Abstract
Aims:
Hybrid closed-loop (HCL) systems have become the standard of care for type-1 diabetes patients. However, they require exact carbohydrate counting (ECC), which can be complex. We conducted a meta-analysis to assess whether simplified meal announcement (SMA) is inferior to ECC by comparing the percentage of time spent in the time in range (TIR) 3.9 to 10 mmol/l.
Materials and Methods:
PubMed, EMBASE, and Cochrane Central database were searched for randomized controlled trials (RCTs) that compared ECC to SMA and reported the outcomes of (1) percentage of 3.9 to 10 mmol/l glucose level time; (2) percentage of 3.9 to 7.8 mmol/l glucose level time; (3) total daily insulin units (TDI) per kg; and (4) coefficient of variation (CV) of glucose (%). Heterogeneity was examined with
Results:
We included 4 RCTs (three of them crossover) with 137 patients, of whom 118 underwent SMA. Follow-up ranged from 2 weeks to 12 months. The pooled mean difference (MD) of percentage of time spent in TIR was −3.28 [−6.00, −0.56], indicating lower proportion with SMA. Two studies reported the percentage of TIR of 3.9 to 7.8 mmol/l, with a pooled effect size MD of −3.36 [−5.80, −0.92]). The pooled MD in TDI units per kg was 0.00 [−0.05, 0.05], and CV of glucose was 0.60 [0.02, 1.18]).
Conclusions:
Simplified meal announcement may be considered as an alternative to ECC, but further research is needed to confirm its broader applicability.
Introduction
Type-1 diabetes is an autoimmune disease that affects the regulation of blood sugar and represents approximately 5% to 10% of all diabetes. 1 Being usually diagnosed in children and young adults, it is the clinical manifestation of β cells destruction in the pancreatic islets, which leads to insulin deficiency and therefore need of persistent insulin therapy. According to a study published in 2021, an increase in prevalent cases to 13.5 to 17.4 million in type-1 diabetes is expected by 2040. 2 The burden of this condition remains immense: from reduced life expectancy and long-term complications like kidney and cardiovascular disease to psychological challenges, such as anxiety and depression. 3
Hybrid closed-loop (HCL) systems—also known as “artificial pancreas” or “automated insulin delivery systems”—are able to monitor glycemia and administer the needed insulin automatically, by linking continuous glucose monitoring (CGM) with insulin pump technology. These systems have demonstrated their efficacy by optimizing glycemic parameters such as time in range (TIR) and HbA1c, achieving the recommended glycemic targets and becoming the standard of care for patients with type-1 diabetes.4 -7 Yet, they still require exact carbohydrate counting (ECC) when announcing meals, 8 which can be complex and challenging for a large proportion of this population, negatively affecting their quality of life.9,10 Meanwhile, simplified meal announcement (SMA) is a more practical approach where patients announce their meal size (small, medium, or large) and the system is responsible for adjusting the insulin dose based on these preset categories.
Several studies have evaluated glucose control by comparing SMA to ECC in type-1 diabetes patients using HCL systems, showing contradictory results. To our knowledge, no previous meta-analysis has assessed the impact of SMA on glucose control in this patient population. Therefore, we aimed to conduct a systematic review and meta-analysis to assess whether SMA is noninferior to ECC by comparing the percentage of time spent in the target glucose range (time in range, TIR) in type-1 diabetes patients using HCL systems. A non-inferiority margin of 5% has been established based on the worldwide consensus on what constitutes a clinically meaningful difference. 11
Materials and Methods
Adherence to Guidelines
This systematic review and meta-analysis of randomized controlled trials (RCTs) followed Cochrane Handbook for Systematic Reviews of Interventions for conducting systematic synthesis and The Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement for reporting meta-analyses. 12 The protocol of this meta-analysis was registered on PROSPERO (ID CRD42024550750).
Inclusion Criteria
Studies were included only if they met the following eligibility criteria: RCTs comparing SMA with ECC. Studies that did not report outcomes of interest in this specific population were excluded. We also excluded studies with overlapping patient populations.
Search Strategy
We systematically searched PubMed, EMBASE, and Cochrane Central databases for RCTs up to December 15, 2024 that compared ECC to SMA in type-1 diabetes patients using HCL systems. The search used terms such as “type 1 diabetes”, “hybrid closed loop,” “automated insulin delivery,” “simplified meal announcement,” “carbohydrate counting,” and “randomized controlled trial.” The complete search strategy is presented in the Supplementary Appendix. Only articles published in English were eligible, and we did not search gray literature. We also performed a backward snowballing technique, identifying eligible studies by reviewing the references cited in prior publications.
