Abstract
In-hospital standard of care for people living with diabetes (PLWD) is based on capillary blood glucose to activate hypoglycemia treatment protocols. PLWD on non-critical care wards often prefer to keep their continuous glucose monitor (CGM) on for their sense of agency. This systematic review assessed the CGM accuracy in the hypoglycemic range for these PLWD. Databases were searched from 2012 to August 2025. We included studies of adult PLWD on non-critical care wards, with CGM levels below 70 mg/dL (3.9 mmol/L) that were compared with paired reference blood glucose levels. Nine included studies reported on 465 hypoglycemic CGM and reference blood glucose pairs. The mean and median absolute relative differences ranged from 7.6% to 53.3%, and from 11.7% to 38.5%, respectively. The methods for pairing CGM with reference blood glucose varied. In eight studies, the mean absolute relative differences between hypoglycemia range CGM and paired reference blood glucose results were greater than 15%. These high mean absolute relative differences suggest that hypoglycemic range CGM results are too inaccurate to guide in-hospital diabetes therapy.
Introduction
In a hospital, the standard of care for people living with diabetes (PLWD) is for nurses to check the capillary blood glucose (CBG) with point-of-care testing glucose meters that are approved for hospital use.1-6 If the hospital-grade glucose meter result does not match with PLWD’s clinical status, then repeat point-of-care testing is advised, and also consider sending for measurement at the hospital laboratory.1-6 The generally-accepted reference standard for blood glucose (BG) is a venous blood draw that is measured at an accredited laboratory (LabBG). 7
For outpatient settings of PLWD self-monitoring, transcutaneous continuous glucose monitors (CGMs) are applied to the skin and measure the subcutaneous tissue interstitial glucose with a reader device or a mobile application downloaded to a cellphone.8-12 Depending on the type of CGM sensor, the interstitial glucose level is measured every one to five minutes.1,8-15 Therefore, CGMs have the potential to detect hypoglycemia episodes better than the point-of-care testing glucose meter protocols which usually only test before meals and at bedtime.1,12-15 Due to their continuous nature, CGMs measure glucose between meals and overnight when glucose meter point-of-care testing is not usually performed.1,12-15 CGMs have algorithms that show a trend arrow forecasting the rate of change for every interstitial glucose level, and provide 24-hour glucose profiles.8,9
Accuracy comparisons for CGMs will pair a CGM interstitial glucose result with a reference blood glucose (CBG or LabBG) result.8,9,16,17 If the BG is in a steady state, the paired interstitial CGM measure can be taken at the same time as the reference BG. However, when there is a change in the blood glucose level, this is first identified by the CBG or LabBG. CGM lag time is a phenomenon whereby the change in blood glucose level takes time for the glucose to diffuse from the blood into the interstitial fluid where it will then be reflected in the CGM measurement.8-11,16-20 Therefore, if the BG level is in a state of change, then a CGM level would be paired with a preceding reference BG level.16-20
The reported lag time for CGMs varies based on the brand and model. A recent study of a popular CGM in adults showed a mean lag time of 3.5 minutes (standard error 0.13 minutes), 10 which is often reasonably interpreted as up to five minutes covers around 95% of lag times. A study of lag time for another popular CGM in adults showed a mean lag time of 2.4 minutes (standard deviation 4.6 minutes). 11 With this larger standard deviation, a reasonable interpretation would be mean plus two standard deviations, rounded up to ~ 15 minutes to cover ~ 95% of lag times.
Currently, inpatient policies require clinical decisions to be based on CBG using hospital-grade point-of-care testing glucose meters.1,8,9,12,16,17,20 There was a transient approval period from health authorities allowing CGM use for inpatients during the COVID-19 pandemic.15,20 After the pandemic, in-hospital CGM use is not approved by regulatory authorities in the United States or in Canada.1,15,20 Nevertheless, because many inpatient studies have shown positive effects for health care teams (e.g. saving nursing time and resources), reducing hypoglycemia episodes, and acceptability from PLWD,9,12-15,20 adjunct use of CGM with the appropriate hospital protocols is recommended by clinical care guidelines.1,12
A critical aspect of whether CGMs can replace CBG in hospitals is the accuracy of CGMs in the hypoglycemia range (below 70 mg/dL; 3.9 mmol/L). Commonly-used inpatient hypoglycemia treatment protocols are activated at CBG below 70 mg/dL (3.9 mmol/L) 1 usually in United States hospitals due to mg/dL units, and below 4.0 mmol/L4-6 (72 mg/dL) in countries using the International System of Units (mmol/L). A high accuracy of CGM levels in the hypoglycemia range is important for nurses to initiate timely treatment of hypoglycemia.
