Abstract
Background:
We tested the safety and performance of the “insulin-only” configuration of the bionic pancreas (BP) closed-loop blood-glucose control system in a home-use setting to assess glycemic outcomes using different static and dynamic glucose set-points.
Method:
This is an open-label non-randomized study with three consecutive intervention periods. Participants had consecutive weeks of usual care followed by the insulin-only BP with (1) an individualized static set-point of 115 or 130 mg/dL and (2) a dynamic set-point that automatically varied within 110 to 130 mg/dL, depending on hypoglycemic risk. Human factors (HF) testing was conducted using validated surveys. The last five days of each study arm were used for data analysis.
Results:
Thirteen participants were enrolled with a mean age of 28 years, mean A1c of 7.2%, and mean daily insulin dose of 0.6 U/kg (0.4-1.0 U/kg). The usual care arm had an average glucose of 145 ± 20 mg/dL, which increased in the static set-point arm (159 ± 8 mg/dL,
Conclusions:
The insulin-only configuration of the BP using either static or dynamic set-points and initialized only with body weight performed similarly to other published insulin-only systems.
Introduction
A closed-loop, blood-glucose (BG) control system, often referred to as an “artificial pancreas” or “bionic pancreas” (BP), provides automated or semi-automated glycemic control and has the potential to decrease the burden of diabetes care and reduce both the short- and long-term complications of diabetes. 1 Closed-loop BG control systems consist of three components: a continuous glucose monitor (CGM), an algorithm to determine the amount of insulin or other hormones to be infused, and infusion pumps to deliver hormones such as insulin or glucagon. Recent advances in this field have arisen from the development of automated drug-dosing algorithms and improvements in the accuracy, reliability, and ease of use of CGM technologies.
The BP system developed at Boston University has been tested in a variety of outpatient and home-use studies2-4 in adults and children with type 1 diabetes (T1D). The BP can be configured in an insulin-only, a glucagon-only, or a bihormonal (insulin and glucagon) configuration. The focus of this study is on the use of the insulin-only configuration of the BP in people with T1D. There are several practical reasons to consider this. In order to use a bihormonal BP in clinical practice, regulatory approval will be required of a stable, pumpable glucagon formulation or glucagon analog. Before that materializes, an insulin-only BP may provide a useful transition therapy in its own right in people with T1D. In addition, practical considerations require that a bihormonal BP system demonstrate safe transition to an insulin-only configuration whenever there are sudden changes in the ability of the device to deliver glucagon.
The BP has several attractive features, most of which are common to both the insulin-only and bihormonal configurations. In particular, the system only requires the user’s body weight for initialization, then the BP algorithm adapts continuously to the user’s ever-changing insulin needs automatically and in real time. 3 In addition, the system is unique in that it does not require carbohydrate counting. Although optional, the system performs best when the user announces meals, which triggers the system to automatically compute and administer a pre-meal insulin bolus. The optional meal-announcement feature only requests that users enter whether the meal is typical in size for that time of day or more or less than typical. Over time the algorithm adapts the size of the meal-announcement insulin bolus, separately for each time of day. The system is also unique in that it does not require any information about pre-programmed basal rates, insulin sensitivity factors, carbohydrate-to-insulin ratios, or the total daily dose (TDD) of insulin.
The objective of this feasibility study was to test the safety and performance of the BP in an insulin-only configuration. As this represented our first home-use study of the insulin-only configuration of the device, we tested the system using a conservative glucose target to reduce the risk of hypoglycemia. Further studies testing other glucose targets of the system are under way.