Screening of the Studies
All titles and abstracts identified from the search were independently reviewed by two authors (CSG and PA). In our meta-analysis, Rayyan Software (Rayyan Systems Inc., Cambridge, MA, USA) was used for the screening and selection process. 13 Three authors (CSG, PA, and PL) conducted the data extraction from included studies. Eventual conflicts were resolved through consensus among the authors.
Extracted Data
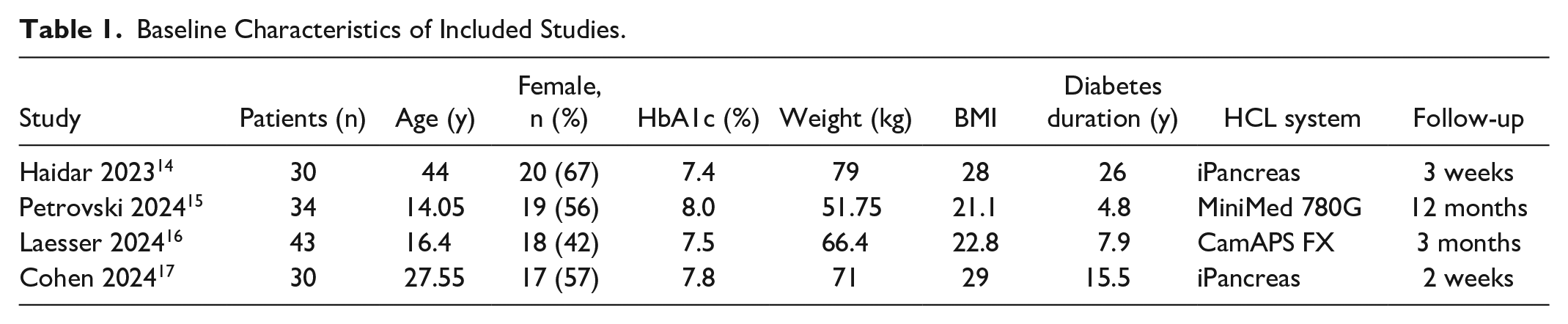
Outcome data extraction was performed independently by two authors (CSG and PL) using a standardized document, with any disagreements resolved by consensus. In addition, two authors (CSG and PA) extracted further baseline data for the individual studies, including study and patient characteristics (Table 1). Participant-level data were not requested.
Baseline Characteristics of Included Studies.
Outcomes of Interest
Our primary outcome was TIR 3.9 to 10 mmol/l. Secondary outcomes of interest were percentage of 3.9 to 7.8 mmol/l glucose level time; total daily insulin units (TDI) per kg; and coefficient of variation (CV) of glucose (%). Safety endpoints included diabetic ketoacidosis and severe hypoglycemia.
During the systematic review process, we observed that the selected articles repeatedly reported additional outcomes that were relevant to our research question and the management of type-1 diabetes patients with HCL systems. Consequently, we included these outcomes in our meta-analysis to provide a more comprehensive understanding of the interventions’ effects. The additional outcomes examined in our study include: glycemia levels below 3 mmol/L, glycemia levels below 3.9 mmol/L, glycemia levels above 10 mmol/L, glycemia levels above 13 mmol/L, basal insulin, bolus insulin, insulin units per day, mean glucose, and the standard deviation (SD) of glucose.
The inclusion of these additional parameters was based on their relevance to the overall understanding of glycemic control and diabetes management in the context of carbohydrate counting. Monitoring low and high blood glucose levels is crucial for identifying risks associated with hypoglycemia and hyperglycemia, respectively. Basal and bolus insulin measures, along with the TDI dosage, provide insights into the effectiveness of insulin therapy and treatment strategies in patients using HCL systems. Mean glucose levels and their variability (SD) are essential for evaluating overall glycemic control, as high values may lead to long-term complications such as retinopathy, nephropathy, and peripheral neuropathy. 18
These outcomes were frequently reported in the literature and are of significant interest to the type-1 diabetes research community. Including them in our meta-analysis allows for a more comprehensive assessment of the management of type-1 diabetes with SMAs.
