, and survey completion did not permit more detailed analysis or comparison of CGM use data and glucose metrics, including HbA1c, which is a significant limitation. Due to small cohort size we cannot draw conclusions about the association between CGM wear and PRO or QoL metrics in youth with T2D. Future research should explore ways to increase CGM wear time, clinic engagement and PROs in relation to CGM uptake in this population with a larger sample size over time. 21 ,38-40
Unlike adult T2D studies showing 87% CGM use at eight months and HbA1c improvements regardless of insulin use, our findings reflect lower sustained use in youth with T2D.41,42 Our study utilized an intermittently scanned CGM device, requiring user engagement which likely contributed to low data capture; real-time CGM or scheduled intermittent CGM may improve engagement in CGM use and outcomes. Further integration of CGM data into patient treatment plans may increase the benefit each adolescent may see in CGM use for their own care, especially for those youth who are treated with noninsulin medications. Studies have also shown use of CGM may lead to behavioral modification in adults with T2D leading to improvement in glycemic control. 43 Further studies using real-time CGM systems in youth with T2D are warranted.
This pilot adds novel PROs data around CGM use in youth with T2D, highlighting elevated diabetes distress and only fair scores on global health and diabetes-related QoL. Greater QoL was linked to higher CGM wear time, suggesting future areas of research to understand sources of lower self-reported health and QoL, and target interventions to increase QoL to support CGM use.
Our pilot study is one of the first to report on barriers to CGM use in this population, which can help inform future studies to evaluate sustained CGM use. Analysis of qualitative reasons for CGM nonuse showed sensors falling off as the main reason for nonuse initially but declined over time. Persistent issues, including insurance logistics, prescription access, and language barriers, identify an area of need to support the sustained use of CGM. Our data adds to the literature in this area to support CGM use in youth with T2D. (Peyyety, Zupa and Hewitt, 2023)(Patel, Sala and Macheret, 2025)38,44
Conclusions
Strategies for CGM use in youth with T2D may differ from adults with T2D. Data from this pilot and feasibility trial fills a gap in the literature for CGM use in youth with T2D. The findings from this trial can provide needed data to impact CGM use in youth with T2D and pave the way for future studies evaluating sustained use of CGM technology in this population. Youth with T2D have a different clinical course and social determinants of health considerations compared with adults with T2D or youth with T1D. As prior studies have identified and addressed barriers and facilitators for sustained CGM use in youth with T1D to optimize diabetes care, additional studies are needed to learn the optimal way to help youth with T2D sustain CGM use to achieve improved diabetes-related outcomes. The higher rates of diabetes-related complications in young onset T2D and use of noninsulin medications necessitate further studies to understand how CGM technology use can be supported to improve long-term glycemic control.
Footnotes
Acknowledgements
We thank all the youth and their guardians that participated in the study, and the members of our research team including research coordinators and clinical staff that supported the study and provided clinical care to study participants. PedsQL™ Diabetes Module 3.2 contact information and permission to use: Mapi Research Trust, Lyon, France, ![]() .
.
Abbreviations
CGM, continuous glucose monitor; PROs, patient-reported outcomes; QoL, quality of life; SES, socioeconomic status; SMBG, self-monitoring blood glucose; T1D, type 1 diabetes; T2D, type 2 diabetes; TODAY, treatment options for type 2 diabetes in adolescents and youth.
Author Contributions
SS: conceptualization (lead); writing—original draft (equal); writing—review and editing (supporting). MLT: data curation (supporting); formal analysis (equal); writing-original draft (supporting); writing—review and editing (equal). AL: Project administration; visualization (supporting). NGLS: data curation (supporting); formal analysis (supporting); writing—review and editing (supporting). HD: formal analysis (lead); methodology (lead); visualization (lead); writing—review and editing (supporting). SH: data curation (lead); formal analysis (supporting). FKB: writing—review and editing (supporting), manuscript preparation. KKH: conceptualization (supporting); methodology (supporting); writing—review and editing (supporting). DMM: conceptualization (supporting); writing—review and editing (lead).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SS, MLT, HD, SH, NGLS, FKB, and AL do not have any disclosures. KKH has received consulting fees from Sanofi and Havas Health and is on the advisory board for MannKind. KKH has also received gift funding from Embecta for investigator-initiated research. DMM has had research support from the NIH, JDRF, NSF, and the Helmsley Charitable Trust and his institution has had research support from Medtronic, Dexcom, Insulet, Bigfoot Biomedical, Tandem, and Roche. DMM has consulted for Abbott, Aditxt, the Helmsley Charitable Trust, Lifescan, Mannkind, Sanofi, Novo Nordisk, Eli Lilly, Medtronic, Insulet, Dompe, Biospex, Provention Bio, Kriya, Enable Biosciences, and Bayer.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by pilot funding from the Stanford Diabetes Research Center (P30DK116074) and the Stanford Maternal & Child Research Institute (MCHRI) Social Injustice and Health Disparities in Maternal and Child Health Pilot Award. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. The Stanford REDCap platform provided additional support. The Stanford REDCap platform (![]() ) is developed and operated by Stanford Medicine Research Technology. The REDCap platform services at Stanford are subsidized by (a) Stanford School of Medicine Research Office, and (b) the National Center for Research Resources and the National Center for Advancing Translational Sciences, NIH, through grant UL1 TR003142.
) is developed and operated by Stanford Medicine Research Technology. The REDCap platform services at Stanford are subsidized by (a) Stanford School of Medicine Research Office, and (b) the National Center for Research Resources and the National Center for Advancing Translational Sciences, NIH, through grant UL1 TR003142.