Methods
This was an open-label, non-randomized, pilot safety and feasibility study lasting 21 days (across three study arms of seven days each) (Figure S1). The institutional review board approved the protocol, and written informed consent was obtained from each participant. All participants were required to have had a diagnosis of T1D for at least one year, to be treated with an insulin pump for at least six months, and to be ≥18 years old. Exclusion criteria included history of coronary artery disease, congestive heart failure, active Graves’ disease, renal failure, and oral antidiabetic medications. Participants were encouraged to engage in their usual physical activity throughout the study period. There were no dietary restrictions or carbohydrate limitations. The participants had four study visits during the 21-day period. The usual-care arm of the study was one week in duration; during this time, subjects wore a blinded CGM. Subjects followed their own usual diabetes therapy for seven days using their own pump and their own CGM (if they normally used one). The usual-care arm was followed by seven days of automated glycemic control on the insulin-only BP initially configured with a fixed glucose target of 130 mg/dL. System performance was re-evaluated remotely 48 hours after initiation of the system, and the glucose target was then switched to either 145 mg/dL (if the subject had two episodes of hypoglycemia with a glucose <50 mg/dL on either one of the two days) or 115 mg/dL (if the subject had an average daily glucose of >180 mg/dL and no glucose values <65 mg/dL on day 2). The participants were informed of any adjustments. During the last one of the three seven-day study arms, subjects continued to use the insulin-only BP but with a dynamic, rather than static, glucose target. The dynamic setting allowed the BP to automatically vary the glucose target online within the range of 110 to 130 mg/dL. Even though the BP was operating in insulin-only mode and no glucagon doses were administered, the BP internally computed “intended” glucagon doses that would be administered if the system were in bihormonal mode. The target is adapted based on those internally computed glucagon doses; the higher the computed glucagon doses, the further the glucose target is raised.
System
The system consisted of a t:slim infusion pump (Tandem), a G4 Platinum AP CGM (Dexcom), and the BP insulin-dosing control algorithms, which were encoded in an app that ran on an iPhone 4S (Apple).3,4 The iPhone and the Dexcom CGM receiver were connected through their external communication ports and housed together in a custom enclosure. The iPhone, CGM, and enclosure together comprised the BP Control Unit. The BP app ran the insulin-dosing control algorithms, managed connectivity between the iPhone and the Dexcom receiver, and controlled the Bluetooth radio, which effectuated communication between the iPhone and the t:slim pump. The BP app had a graphical user interface (GUI) that displayed the current CGM glucose, the CGM trend, and the insulin doses. The BP app also provided the interface to input meal announcements. Meal announcements (1) specified a type of meal (as “breakfast,” “lunch,” or “dinner”), (2) designated the size of the meal (as “larger than typical,” “typical,” “smaller than typical,” or “just a bite”), and (3) triggered a partial meal-priming bolus, the size of which automatically adapts during the course of the trial to meet a target of 75% of the insulin required for that size and type of meal. The participants were encouraged to use the meal-announcement option throughout the study.
The BP managed all insulin dose calculations when the CGM was online and offline. When the CGM was offline, the control algorithm administered correction boluses of insulin as appropriate in response to any entered BG value, just as if they were CGM values. The GUI also displayed visual alarms associated with an audio signal if communication was dropped between the BP app and the t:slim pump, or if the CGM glucose was below a low threshold. The manual correction is not an option in this system.
The BP Control Unit communicated to a server that allowed the BP to support remote telemetry of CGM data. During both the usual-care and BP arms, if there were communication failures that were not resolved within 15 minutes, or if the sensor glucose was <50 mg/dL, an alert was sent to a study staff, who would then contact the subject. Subjects were trained on how to resolve communication failures between the BP and the CGM or pump and how to resolve problems related to communication with the cloud server.
Study subjects were also provided with a commercial FDA-approved glucometer (StatStrip Xpress, Nova Biomedical) and a second iPhone, so that their current location could be determined remotely with a dedicated study staff iPhone using the “Find My iPhone” utility.
Human factors (HF) testing was performed at enrollment and at study completion. HF testing included validated surveys on well-being (WHO-5; 5 items, higher score indicates better well-being), diabetes distress (Diabetes Distress Scale—T1D; 28 items; higher score means more distress), confidence to prevent or manage hypoglycemia (Hypoglycemia Confidence Scale; 8 items; higher score indicates more confidence), satisfaction with glucose monitoring methods (Glucose Monitoring Satisfaction Scale; 15 items; higher score indicates more satisfaction), and satisfaction with overall diabetes treatment (Diabetes Treatments Satisfaction Questionnaire; 8 items; higher score equals more satisfaction). Focus groups were held at the end of the study and were moderated by a psychologist to guide the discussion. Sessions were recorded, and conversations were transcribed and analyzed for recurrent themes and content.