Quality Assessment
Each included study was assessed for risk of bias using the Cochrane Risk of Bias Assessment Tool (RoB-2) for RCTs or the RoB-2 with crossover extension. The evaluations were independently conducted by two investigators (CSG and PA). Any discrepancies were resolved through discussion and consensus.
Statistical Analysis
A random-effect generic inverse variance (GIV) method was applied to all analyses. Ninety-five confidence intervals were used in the study (95% confidence interval [CI]). We have presented results of continuous outcomes as GIV mean difference (MD). Heterogeneity was assessed with
Results
Our systematic search retrieved a total of 547 articles, as detailed in Figure 1. Following the removal of duplicates, 390 articles were screened based on title and abstract review. Ultimately, four RCTs,14 -17 three of which were crossover studies, met the inclusion criteria. A total of 137 patients were included, of whom 118 were assigned to SMA. Study characteristics are presented in Table 1. Most of the studies included a mixed population of adults and adolescents. Among the participants, 54% were female, with a mean age of 24.29 years and a mean body mass index (BMI) of 25.19. The mean duration of diabetes was 12.76 years, and baseline HbA1c levels ranged from 7.2 to 8.4. Follow-up periods varied from 2 weeks to 12 months.
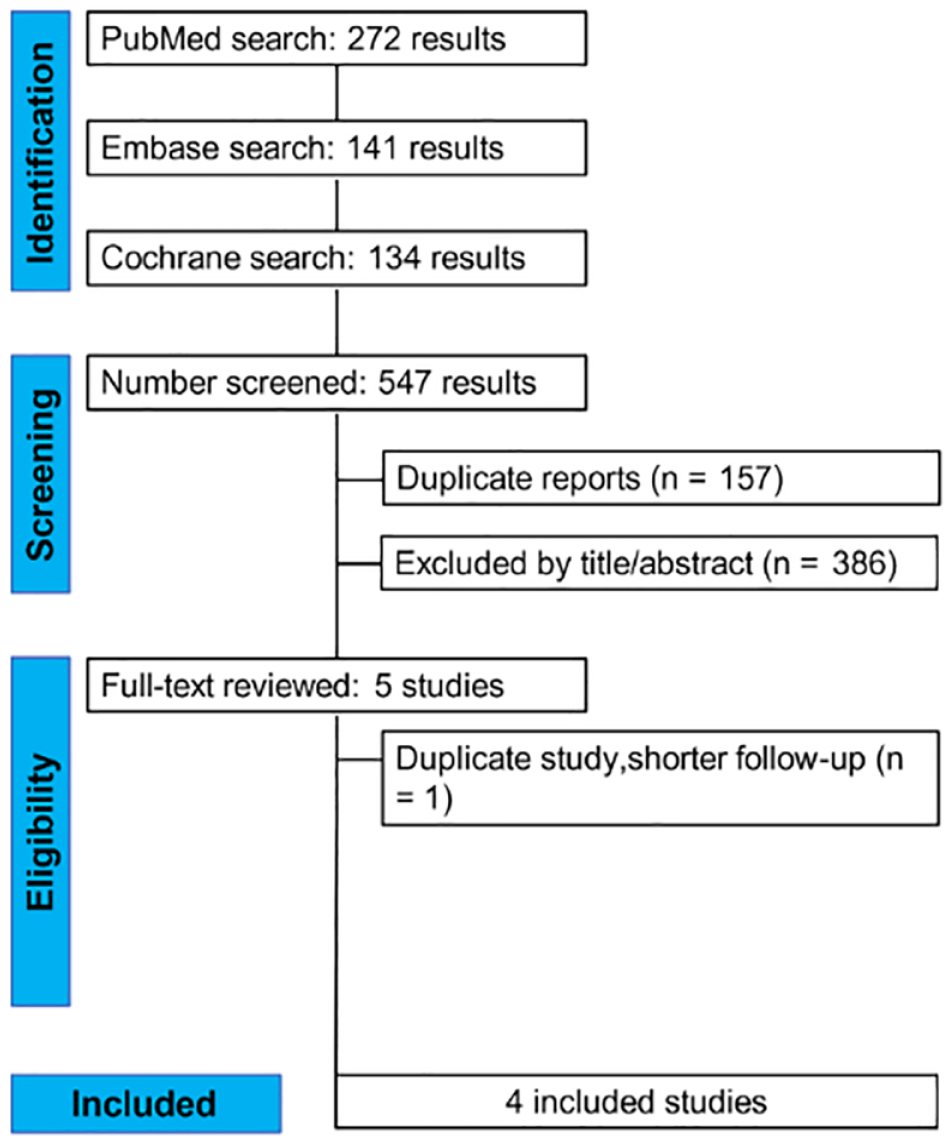
PRISMA flow diagram of study screening and selection.