The PLWD who are admitted to a hospital’s non-critical care wards are most likely to want to keep their personal CGM on as their preferred method for glucose monitoring. These non-critical care ward PLWD strongly favor their CGM for glucose trend prediction, alarms, and sense of agency that they can self-monitor.21,22 Another practical consideration is the cost of CGMs that has a duration of 10 to 15 days, which would be money wasted if taken off prematurely. Also, in-hospital care can include settings where the CGM manufacturers have not tested the sensors and readers. Current common clinical practice includes the removal of CGMs prior to magnetic resonance imaging, and surgeries with cauterization (diathermy). 15
The objective of this systematic review is to determine the accuracy of CGMs in the hypoglycemia range for PLWD when they are hospitalized in a non-critical care ward.
Methods
Inclusion criteria:
Exclusion criteria:
The study population focused on a medical condition for which CGMs are not approved for use by the manufacturer, such as PLWD who are on dialysis.
Not a full-text article.
Not a primary research study.
Published in a language other than English or French.
We searched the Medline, Embase, and Web of Science databases for studies that evaluated the accuracy of CGM in inpatient settings, and published in English or French, from 1 January 2012 to 18 August 2025. The search strategy combined the Medical Subject Headings term “continuous glucose monitoring” with text words such as “flash glucose monitoring” and “CGM.” These results were intersected with Medical Subject Headings terms “inpatients” and “hospitals”, and supplemented by text word searches for “inpatient*” and “hospital*" to capture relevant literature comprehensively.