Statistical Methods
The co-primary outcomes were (1) mean CGM glucose from day 3 to 7 (last five days of each study arm) on the system (until 2
Repeated-measures regression models (ANOVA) were used to test the differences between groups (three arms), and Bonferroni was used for within-group analyses. Statistical hypotheses were tested using a two-tailed
Results
Table 1 lists subject baseline data. The subjects were well controlled with an average HbA1c of 7.2% at baseline. CGM data from their usual-care arm showed an average CGM glucose of 145 mg/dL with an average time <70 mg/dL of 5.5% ± 4.2%.
Baseline Characteristics.
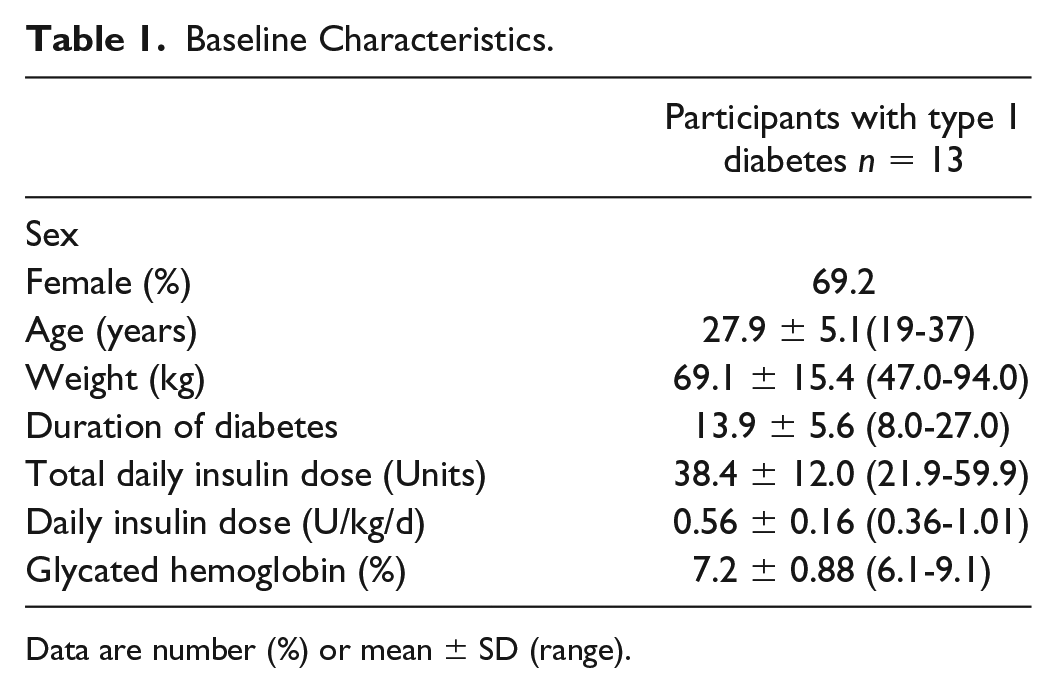
Data are number (%) or mean ± SD (range).
The study was conducted using two cohorts of eight subjects (Figure 1, Supplemental Figures S2-S16, Supplemental Tables S1 and S2). In the first cohort, there were three subjects who, in the first 48 hours, had no CGM glucose values <65 mg/dL and a mean CGM glucose of ≥180 mg/dL; thus their static set-point was lowered from 130 to 115 mg/dL for the next five days. No subject had two hypoglycemic events with a meter glucose <50 mg/dL in the first 48 hours, thus we did not utilize the set-point of 145 mg/dL. The first cohort completed the study arm using the dynamic set-point range of 115 to 145 mg/dL, and it was determined that this dynamic range was too wide, so a second cohort was enrolled with the dynamic set-point narrowed to 110 to 130 mg/dL. In addition, five subjects from the first cohort repeated the dynamic set-point arm using the new range of 110 to 130 mg/dL. This created a cohort of 13 subjects that was used for efficacy analysis of the dynamic set-point. Of these 13 subjects, all had used a static set-point of 130 mg/dL except for 1 subject, whose set-point was lowered to 115 mg/dL in week 2. Adverse events and safety data are reported for all 16 subjects who were enrolled in the study. All subject-level data for each of the 16 subjects are provided in supplemental Figures S2 to S17. The focus of this paper is on the 13 subjects who completed all three study arms with the final arm using a dynamic set-point that varied between 110 and 130 mg/dL. Data were analyzed using the last five days of each study week (Figure 2 and Table 2).
