Abstract
Background:
Time at high risk of hypoglycemia (THRH), 3.9 to 5.6 mmol/L, is a continuous glucose monitoring (CGM)-based metric recommended for reporting in hospitalized patients. This study aims to validate THRH as a predictor of hypoglycemia.
Methods:
The CGM data from 166 non-intensive care unit (non-ICU) inpatients with type 2 diabetes from the DIATEC trial were analyzed. All participants received basal-bolus insulin therapy. Of these, 82 were monitored with point-of-care glucose testing and blinded CGM, while 84 had open CGM. Linear and negative binomial regression analyses assessed the relationship between THRH and time below range (TBR) (<3.0 mmol/L, 3.0-3.9 mmol/L, and <3.9 mmol/L) and hypoglycemic events. Analyses were conducted for day (07:00-23:00), night (23:01-06:59), and 24-hour periods.
Results:
For CGM-monitored patients, every 10%-point increase in THRH was associated with a 0.13%-point increase in TBR (<3.0 mmol/L) (95% confidence interval [CI] = 0.06-0.21), 0.66%-point increase in TBR (3.0-3.9 mmol/L) (95% CI = 0.47-0.86), and 0.74%-point increase in TBR (<3.9 mmol/L) (95% CI = 0.51-0.97), all P < .001. A THRH threshold below 50% was linked to a TBR <3.9 mmol/L of less than 4%, as recommended. Similar results were observed during both day and night analyses and for point-of-care monitored patients, also for hypoglycemic events.
Conclusions:
The THRH is strongly associated with hypoglycemia in non-ICU hospitalized patients with type 2 diabetes on basal-bolus insulin. Aiming for THRH below 50% aligns with the recommended TBR target of <3.9 mmol/L below 4%, supporting THRH’s role in guiding hypoglycemia prevention strategies.
Keywords
Introduction
Hypoglycemia remains a significant concern in the management of hospitalized patients with diabetes, particularly those receiving insulin therapy. 1 Continuous glucose monitoring (CGM) has become an invaluable tool for closely tracking glucose levels in real-time in the outpatient setting, and CGM efficacy and safety are currently being assessed in inpatient settings.2,3 Recently, a new CGM-based metric, time at high risk of hypoglycemia (THRH), has been strongly recommended to be reported in a consensus statement for CGM metrics for inpatient clinical trials. 4 The THRH assesses the proportion of time spent with sensor glucose values between 3.9 and 5.6 mmol/L and has been suggested as indicative of an imminent risk of developing hypoglycemia, prompting therapeutic adjustments. However, the validity of THRH as a standalone metric, as well as its added value to time in range (TIR) 3.9 to 10.0 mmol/L and established hypoglycemia indicators such as hypoglycemic events and time below range (TBR) (<3.0 mmol/L, 3.0–3.9 mmol/L, and <3.9 mmol/L), 4 remains unexplored.
The aim of this study is to evaluate the utility of THRH as a predictor of hypoglycemia.
Methods
We included CGM data from 166 non-intensive care unit patients (non-ICU) with type 2 diabetes treated with a basal-bolus insulin regimen from the DIAbetes TEam and Cgm (DIATEC) trial.2,5 The DIATEC trial is a two-armed, two-site, prospective, randomized, open-label, blinded-endpoint (PROBE) trial conducted at Copenhagen University Hospital—North Zealand and Herlev-Gentofte Hospital. We included patients from both orthopedic, geriatric, pulmonary, and infectious disease departments. The trial was prospectively registered with ClinicalTrials.gov as NCT05803473.
In the DIATEC trial, we hypothesized that diabetes management led by in-hospital diabetes teams with CGM-based insulin titration algorithms would be superior to treatment led by the same diabetes teams and point-of-care (POC)-based insulin titration algorithms, in non-intensive care unit patients with type 2 diabetes. Of these, 82 were managed during hospitalization by POC glucose testing and blinded CGM and 84 with open CGM with hypoglycemic alarm settings at 3.9 mmol/L.