In this meta-analysis, the pooled MD for the percentage of time spent in the target glycemia in TIR of 3.9 to 10 mmol/L was −3.28 (95% CI [−6.00, −0.56]), indicating a lower proportion of time within the target range compared to the control group (Figure 2). The analysis revealed significant heterogeneity among the included studies (
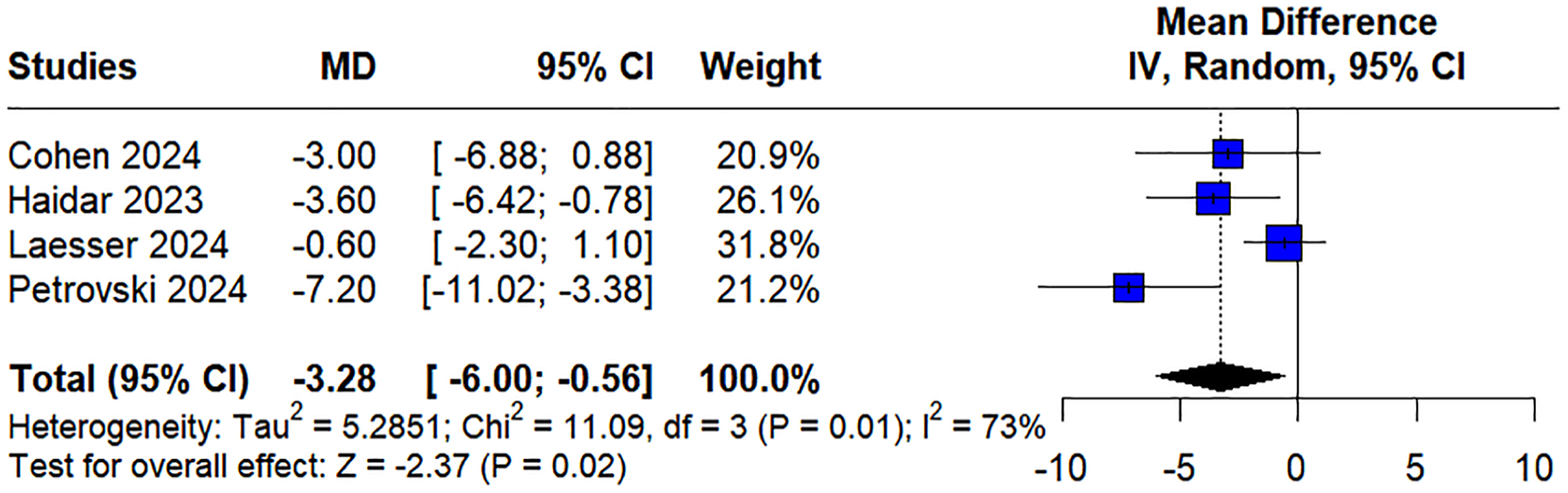
Forest plot for TIR 3.9 to 10 mmol/L.