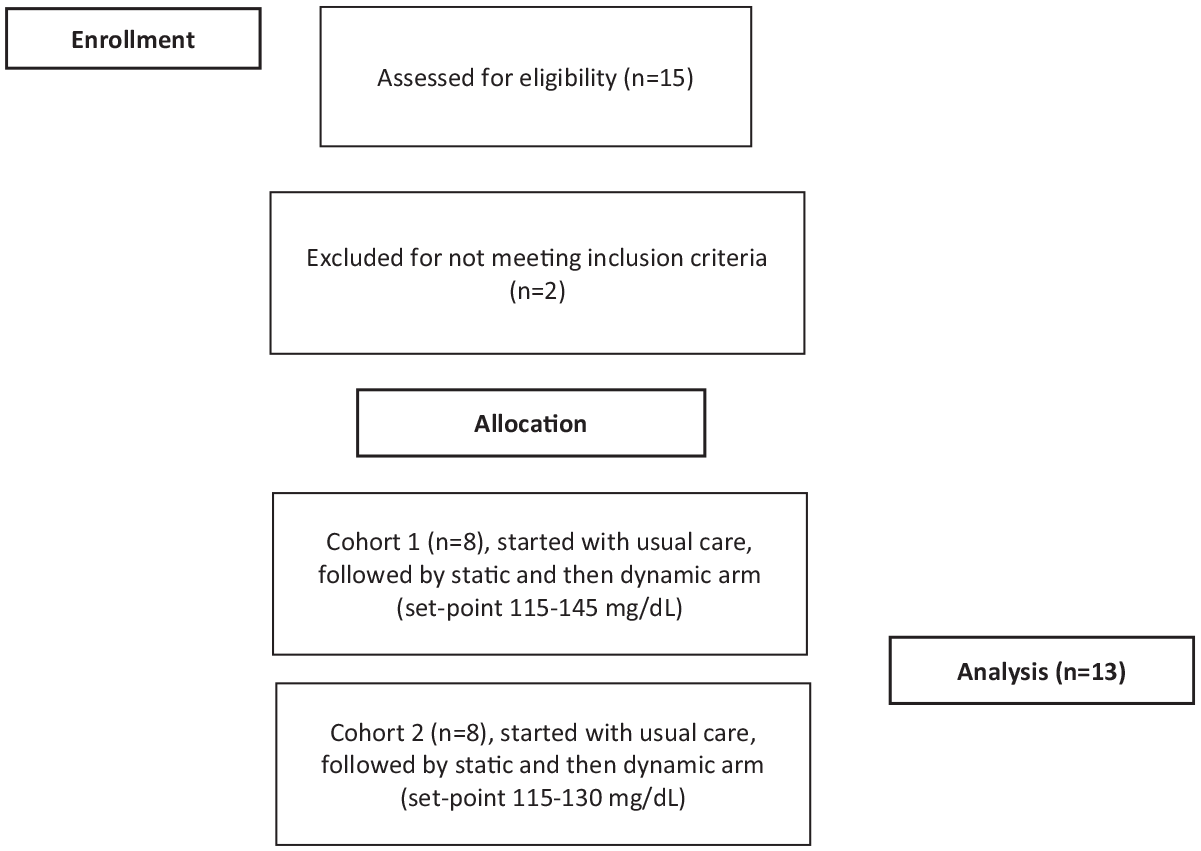
Consort flow diagram.
