Abstract
Background:
Diabetic foot ulcers (DFUs) affect 19% to 34% of individuals with diabetes during their lifetime and account for over one million nontraumatic lower-limb amputations annually. Standard care often fails to detect early, subclinical changes, leading to delayed intervention and high mortality rates. This review examines how artificial intelligence (AI) and machine learning (ML) can extract complex patterns from diverse data modalities to advance DFU care.
Methods:
We examined AI/ML applications in DFU care across four domains: diagnosis (automated image and thermogram classification, biomechanical risk stratification), treatment optimization (AI-driven offloading prescriptions, tele-rehabilitation, molecularly informed wound care), prognosis (neural network and random forest models for risk assessment), and novel strategy development (generative AI, transcriptomic target discovery, wearable digital biomarkers).
Results:
Artificial intelligence/ML methodologies have demonstrated promising results in DFU image and thermogram analysis, with reported accuracies ranging from 81-97% across different studies. Biomechanical ML models show potential for dynamic risk stratification, and prognostic models achieve moderate performance with area under the curve values around 0.74-0.82. Generative AI approaches have shown promise for data augmentation, improving segmentation performance in limited datasets.
Conclusion:
Despite promising advances, several challenges impede clinical translation, including data standardization, model explainability, regulatory compliance, clinical workflow integration, prospective validation, and equitable implementation. Collaborative efforts among clinicians, data scientists, regulators, and patients are essential to translate AI-driven innovations into routine DFU management, potentially reducing amputations and improving outcomes for this global health burden.
Keywords
Introduction
Diabetic foot ulcers (DFUs) are a major global health burden, affecting approximately 19-34% of individuals with diabetes during their lifetime and accounting for over one million nontraumatic lower-limb amputations annually.1,2 Standard care—visual inspection, monofilament neuropathy testing, graduated offloading—often fails to detect early, subclinical changes in skin perfusion or pressure distribution, leading to delayed intervention, high recurrence, and five-year mortality rates exceeding 50% in some cohorts. 3
The pathophysiology of DFUs is multifactorial: sensory and motor neuropathy impair protective sensation and foot biomechanics; peripheral arterial disease (PAD) limits tissue perfusion; abnormal gait and pressure distribution precipitate tissue breakdown; and infection further exacerbates tissue damage. 4 Early detection of high-risk feet, personalized offloading strategies, and accurate prognostication are critical to reducing morbidity and health care costs.
Artificial intelligence (AI) and machine learning (ML) enable extraction of complex, nonlinear patterns from diverse data modalities—wound images, infrared thermograms, biomechanical signals, electronic health records (EHRs), and high-throughput omics—to support DFU care. These technologies offer significant potential to improve DFU management by enabling earlier detection, personalizing treatment strategies, and providing continuous monitoring capabilities that complement traditional clinical approaches.5,6 This review examines AI/ML applications in four domains:
We critically appraise methodologies, performance metrics, clinical integration challenges, and propose future research directions.
Search Methodology
We conducted a comprehensive narrative review following established guidelines for AI/ML health care applications. Literature search was performed in PubMed, IEEE Xplore, and Google Scholar databases using terms: “artificial intelligence,” “machine learning,” “diabetic foot ulcers,” “biomechanics,” “thermography,” and “prognostic modeling” from 2020 to 2024. Studies were included if they: (1) applied AI/ML methods to DFU diagnosis, treatment, or prognosis; (2) used quantitative outcome measures; (3) were published in peer-reviewed journals.
AI/ML in DFU Diagnosis
CNN-Based Wound Image Classification
The use of convolutional neural networks (CNNs) for DFU image analysis has shown promising diagnostic accuracy. Almufadi et al fine-tuned seven pretrained neural network architectures on DFU part B dataset including high number of clinical and thermographic images labeled ischemia versus infection. The EfficientNetB0 architecture combined with a logistic regression classifier achieved 97% accuracy and 0.96 F1-score for ischemia, while EfficientNetB0 plus AdaBoost reached 93% accuracy for infection. 7 This level of accuracy suggests potential for point-of-care diagnosis, which could reduce diagnostic delays and support earlier clinical intervention.