Regarding the glycemia range of 3.9 to 7.8 mmol/L, two studies reported this outcome, with a pooled MD of −3.36 (95% CI [−5.80, −0.92]) and minimal heterogeneity (
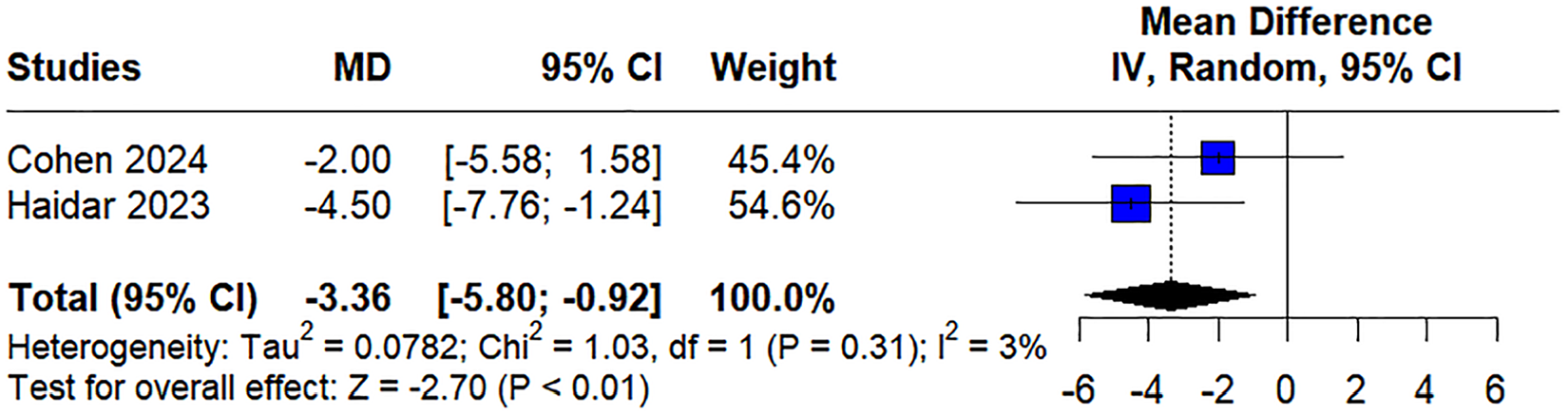
Forest plot for TIR 3.9 to 7.8 mmol/L.
For TDI per kg, the pooled MD was 0.00 (95% CI [−0.05, 0.05]), indicating no significant difference between the groups (Figure 4). Significant heterogeneity was observed (
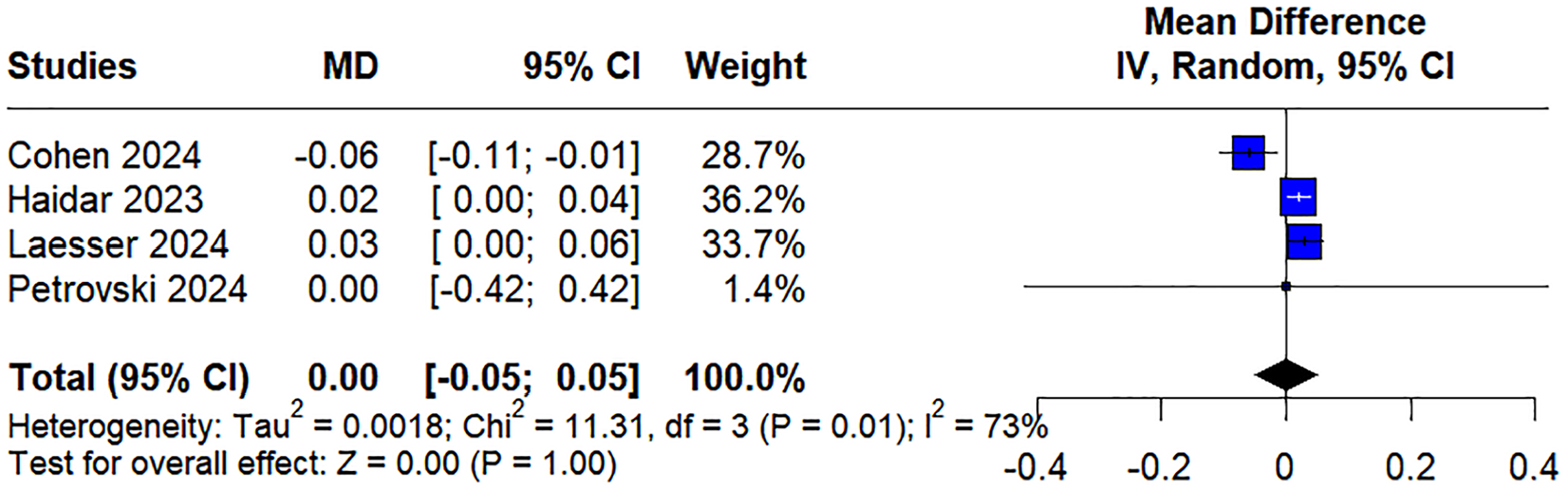
Forest plot for total daily insulin units (TDI) per kg.