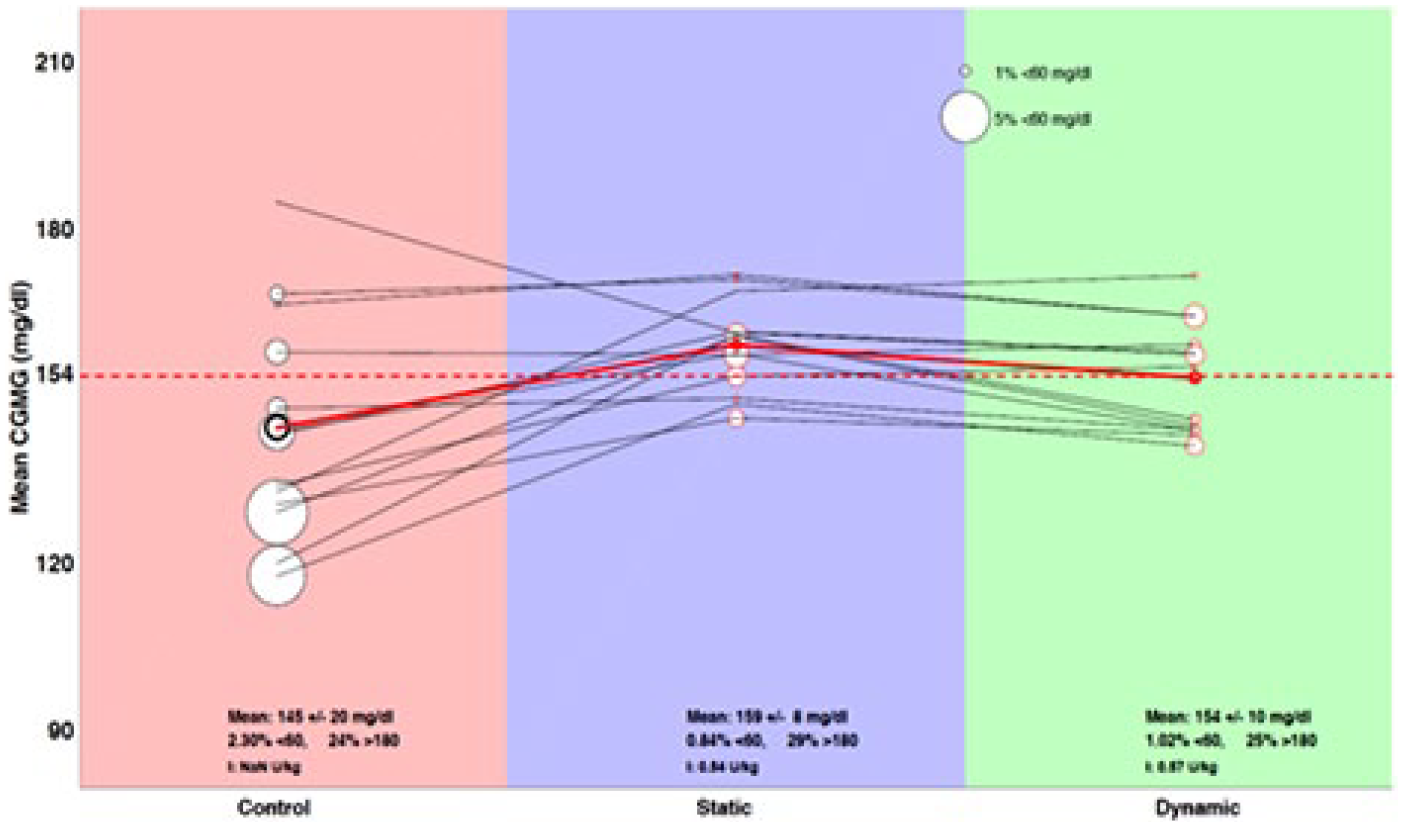
Plot shows mean CGM glucose concentration and hypoglycemia for each of the 13 participants across the three study arms (usual care, static set-point, and dynamic set-point). The diameter of each circle is proportional to the percentage of time that the user spent with a CGM glucose level <60 mg/dL. The solid red line represents the mean for all the participants. The dashed horizontal red line shows a mean CGM glucose level of 154 mg/dL.
CGM Metrics for the Three Arms.