Inclusion criteria were patients with type 2 diabetes, aged 18 years or older, who were expected to stay in the hospital for at least two days following enrolment. This was determined based on the investigator’s clinical judgment, considering the patient’s diagnosis, severity, comorbidities, and other relevant factors. Key exclusion criteria were patients receiving out-of-hospital basal insulin with a duration of action greater than 24 hours (eg, insulin glargine 300 U/mL or insulin degludec), those undergoing systemic glucocorticoid therapy with a prednisone equivalent dose exceeding 5 mg/day, and individuals on dialysis or with an estimated glomerular filtration rate less than 15 mL/min/1.73 m2. Additional criteria are published.2,5
Statistics
We assessed the correlation between TBR (<3.0 mmol/L, 3.0–3.9 mmol/L, and <3.9 mmol/L) and THRH in linear regression analyses and hypoglycemic events and THRH in negative binomial regression analyses. The analysis of the associations between TBR and THRH was conducted separately for all 24 hours of the day, as well as during daytime (07:00 to 23:00) and nighttime (23:01 to 06:59). For the THRH and TBR associations, the THRH variable was scaled by a factor of 10 to facilitate the clinical interpretability of the results, allowing the association between THRH and TBR to be expressed as an increase in TBR for every 10%-point increase in THRH. The associations between hypoglycemic events and THRH were only analyzed for all 24 hours due to the infrequency of hypoglycemic events, which resulted in zero-inflation of the outcome in daytime and nighttime analyses. We assessed whether known risk factors (i.e., age, gender, admission diagnosis [medical or surgical], insulin treatment prior to admission, hemoglobin A1c (HbA1c) levels at admission, estimated glomerular filtration rate [eGFR] at admission, plasma glucose at admission, and duration of diabetes diagnosis) 6 for inpatient hypoglycemia modified the association between THRH and the occurrence of TBR or hypoglycemic events in interaction (subgroup) analyses. Analyses were conducted for patients with blinded CGM and open CGM separately, as CGM alarms and access to non-blinded CGM data may impact the associations between THRH, TBR, and hypoglycemic events. Statistical significance was set at a 2-sided P-value ≤ .05.
Results
Baseline Characteristics
Baseline characteristics from the 166 non-ICU patients with type 2 diabetes are published. 2 In summary, subjects had a mean (SD) age of 76.1 (9.8) years, of which 106 (63.9%) were males. The duration of diabetes was on average (SD) 13.1 (8.7) years. Patients were equally distributed between orthopedic patients (48%) and medical patients (52%), including those with infectious, pulmonary, and geriatric conditions. As outpatients, 86% were on non-insulin antidiabetic medications, while 25% received insulin therapy.
Associations between THRH and TBR as well as THRH and hypoglycemic events are depicted in Table 1. In summary, we observed strong associations between THRH and both TBR and hypoglycemic events in patients monitored by both blinded and real-time non-blinded CGM. None of the potential risk factors for inpatient hypoglycemia—such as advanced age, gender, admission diagnosis (medical or surgical), insulin treatment prior to admission, HbA1c levels at admission, eGFR at admission, plasma glucose at admission, and duration of diabetes diagnosis—had any effect modification on the associations between THRH and TBR or hypoglycemic events (interaction P > .05, data not shown). This suggests that the risk of hypoglycemia associated with high levels of THRH is consistent across all inpatients with type 2 diabetes on a basal-bolus regimen.
Associations Between Time at High Risk of Hypoglycemia (THRH), 3.9 to 5.6 mmol/L, and Three Levels of Time Below Range (TBR) and Hypoglycemic Events.

Abbreviations: TBR, time below range; THRH, time at high risk of hypoglycemia; “–” indicates zero-inflation of the outcome.
Figure 1 depicts the associations between THRH and TBR (<3.0 mmol/L and <3.9 mmol/L) in scatterplots. Figure 1 illustrates that, based on the recommendation for TBR (<3.9 mmol/L) to remain below 4%, 7 THRH should not exceed around 40% for patients monitored by POC glucose testing (blinded CGM) and around 50% for those monitored by open CGM. For the recommendation of TBR (<3.0 mmol/L) to remain below 1%, 7 THRH should not exceed around 60% in both POC glucose testing and CGM-monitored patients.