The pooled MD for the CV of glucose was 0.60 (95% CI [0.02, 1.18]) with no significant heterogeneity among the included studies (
For glycemia levels below 3 mmol/L, the pooled MD was −0.03 (95% CI [−0.08, 0.02]), with low heterogeneity (
For glycemia levels above 10 mmol/L, the pooled MD was 2.28 (95% CI [0.07, 4.49]), with moderate heterogeneity (
Basal insulin demonstrated a pooled MD of 1.60 (95% CI [0.98, 2.23]) with low heterogeneity (
For insulin units per day, the pooled MD was −1.12 (95% CI [−5.30, 3.05]) with high heterogeneity (Tau² = 10.6282, Ch
Finally, the pooled MD for mean glucose was 0.22 (95% CI [0.06, 0.39]) with low heterogeneity (
Safety Endpoints
Only Laesser study reported safety endpoints, identifying one serious adverse event (diabetic ketoacidosis) with SMA, and two with ECC (diabetic ketoacidosis and hypoglycemia). All of these events were deemed unrelated to the study.
Risk of Bias in Included Studies
The risk of bias of each RCT is presented in the Supplementary file (Table S1). We assessed the risk of bias in the four RCTs using Risk of bias by Cochrane Risk of Bias tool for randomized trials (ROB2) or the RoB-2 with crossover extension. All four trials exhibited “some concerns” regarding deviations from intended interventions, leading to an overall risk of bias categorized as “some concerns.”
Discussion
In this systematic review and meta-analysis of four RCTs involving 137 patients, we compared the glycemic outcomes of the SMA strategy versus ECC in type-1 diabetes patients using HCL systems. Our primary findings were: (1) a small but statistically significant reduction in TIR (3.9-10 mmol/L) with SMA; (2) a statistically significant decrease in TIR (3.9-7.8 mmol/L) in the SMA group; (3) no significant difference in TDI per kg, and (4) a significantly higher CV of glucose in the SMA group.
In addition to the primary analysis, we explored several secondary outcomes that were consistently reported across the included studies, providing additional insights into the effects of the interventions. The secondary findings were as follows: (1) no significant difference between the groups in glycemia levels below 3 mmol/L; (2) significantly lower glycemia levels below 3.9 mmol/L in the SMA group; (3) significantly higher glycemia levels above 10 mmol/L in the SMA group; (4) a significant increase in glycemia levels above 13 mmol/L in the SMA group; (5) a significant increase in basal insulin in the SMA group; (6) no statistically significant increase in bolus insulin in the SMA group; (7) no statistically significant reduction in insulin units per day in the SMA group; (8) a small but statistically significant improvement in mean glucose in the ECC group; and (9) a small but statistically significant increase in the standard deviation of glucose in the SMA group.
Maintaining glycemic control in type-1 diabetes while avoiding hypo- or hyperglycemia remains a significant challenge for patients and requires a difficult balance between medication, diet and lifestyle. The SMA strategy achieved a high TIR (above the recommended 70%). However, it did not meet the established noninferiority margin of 5%. The small but statistically significant reduction in TIR (3.9-10 mmol/L) and TIR (3.9-7.8 mmol/L) with SMA were accompanied by significantly lower glycemia levels below 3.9 mmol/L, higher glycemia levels above 10 mmol/L and 13 mmol/L, and an increase in basal insulin. While these findings are statistically significant, the confidence intervals indicate that the observed variations are unlikely to have substantial clinical relevance. Although the CV of glucose was slightly higher in the SMA group, this increase may not be significant enough to suggest a definitive impact on oxidative stress. Despite no significant difference in TDI per kg, the increase in basal insulin and stable bolus insulin may highlight the different insulin management required with SMA. The small but statistically significant improvement in mean glucose in the ECC group and the increase in the standard deviation of glucose in the SMA group further underscore the differences in glycemic control between the two strategies.
Nevertheless, SMA may benefit patients who find ECC burdensome. Similar outcomes have been observed in other studies. For example, in a study with 14 children and adolescents with type-1 diabetes using the Medtronic MiniMed 780G system, 19 researchers found that unannounced snacks of up to 20 g of carbohydrates can prevent a blood glucose increase of ≥50 mg/dL, while unannounced meals of up to 30 g are safe. The mean TIR was ≥70% in the 2 hours following each snack, too. In addition, a single-arm design study of 14 adults with type-1 diabetes 20 evaluated the safety and efficacy of the HCL systems with and without meal announcements. Results showed that unannounced small meals (≤20 g carbohydrates) did not worsen glycemic control. Furthermore, Blervaque et al 21 published findings in a letter to the editor, based on a sample extracted from their type-1 diabetes database. In the study, 1958 patients from seven European countries were included, and a total of 299 387 meals were recorded using ECC and 315 432 using SMA. During the postprandial period, the mean TIR was 63.0% in the ECC group and 62.6% in the SMA group. While this study provides valuable real-world data from a diabetes registry, our meta-analysis takes a different approach by focusing exclusively on RCTs that directly compare ECC and SMA strategies under controlled conditions. Despite the smaller sample size, including RCTs strengthens the evidence on the efficacy of SMA.