Panamonta et al 8 similarly applied deep CNNs to plantar thermogram images for diabetic foot risk classification, reporting sensitivity of 81.2% and specificity of 64.0% using five-fold cross-validation on 153 images. While further optimization is needed for clinical implementation, the sensitivity achieved makes this approach potentially valuable for screening applications where high detection rates are prioritized.
Ensemble methods combining multiple CNN architectures consistently outperformed single-model approaches, with improvement in diagnostic accuracy. 9 This improvement represents meaningful progress that could translate to better patient outcomes when implemented at scale.
Thermogram-Based Severity Stratification
Khandakar et al addressed multiclass severity classification by first performing
Chemello et al’s 5 narrative review emphasizes the integration of infrared thermography with other sensor modalities feeding support vector machine (SVM) and random forest classifiers, achieving >85% sensitivity and specificity across studies. This multimodal approach addresses the inherent limitations of single-modality assessments for complex clinical conditions like DFU risk.
Basiri et al developed a comprehensive protocol for multimodal DFU image collection that incorporated thermal imaging alongside RGB and depth cameras for enhanced wound assessment. Using FLIR ONE Gen 3 smartphone thermal infrared cameras, they collected 3871 thermal images from 269 participants, demonstrating the practical implementation of thermography in clinical DFU management. 11 Their approach included predebridement and postdebridement thermal imaging, revealing important physiological insights: wounds exhibited lower temperatures prior to debridement due to necrotic tissue with poor blood supply, while postdebridement thermal imaging showed temperature increases reflecting improved blood circulation and acute inflammatory response. This thermal mapping capability, when projected onto RGB images, provided quantitative assessment of debridement effectiveness and could enable early detection of tissue deterioration before visible clinical changes occur. 11 The integration of standardized thermal imaging protocols with comprehensive clinical data collection represents a significant step toward establishing thermography as a routine diagnostic tool in DFU care.
Biomechanical Risk Classification via Wearables
Integration of computational models with ML
Recent advances combine biomechanical modeling approaches with ML to enhance DFU prediction accuracy. Zhang et al 12 developed an approach using multilayer perceptron (MLP) neural networks to predict dynamic plantar pressure from footprint images, addressing the need for more accessible pressure assessment methods compared to traditional sensor-based equipment. This approach could help make biomechanical assessment more widely available in clinical settings.
Singh et al 13 employed finite element modeling to conduct extensive biomechanical analysis of DFUs, developing full-scale foot models with ulcers of differing sizes and depths at different plantar sites. This computational modeling approach provides insights into tissue mechanics that could inform both prevention and treatment strategies.
Technical advances in biomechanical signal processing
Wearable sensors enable continuous, noninvasive monitoring of gait and foot mechanics. Guiotto et al captured EMG and ground reaction force (GRF) data from 80 participants during gait. Their ML approach using biomechanical features demonstrated the potential for distinguishing diabetic patients from healthy controls. 14 This validates biomechanical alterations as potential digital biomarkers for clinical assessment.
Agrawal et al leveraged smart insole-embedded pressure sensors in 150 subjects. They computed dynamic plantar pressure features and compared static thresholding versus ML models. The AdaBoost pipeline achieved 85% classification accuracy, significantly outperforming the 67% accuracy of static methods. 15 This improvement demonstrates the value of ML approaches over traditional threshold-based methods for pressure analysis.
Data standardization for biomechanical applications
Ferber et al 16 provided important perspectives on how data science methods can be incorporated into gait biomechanics research to advance understanding and improve treatment planning procedures. Their work highlights the challenges that remain for effectively using data science approaches in clinical gait analysis, including the need for better infrastructure and incentives for sharing data, and education across the disciplines of biomechanics and data science.
AI/ML in DFU Treatment
Personalized Offloading Prescription
Offloading is a cornerstone of DFU management, yet adherence and effectiveness vary. 6 Traditional statistical approaches to biomechanical assessment demonstrate important limitations when compared to AI/ML methodologies (Table 1). The enhanced performance of ML approaches across multiple assessment domains suggests significant clinical value for implementation in DFU prevention and management programs.
