Abstract

Data on use of hybrid closed-loop (HCL) therapy during labor and delivery (L&D) are sparse. We conducted a retrospective chart review of six individuals with type 1 diabetes using Tandem Control IQ (CIQ) during pregnancy, including L&D. We obtained hospital records to verify time of hospital admission for L&D and time of infant delivery.
Pregnant individuals used CIQ with sleep mode (SM) 24 hours/day, which has a glucose target range of 112.5 to 120 mg/dL (6.3-6.7 mmol/L). They were counseled on use of CIQ during L&D, if allowed by the obstetric and hospital teams. Individuals were instructed to use SM the first 4 to 6 hours of spontaneous labor or at induction initiation then remain in SM until delivery or switch to exercise mode (glucose target range of 140-160 mg/dL [7.8-8.9 mmol/L]) if experiencing frequent hypoglycemia. After delivery, they were instructed to use postpartum doses recommended by their provider. For scheduled cesarean sections, individuals were instructed to run CIQ and switch to postpartum pump settings just before surgery. After delivery, for all L&D scenarios, individuals were counseled to skip the first carbohydrate meal bolus and change the SM schedule to be active at nighttime.
Continuous glucose monitoring (CGM) data were obtained from Tandem’s t:connect® software and were analyzed for glycemic metrics of mean sensor glucose (mg/dL), pregnancy-specific time-above-range (psTAR, >140 mg/dL [>7.8 mmol/L]), pregnancy-specific time-in-range (psTIR, 63-140 mg/dL [3.5-7.8 mmol/L]), and pregnancy-specific time-below-range (psTBR, <63 mg/dL [<3.5 mmol/L]).
The average admission interval was 18.7 ± 13.9 hours, during which 66.7% of individuals (4/6) met all pregnancy-specific glucose targets. The cohort’s mean time spent in psTIR was 79.5% ± 16.9% with an average of <1% of values in the psTBR (Table 1). All but one individual used SM at the time of delivery. After delivery, two individuals turned off SM and the remaining three stayed in SM through discharge.
Control IQ Use During Labor and Delivery.
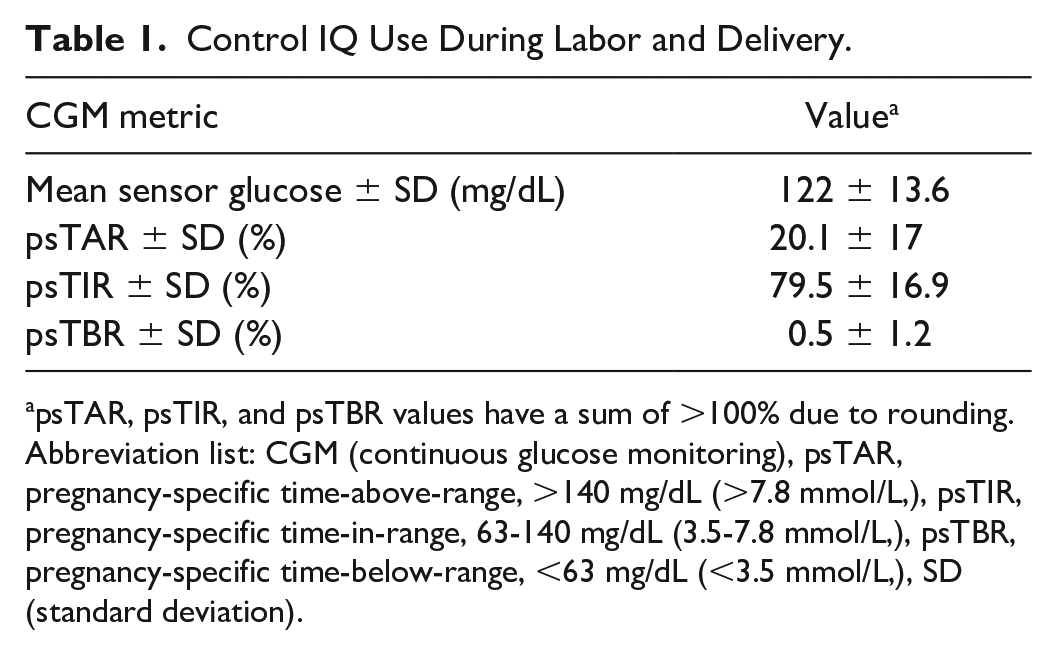
psTAR, psTIR, and psTBR values have a sum of >100% due to rounding.
Abbreviation list: CGM (continuous glucose monitoring), psTAR, pregnancy-specific time-above-range, >140 mg/dL (>7.8 mmol/L,), psTIR, pregnancy-specific time-in-range, 63-140 mg/dL (3.5-7.8 mmol/L,), psTBR, pregnancy-specific time-below-range, <63 mg/dL (<3.5 mmol/L,), SD (standard deviation).
Analysis of L&D data collected from the AiDAPT randomized controlled trial, CamAPS® FX versus standard insulin delivery, has not been published. 1 Data from the two precursor studies to this system were pooled by Stewart et al to analyze glycemic indices for individuals choosing to use HCL therapy during L&D (n = 27/32, 84.4%). 2 The mean glucose level among participants was 124 mg/dL (6.9 mmol/L). The median psTIR ranged from 77% to 84%, depending on mode of delivery. 2 Wang et al reported a case series of four pregnant individuals who used CIQ during gestation and L&D. 3 Three individuals in SM had psTIR glycemic levels during labor or prior to cesarean section, while one had above-target glucose levels during labor and switched from SM to exercise mode upon pushing. 3 Continuous glucose monitoring metrics in these publications were similar to those observed in this case series.2,3
Data from these small studies of pregnant individuals with type 1 diabetes suggest that HCL wear may be safe and effective for use in the L&D setting. More research is needed to further explore HCL therapy for individuals admitted for L&D in terms of safety, efficacy, cost-effectiveness, and patient-reported outcomes.
Footnotes
Acknowledgements
The authors thank the participants of this study for contributing their data.
Abbreviations
CIQ, control IQ; HCL, hybrid closed-loop; L&D, labor and delivery; psTAR, pregnancy-specific time above range; psTBR, pregnancy-specific time below range; psTIR, pregnancy-specific time in range; RCT, randomized controlled trial; SM, sleep mode.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SP reports research funding from Dexcom Inc, Eli Lilly, JDRF, Leona & Harry Helmsley Charitable Trust, NIDDK, and Sanofi U.S. Services; research support from Diasome Pharmaceuticals Inc, Medtronic MiniMed Inc, Sanofi U.S. Services; contributing writer: diatribe; Medical Advisory Board: Medtronic MiniMed Inc (2020). MPK, TLB, and JKS report no conflicts of interest
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