Two authors independently completed the literature search, results screening, and data extraction. Any differences were discussed amongst the authors, and resolved by reaching consensus. The Newcastle-Ottawa scale 23 was applied for the risk of bias assessment.
The accuracy metric for the CGM results and its paired reference BG result that the studies reported was the absolute relative difference (ARD). The ARD calculation is the numerator, comprised of the absolute difference between CGM result and its paired reference BG result, divided by the denominator, which is the reference BG result.8-11,24 The higher the ARD, the lower the accuracy of the CGM level compared to the reference BG result. Some studies reported the mean ARD (MARD), or the median ARD (med-ARD), or both MARD and med-ARD. The effect measures were the standard deviation (SD) for the MARD, and the interquartile range (IQR) for the med-ARD for the pairs of CGM and reference BG results. Data synthesis was planned if there were studies that used the same CGM brand/model and the same type and pairing for the reference BG results. We did not expect the included studies to be biased or have uncertainty because the intervention and comparator were measured CGM and CBG or LabBG levels. The outcome of interest was well-defined accuracy metrics.
Results
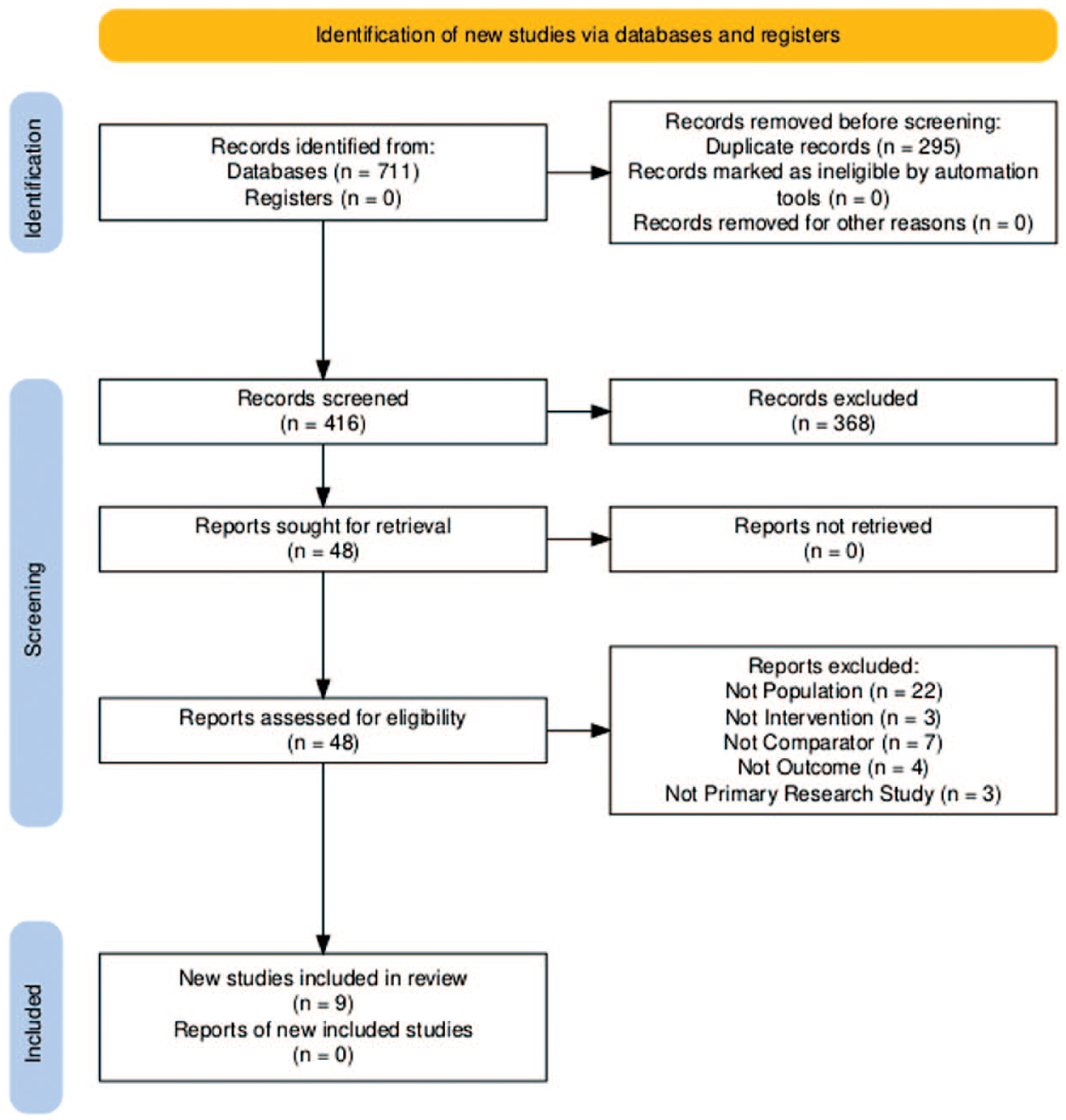
Figure 1 presents the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) Protocol25,26 flow chart. Nine studies were included.19,27-34 Altogether, data were extracted for 465 CGM hypoglycemic results and their paired reference BG (Table 1).
Data Extracted From the Nine Included Studies.
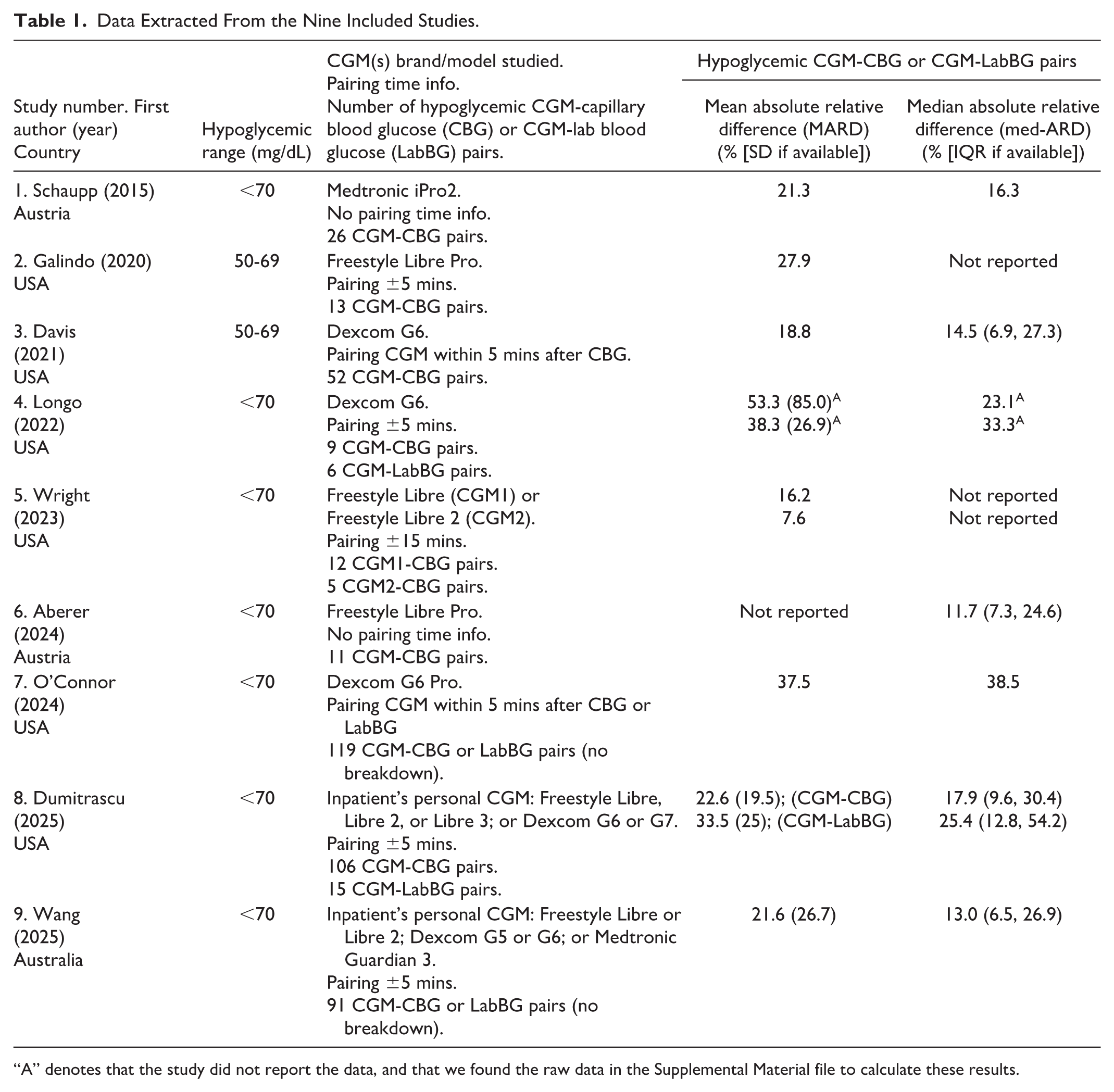
“A” denotes that the study did not report the data, and that we found the raw data in the Supplemental Material file to calculate these results.
Pairing of CGM Result with Reference BG Result
Twenty-one CGM results were paired with reference LabBG results. Two hundred thirty-four CGM results were paired with reference CBG results. Two hundred ten CGM results were paired with a reference BG that was either CBG or LabBG.