Comparison of Traditional Statistical Versus AI/ML Approaches in Biomechanical Assessment for DFU.
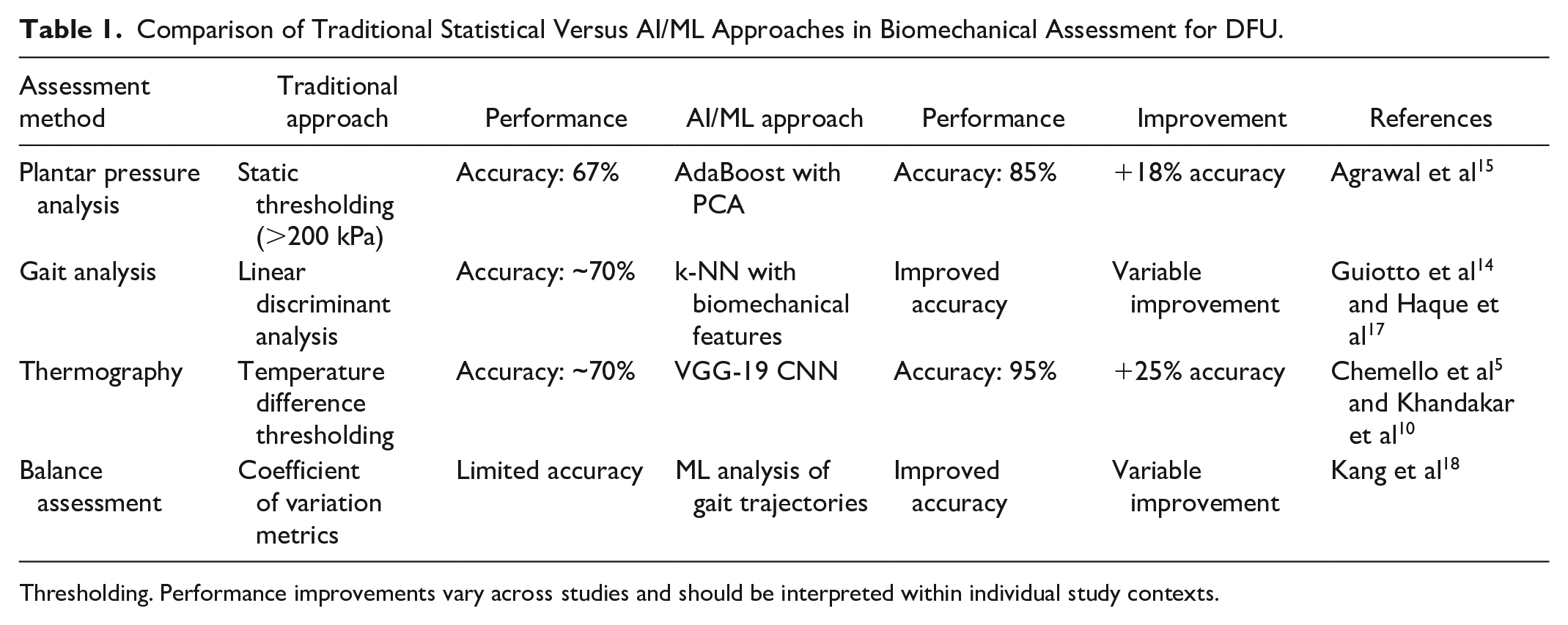
Thresholding. Performance improvements vary across studies and should be interpreted within individual study contexts.
Biomechanical modeling for treatment planning
Computational approaches are increasingly being applied to optimize treatment strategies. Singh et al’s 13 finite-element modeling work provides a foundation for understanding tissue mechanics in DFUs, which could inform personalized treatment approaches. Such biomechanical insights may enable detection of tissue deterioration before visible clinical changes occur.
Zhang et al’s 12 approach to predicting plantar pressure from footprint images using MLP neural networks demonstrates how AI can make sophisticated biomechanical analysis more accessible for routine clinical use. This computational efficiency enables practical biomechanical analysis during clinical visits for treatment optimization.