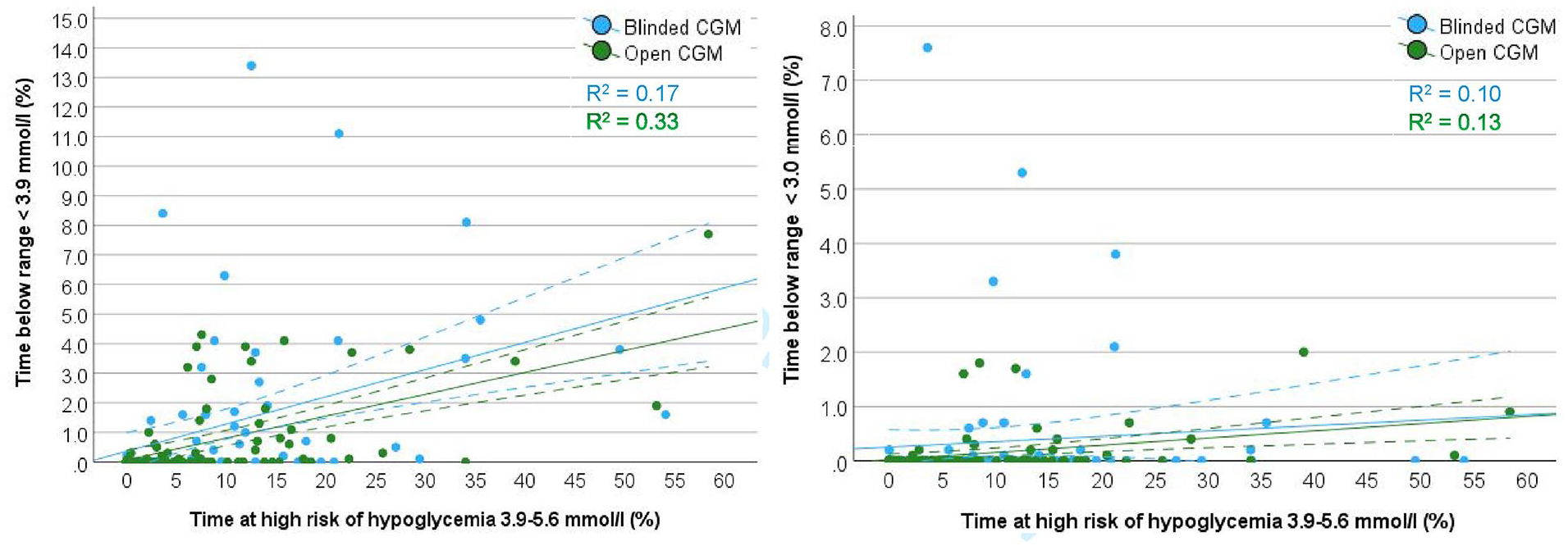
Scatterplots with regression lines (solid lines) and 95% confidence intervals (stippled lines) for the associations between time at high risk of hypoglycemia (3.9-5.6 mmol/L) and time below range <3.9 mmol/L (left panel) and time below range <3.0 mmol/L (right panel) in non-intensive care unit patients with type 2 diabetes monitored by blinded or open continuous glucose monitoring (CGM) during the entire hospitalization.
Discussion
In this study, we validated the newly proposed CGM-based metric THRH as a predictor of hypoglycemia in a cohort of 166 non-ICU patients with type 2 diabetes receiving basal-bolus insulin therapy. Our results demonstrate that THRH is significantly associated with increased TBR and hypoglycemic events, supporting its potential use for predicting hypoglycemia in patients monitored with CGMs with alarms of hypoglycemia <3.9 mmol/L. Our analysis revealed that THRH was most strongly associated with level 1 (3.0-3.9 mmol/L) TBR and hypoglycemic events, with 7-fold and 2-fold higher risks, respectively, compared to level 2 (<3.0 mmol/L) TBR and hypoglycemia. These findings suggest that glucose levels within the THRH range of 3.9 to 5.6 mmol/L may represent a clinically significant warning zone, indicating the need for therapeutic adjustments to prevent imminent hypoglycemia in hospitalized patients receiving insulin therapy. Moreover, THRH could serve as an early warning trigger, even before level 1 hypoglycemia occurs, potentially preventing clinically significant level 2 hypoglycemia. Interestingly, the association between THRH and overall hypoglycemia, measured as TBR <3.9 mmol/L, was reduced by approximately 20% in patients using non-blinded CGM compared to those with blinded CGM. This suggests that with CGM monitoring, it is possible to achieve tighter glycemic control while still minimizing the risk of hypoglycemia.8,9
The associations between THRH, TBR, and hypoglycemic events highlight THRH’s role as a complementary metric to the core metric TIR. While TIR encompasses glucose levels from 3.9 to 10.0 mmol/L, THRH offers additional insight by identifying patients who may have a high TIR but are still at risk of hypoglycemia, as THRH falls within the lower end of the TIR range. This may particularly be of importance for recognizing patients at risk, even when their glucose levels are not overtly low, helping to guide more proactive insulin adjustments. In a study by Mathioudakis et al, the authors investigated whether decreasing insulin doses were made to prevent insulin-associated hypoglycemic events in non-critically ill hospitalized patients monitored by POC glucose testing, focusing on subranges within the range of 3.9 to 5.6 mmol/L, corresponding to THRH. The study found that adherence to local guidelines for reducing insulin doses in patients with borderline hypoglycemia was low. The authors concluded that further studies are needed to better understand the associated risks and to establish more effective therapeutic adjustments for insulin-treated patients experiencing both borderline and overt hypoglycemia. 10 Indeed, the risk of hypoglycemia when glucose levels fall within the THRH range of 3.9 to 5.6 mmol/L is also indirectly accounted for in international treatment guidelines for inpatient diabetes care based on POC glucose testing. For POC-monitored patients, the glycemic target range is not 3.9 to 10.0 mmol/L as for CGM-monitored patients. Instead, for non-ICU patients, most guidelines recommend a glycemic goal of 5.6 to 10.0 mmol/L,1,11 provided it can be achieved without significant hypoglycemia. This POC-based recommendation accounts for THRH by excluding it from the range of 3.9 to 10.0 mmol/L as POC-monitoring has a higher risk of missing hypoglycemic events, which necessitates a more cautious approach in setting the lower limit of the target range to avoid hypoglycemia, compared to CGM. The THRH metric has not been introduced in outpatient CGM metric consensus statements,7,12,13 but may also be of interest for further investigation in outpatients in risk of hypoglycemia, eg, elderly and people with impaired hypoglycemic awareness and/or cognitive impairment. 14 However, THRH 3.9 to 5.6 mmol/L may conflict with the range of time in tight range (TITR) 3.9 to 7.8 mmol/L, which has been introduced for outpatient management, 12 while not being recommended for most inpatients according to current consensus guidelines, although it could be considered for cardiac surgical patients. 4 The significant overlap between the ranges of THRH and TITR creates a paradox, as striving for TITR may inadvertently increase the risk of hypoglycemia. Furthermore, achieving TITR can be challenging even with CGM in an inpatient setting. 15 Therefore, TITR is likely not advisable for most hospitalized patients due to the increased risk of hypoglycemia and related adverse clinical outcomes.