Given these findings, the SMA strategy may present a viable alternative to the ECC method, especially for patients who struggle with carbohydrate counting. Patients and health care providers might consider SMA as a simpler and yet effective approach for managing type-1 diabetes with HCL systems. While SMA shows promise, its use should be customized to individual patient preferences, needs, and clinical circumstances. Further research with larger sample sizes and standardized outcome measures is necessary to confirm and extend these findings, but it is likely that recommendations from meal management during HCL use will be revised. This is especially relevant as ongoing research focuses on artificial intelligence-enhanced meal detection and carbohydrate size estimation. 22
This study has several strengths worth acknowledging. The inclusion of only RCTs ensures a high level of evidence quality. Furthermore, one of the included studies had a follow-up period of 12 months, providing robust data on the intervention’s effects.
However, despite these strengths, there are important limitations to consider. First, the analysis included only four studies with a total sample size of 137 patients, limiting statistical power and generalizability. Although RCTs are considered high-quality evidence, some of the included studies had some concerns regarding risk of bias, potentially weakening the overall strength of the evidence. Due to these limitations, we did not conduct a GRADE assessment. In addition, variability in study designs, patient populations, and intervention protocols introduced heterogeneity, affecting comparability. One notable source of variability was the differences in SMA strategies used across the four studies, which may have contributed to the observed heterogeneity. While all studies implemented some form of meal announcement without ECC, differences in implementation—such as predefined meal categories (e.g., snack, small, medium or large meals) could have influenced glycemic variability and overall outcomes. Variability in meal classification and insulin adjustments across studies may explain some of the heterogeneity observed in the pooled results, underscoring the need for standardized SMA definitions in future research. Moreover, the follow-up periods among the included studies varied significantly, ranging from two weeks to 12 months, potentially impacting the consistency of long-term outcomes. Finally, the lack of standardized outcome measures across studies, especially the use of different HCL systems with varying algorithms and protocols, might have introduced bias and limited the ability to perform a comprehensive meta-analysis.
Conclusion
Simplified meal announcement appears to be a promising alternative to the ECC method, delivering good clinical results while potentially reducing the effort and burden for people with type-1 diabetes. These findings suggest simplified meal announcement could be considered for patients using HCL systems. However, further studies are needed to validate these results, particularly in different subgroups of patients, to better understand its applicability across diverse populations and clinical scenarios.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251351994 – Supplemental material for Is Simplified Meal Announcement an Alternative to Exact Carbohydrate Counting in Patients With Type-1 Diabetes Using an Advanced Hybrid Closed-Loop System? A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-dst-10.1177_19322968251351994 for Is Simplified Meal Announcement an Alternative to Exact Carbohydrate Counting in Patients With Type-1 Diabetes Using an Advanced Hybrid Closed-Loop System? A Systematic Review and Meta-Analysis by Cristina Sicorschi Gutu, Paweł Łajczak, Patricia-Maria Anton, Krisztina Schmitz-Grosz, Carsten Sommer-Meyer, Cintia Gonzalez Blanco and Günter Niklewski in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
HCL, hybrid closed loop; ECC, exact carbohydrate counting; SMA, simplified meal announcement; TIR, time in range; RCT, randomized controlled trials; TDI, total daily insulin units; CV, coefficient of variation; SD, standard deviation; MD, mean difference; CI, confidence interval; ROB-2, risk of bias.
Author Contributions
CSG contributed to the study’s conception and design, conducted the literature search, study triage, data extraction, risk of bias, and was responsible for drafting, writing, and preparing the original manuscript. PL participated in data extraction and conducted statistical analyses. PA participated in data extraction and conducted risk of bias. KSG assisted in manuscript writing. CSM assisted in manuscript writing. CGB supported the literature search and reviewed the manuscript. GN reviewed the manuscript and provided final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