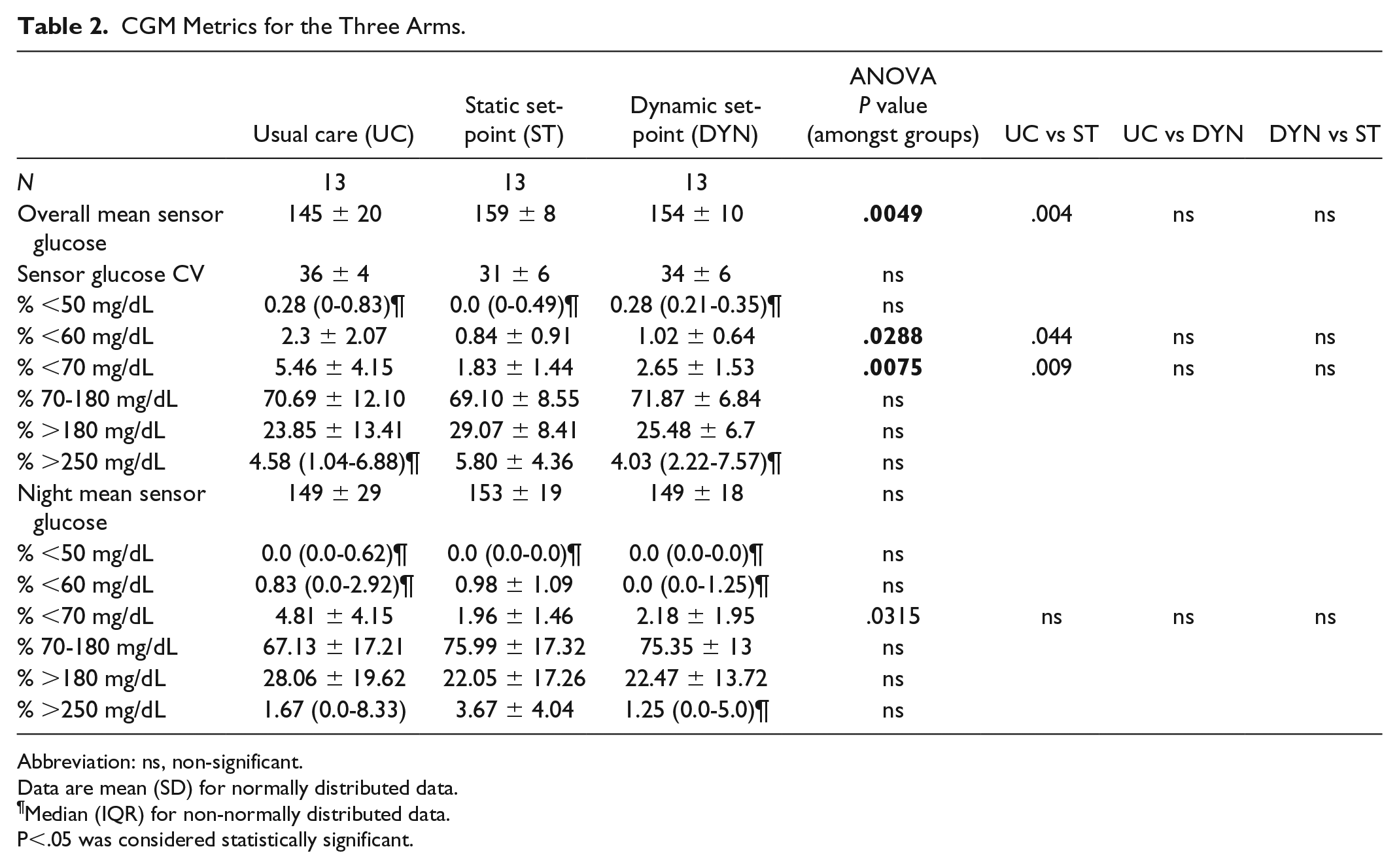
Abbreviation: ns, non-significant.
Data are mean (SD) for normally distributed data.
Median (IQR) for non-normally distributed data.
P<.05 was considered statistically significant.
Summary glycemic control results are shown in Table 2 and Figure 2. There was a significant increase in the mean CGM glucose level in the BP arm relative to the usual-care arm in the case of the static set-point (mean: 159 ± 8 vs 145 ± 20 mg/dL,
Results from HF testing revealed no significant changes from study enrollment to completion on all five surveys administered (
HF Testing Results.
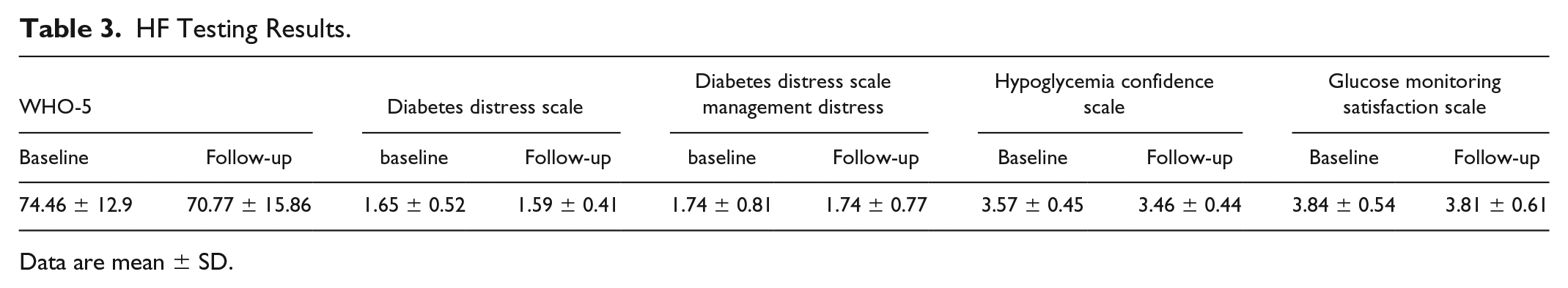
Data are mean ± SD.