Virtual Coaching and Tele-Rehabilitation
Telemedicine platforms enhanced by ML can deliver tailored exercise regimens and support patient engagement in DFU care. Research in this area shows promise for extending specialized DFU care through digital platforms, particularly valuable for patients in underserved areas. 19 This approach demonstrates the feasibility of using technology to improve access to specialized care.
Molecularly Informed Wound Care
Transcriptomic ML pipelines enable mechanism-based wound interventions by identifying molecular targets for therapeutic intervention. Advanced ML algorithms including LASSO regression and support vector machine recursive feature elimination (SVM-RFE) show promise for identifying healing-associated genes from tissue transcriptome data. 20 This computational approach suggests potential opportunities for developing targeted therapeutic interventions, though clinical validation would be required.
AI/ML in DFU Prognosis
Neural Network Survival Models
Accurate long-term prognosis guides resource allocation and patient counseling. Popa et al developed MLP classifiers predicting five- and ten-year mortality in 635 DFU patients. Inputs included demographics, ulcer grading systems, kidney function, and comorbidities. The models achieved area under the curve (AUC) values around 0.74. 21 This level of prognostic accuracy could support more informed discussions about treatment planning and resource allocation.
Random Forests for Sepsis-Related Mortality
Random forest models excel at identifying the most important clinical features for prognosis, providing valuable insights for clinical decision-making. Matsinhe et al applied random forests to 114 diabetic foot sepsis admissions during the COVID-19 pandemic. The model’s feature importance ranking revealed serum potassium, urea, creatinine, and HbA1c as the most predictive variables, with SHapley Additive exPlanations (SHAP) values quantifying each feature’s contribution to individual predictions. The model achieved 87% accuracy and 0.82 AUC for in-hospital mortality. 22
Feature importance analysis enables clinicians to understand which parameters most strongly influence prognosis, supporting more focused monitoring and intervention strategies. This transparent ranking of predictive features can help optimize clinical workflow by highlighting the most critical parameters for monitoring.
Data Augmentation for Prognostic Modeling
Limited DFU cohort sizes challenge robust prognostic modeling. Alkhalefah et al implemented a conditional generative adversarial network (cGAN) to synthesize thermogram patches from real images. Training models on real + synthetic data improved intersection over union (IoU) performance, demonstrating how generative AI can enhance model performance in limited-data settings. 1 This improved accuracy in healing assessment could support better resource planning and patient expectation management.
AI-Driven Development of Novel Therapeutic Strategies
Generative AI for Data Generation
Generative models address data paucity and class imbalance in DFU research. Alkhalefah et al’s cGAN architecture produced physiologically realistic thermogram samples that preserved vascular and inflammatory patterns crucial for analysis. 1 Such synthetic datasets enhance model robustness in limited-data settings and could accelerate research progress in DFU subtypes where collecting sufficient real-world data presents significant challenges.
Large Language Models in DFU Care Coordination
Recent advances in LLMs represent an emerging frontier in diabetic foot care applications, particularly for patient education, symptom triage, and care coordination. These AI systems can process natural language queries and potentially provide evidence-based responses to patient concerns about foot care, wound management, and prevention strategies.
Several potential applications warrant investigation for future implementation. Large language model-powered systems could theoretically provide patient education about proper foot hygiene, early warning signs of complications, and guidance on when to seek medical attention. Important considerations include the need for rigorous clinical validation, potential liability concerns, regulatory requirements, and the necessity for robust safeguards to ensure appropriate medical guidance. Current applications remain largely experimental and require substantial research before clinical implementation.
Wearable-Derived Digital Biomarkers
Wearable IMUs and pressure sensors furnish continuous digital biomarkers for DFU risk and recurrence. Research has consolidated evidence that various gait parameters including gait speed, stride time variability, and postural sway correlate with ulcer risk. 18 Integration of these digital biomarkers into mobile health platforms could enable remote monitoring and support timely clinical intervention.