It remains to be explored whether the THRH range of 3.9 to 5.6 mmol/L should be considered a risk zone for hypoglycemia in different populations, such as ICU patients, where treatment regimens and glycemic targets may differ from those in non-ICU settings, 1 or whether a narrower or broader THRH range should be defined in specific populations. In addition, it is uncertain whether the associations between THRH, TBR, and hypoglycemic events hold true for patients treated with various insulin regimens. However, given that the basal-bolus regimen is the most recommended approach and the risk of hypoglycemia associated with high levels of THRH was consistent across all inpatients with type 2 diabetes, regardless of known risk factors for inpatient hypoglycemia, the findings presented here are likely applicable to a broad range of hospitalized patients with diabetes.
Before incorporating the findings above into clinical inpatient diabetes management, it is crucial to recognize that the US Food and Drug Administration (FDA) has not granted official approval for the use of CGM in hospital settings. However, the FDA did not object to its use for managing hospitalized patients during the COVID-19 pandemic. 16 Interest in CGM increased during the pandemic to streamline glycemic monitoring, reduce the spread of infectious particles, and minimize the reliance on personal protective equipment. 17 Despite this growing interest, in-hospital CGM is still considered investigational. This is primarily due to concerns about the accuracy of CGM devices in both non-ICU 18 and ICU setting, 19 as well as the lack of robust evidence supporting its glycemic and clinical benefits over POC glucose testing. 3 In addition, there is no well-defined strategy for CGM’s implementation in inpatient care. 20
Strengths of the study include the inclusion of a relatively large sample size (N = 166), with an even distribution between medical and orthopedic patients, all treated according to international guidelines with a basal-bolus insulin regimen.1,21,22 This increases the external validity of the findings on the associations between THRH, TBR, and hypoglycemic events. However, several limitations should be considered in this work. First, the analysis was conducted in a cohort of non-ICU patients with type 2 diabetes, and the results may not be generalizable to other patient populations, such as those with type 1 diabetes or in ICU patients. Specifically, the mean (SD) age of participants, 76.1 (9.8) years, may limit the generalizability of these findings to younger hospitalized patients with diabetes.
Conclusions
This study validates the utility of THRH as a predictor of hypoglycemia risk in non-ICU hospitalized patients with type 2 diabetes treated with a basal-bolus insulin regimen. The significant associations between THRH, TBR, and hypoglycemic events suggest that THRH could serve as a valuable tool for identifying patients at risk of hypoglycemia. Our data indicate that aiming for a THRH <50% would align with the recommendation of keeping TBR <4% for CGM-monitored patients. Given its potential to inform clinical decision-making, further investigation into the optimal thresholds in different populations and the optimal management strategies for patients with elevated THRH is warranted. This could lead to more personalized and proactive management strategies, ultimately improving patient safety and reducing the incidence of hypoglycemic events and related adverse outcomes in hospitalized patients receiving insulin therapy.
Footnotes
Abbreviations
CGM, continuous glucose monitoring; DIATEC, DIAbetes TEam and Cgm; ICU, intensive care unit; IRR, incidence rate ratio; PROBE; prospective, randomized, open-label, blinded-endpoint; TBR, time below range; THRH, time at high risk of hypoglycemia; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: UPB received speaker fees from Sanofi and Novo Nordisk and has served on advisory boards for Sanofi, Novo Nordisk, and Vertex. Additional authors declare no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
The data sets generated and/or analyzed during the trial are available from the corresponding author upon reasonable request.