Discussion
This was the first home-use study testing the insulin-only configuration of the BP system; all previous studies tested the bihormonal configuration of this system. The primary aim of this pilot study as an initial step of the future randomized control trials was to ensure safety, and to that end, no severe hypoglycemic or severe hyperglycemic events occurred. In the interest of safety, this first home-use study used a conservative set-point of 130 mg/dL, which was 30 mg/dL higher than the set-point previously used with the bihormonal configuration.2-4 This allowed us to safely gather preliminary data on the efficacy of the insulin-only configuration of the BP. A lower set-point of 115 mg/dL was used in only one subject who had an average daily glucose >180 mg/dL and had no glucose values <65 mg/dL on the second day of using the system with a set-point of 130 mg/dL. We also tested a dynamic set-point mode, where the BP autonomously adjusted the set-point within a prescribed range based on the subject’s risk of hypoglycemia as assessed autonomously by the device based on the subject’s real-time CGM data. In terms of both mean CGM glucose and percentage of time spent <60 mg/dL, the static set-point of 130 mg/dL performed similarly to the dynamic set-point of 110 to 130 mg/dL over the last five days of these two study arms.
There was a wide range in the time spent in hypoglycemia during the usual-care arm (0.6%-15%), and, by comparison, the BP provided a significant reduction in hypoglycemia during both days and nights relative to usual care. The mean CGM glucose was higher in the dynamic and static set-point BP arms during the day relative to usual care; however, the time spent in target range (70-180 mg/dL) remained similar to the usual-care arm. This might partly be due to a study effect, since baseline A1c would not have predicted such a low mean glucose and such a high percentage of time in range for the usual-care arm. Because the system did not require carbohydrate counting, adjustment in basal infusion rates, or determination of prandial or correction doses of insulin, participants experienced a lower burden of care throughout the day and night.
Although this was only a pilot study, and the BP used a conservative glucose target, it is interesting to compare our results with those of recently published insulin-only closed-loop studies that followed a similar study design. 1 Our study population was well controlled at baseline and during usual care before initiation of closed-loop therapy. Relative to other studies, our subjects at baseline were one of the better controlled groups with an average A1c of 7.2%, although most subjects entering these early closed-loop studies tend to have lower A1c levels (average 7.9%, range 7.0%-9.0%)5-13 than is typical for patients in these age groups (average 8.4%). 14 The mean CGM glucose obtained with the insulin-only configuration of the BP was 154 mg/dL for the dynamic set-point, which is consistent with what has previously been reported for 10 insulin-only closed-loop studies (mean 152 mg/dL, range 143-157 mg/dL). 1 In the case of the static set-point, the mean CGM glucose was slightly higher, at 159 mg/dL, although not statistically significant. This is not surprising given that the static set-point used in this first feasibility study was deliberately chosen to be conservative to reduce the risk of hypoglycemia. Preliminary analysis of a concurrent study testing a range of static glucose targets of the insulin-only and bihormonal configurations of the BP revealed that lower mean CGM glucose levels corresponded to lower fixed set-points over a range of 110 to 145 mg/dL. 15
The average time within 70 to 180 mg/dL was 69% and 72% for the static and dynamic set-points of the insulin-only configuration of the BP, respectively. These results are similar with another 10 studies 1 (mean 72%, range 68%-79%) as well as with the results reported by a meta-analysis of 22 studies testing a variety of insulin-only artificial pancreas systems (average 68%, range 64%-72%). 1
The percentage of time <70 mg/dL (1.8% for static and 2.7% for dynamic set-points) was also similar to those studies reported in the same meta-analysis of 22 insulin-only artificial pancreas systems (mean 2.5%, range 1.0%-4.0%). 1 When compared to other insulin-only studies, our findings support what previous studies have shown; overnight closed-loop control allows for reduction in nocturnal hypoglycemia, improved time spent in target range, and a reduction in hyperglycemia5,9,10,16-18(Supplemental Table S3). Importantly, the insulin-only configuration of the BP achieved results that were consistent with other insulin-only systems in terms of mean CGM glucose, mean time in range, and mean time <60 and <70 mg/dL, but, unlike other insulin-only systems, did so without the need for carbohydrate counting and with no system knowledge of the subject’s TDD of insulin, basal insulin infusion rates, insulin sensitivity factors, or carbohydrate-to-insulin ratios. The ability of the system to perform as autonomously and as well as it did, and with virtually no input from the subject or clinical study staff, has important implications for the promise of this system, in particular, in terms of accessibility and adoptability of the technology across a broad range of patients and providers.