Discussion
This review reveals that AI/ML methodologies have shown promise across multiple domains of DFU care, with diagnostic accuracies ranging from 81% to 97% for image and thermogram analysis,5,7,10 potential for biomechanical risk stratification,14,15 and prognostic models achieving AUC values around 0.74 to 0.82.21,22 Generative AI approaches show promise for addressing data limitations, 1 while emerging applications in transcriptomics suggest opportunities for targeted therapeutic development. 20
Value-Added Analysis of AI/ML in Biomechanical Assessment
While traditional biomechanical assessments rely on threshold-based metrics and linear statistical methods, AI/ML approaches demonstrate potential added value through several mechanisms. First, they may capture nonlinear relationships between biomechanical variables that traditional methods overlook.14,16 Second, ML models can potentially integrate multimodal biomechanical data that would be challenging to analyze simultaneously with conventional approaches.12,17 Third, deep learning methods may automatically extract relevant features from raw biomechanical signals, reducing the need for manual feature engineering.13,18
However, this potential improved performance comes with trade-offs: increased computational requirements, reduced interpretability in some models, and dependence on larger training datasets. 16 The decision to employ AI/ML should be guided by the complexity of the biomechanical assessment task, with simpler statistical approaches remaining appropriate for straightforward analyses.
Despite these advances, several challenges impede translation to routine care:
1. ● Imaging: Diverse wound photography lighting and angles hinder model generalizability.7,9,11 ● Thermography: Varied camera specifications and ambient temperature settings introduce noise.5,10 ● Sensors: IMU placement, sampling rates, and insole sensor calibration differ across studies.16,18 ● EHR schemas: Heterogeneous comorbidity and outcome coding complicate multicenter model training.19,21
2.
3.
4. ● Seamless embedding into EHR and mobile platforms demands interoperability (FHIR, HL7) and user-friendly interfaces. ● Training clinicians and patients to use AI-driven tools is essential to uptake.
5.
6.
Critical Evaluation of Validation Methodologies
The quality of validation approaches varies considerably across the reviewed studies. Several methodological limitations warrant caution when interpreting reported performance metrics:
These methodological limitations may affect the generalizability of reported performance estimates to real-world clinical settings and should be considered when interpreting study results.
Research Priorities and Future Directions
Based on our review, we propose the following specific research priorities:
These priorities should be pursued through interdisciplinary collaborations spanning biomechanics, computer science, endocrinology, vascular medicine, and implementation science to ensure both technical innovation and clinical relevance.
Conclusion
Artificial intelligence and ML have demonstrated potential to advance DFU care across diagnosis, treatment personalization, prognostication, and novel strategy development. These technologies show promise for improving clinical outcomes through enhanced diagnostic capabilities, personalized treatment approaches, continuous monitoring, and data-driven prognostic insights. However, significant challenges remain including data standardization, model explainability, regulatory compliance, prospective validation, and equitable implementation.
The evidence supports continued investment in AI/ML development for DFU care, with priority given to prospective validation studies, standardization initiatives, and implementation strategies that ensure these technologies can benefit diverse patient populations across various health care settings. Collaborative efforts among clinicians, data scientists, regulators, and patients will be essential to translate AI-driven innovations into routine DFU management.
Footnotes
Author’s Note
Investigation was performed at the Bahcesehir University, Department of Orthopedics and Traumatology
List of Abbreviations
AI, artificial intelligence; AUC, area under the curve; CDSS, clinical decision support system; cGAN, conditional generative adversarial network; CNN, convolutional neural network; DFU, diabetic foot ulcer; EHR, electronic health record; EMA, European Medicines Agency; EMG, electromyography; FAIR, findable, accessible, interoperable, reusable; FDA, food and drug administration; FEM, finite-element modeling; FHIR, fast health care interoperability resources; GDPR, general data protection regulation; Grad-CAM, gradient-weighted class activation mapping; GRF, ground reaction force; HIPAA, health insurance portability and accountability act; HL7, health level seven; IMU, inertial measurement unit; IoU, Intersection over Union; LIME, local interpretable model-agnostic explanations; LLM, large language model; LSTM, long short-term memory; ML, machine learning; MLP, multilayer perceptron; PAD, peripheral arterial disease; PCA, principal component analysis; RCT, randomized controlled trial; ROI, return on investment; SaMD, Software as a medical device; SHAP, SHapley additive exPlanations; SVM, support vector machine; VGG, visual geometry group; XAI, explainable artificial intelligence
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