In sum, the insulin-only configuration of the BP system performed similarly to other published insulin-only systems, achieving mean glucose levels that would be consistent with current treatment recommendations and with minimal hypoglycemia. Further studies comparing the insulin-only and bihormonal configurations of the BP are underway. 15 These studies are intended to help elucidate optimal set-points for both insulin-only and bihormonal configurations of the BP and to identify the ranges over which these set-points should be allowed to vary. Further studies are needed to determine if a dynamically adjustable set-point will offer practical clinical advantages over user- and clinician-determined static set-points.
Supplemental Material
Ekhlaspour.Supplement – Supplemental material for Feasibility Studies of an Insulin-Only Bionic Pancreas in a Home-Use Setting
Supplemental material, Ekhlaspour.Supplement for Feasibility Studies of an Insulin-Only Bionic Pancreas in a Home-Use Setting by Laya Ekhlaspour, Laura M. Nally, Firas H. El-Khatib, Trang T. Ly, Paula Clinton, Eliana Frank, Molly L. Tanenbaum, Sarah J. Hanes, Rajendranath R. Selagamsetty, Korey Hood, Edward R. Damiano and Bruce A. Buckingham in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
We thank the volunteers, their families, and their designated contacts for their participation. We thank Eli Lilly, USA, for donating the glucagon used in this study. Tandem Diabetes Care (San Diego, CA, USA) and Dexcom (San Diego, CA, USA) provided access to their technology. We thank the study staff Suzette Reuschel-DiVirgilio, Liana Hsu, and Darrell Wilson for making the study possible. Some of the data were presented at the American Diabetes Association Scientific Session, 2015, in New Orleans.
Contributors
BAB, FHE-K, and ERD designed the study. BAB, LMN, TTL, MLT, KH, SJH, PC, and EF performed the experiments and supervised the clinical study. BAB, ERD, FHE-K, RRS, and LE analyzed, interpreted the data, and wrote the manuscript. FHE-K and ERD designed and built the closed-loop control algorithm and the BP device. All of the authors reviewed the draft report for content.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Laya Ekhlaspour, Laura Nally, Eliana Frank, Paula Clinton, Sarah Hanes, Trang Ly, and Molly L Tanenbaum have no conflict of interests. Korey Hood receives research support from Dexcom, Inc, for an investigator-initiated study and has received consulting fees in the past year from Bigfoot Biomedical, Insulet, J&J Diabetes Institute, and the Lilly Innovation Center. Firas El-Khatib is affiliated with the Biomedical Engineering Department at Boston University, and an officer, co-founder, and equity holder in Beta Bionics, Inc. Rajendranath Selagamsetty is a PhD candidate in the Biomedical Engineering Department at Boston University and is also an employee of, and holds options to purchase stock in, Beta Bionics, Inc. Edward Damiano is a professor in the Biomedical Engineering Department at Boston University, and an officer, director, co-founder, and equity holder in Beta Bionics, Inc. El-Khatib, Selagamsetty, and Damiano are inventors on patents related to the bionic pancreas technology, which are assigned to Boston University and licensed to Beta Bionics, Inc. Bruce Buckingham is on medical advisory boards for Novo Nordisk and Convatec, has received honorarium from Insulet, Medtronic, and Tandem Diabetes, and has received research support from Medtronic, Dexcom, Tandem, Xeris, and Convatec.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by The Leona M. and Harry B. Helmsley Charitable Trust.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