Mix of PLWD in Critical Care (Intensive Care Unit) and Non-Critical Care Wards
For both Longo et al 30 and Wang et al 19 studies, the proportion of the hypoglycemic range CGM-CBG or CGM-LabBG pairs that occurred in the PLWD admitted to non-critical care wards was not reported.
Accuracy Metric Reported in all Included Studies
We based our cutoff of mean or median ARD >15% on the following: (1) Bellido et al 15 recommended that for CGMs used in-hospital, aim for MARD <14%, and (2) the accuracy metric referred to as “15/15,” which recommends for CGM interstitial glucose <100 mg/dL (5.6 mmol/L) to be ±15 mg/dL (0.8 mmol/L) difference when compared with its paired reference BG.8-10
Eight studies19,27-31,33-34 reported MARD >15%: their MARD range was 16.2% to 53.3% with SD range (in those that reported SD) of 19.5% to 85.0%.
Three studies19,29,32 reported med-ARD below 15%: their med-ARD range was 11.7% to 14.5%, with IQR between 6.5% and 27.3%. Four studies27,30,33,34 reported med-ARD >15%: their med-ARD range was 16.3% to 38.5%, and IQR was only presented in one study. 34
Four studies reported both MARD and med-ARD >15%.
Risk of Bias Assessment
The included studies had a low risk of bias (Supplemental Table 1).
Results of Syntheses, Reporting Biases, and Certainty of Evidence
Data syntheses were not performed because the included studies differed in the CGM brands/models, and parameters for pairing CGM results with reference BG results.
Reporting biases are also less applicable because the studies’ interventions (CGM brands/models, and pairing with reference BG results) were different. Since all results are measured from CGMs or CBG meters or laboratory testing, the certainty of the evidence is high.
Discussion
General Interpretation of Results
For the hypoglycemic range, the mean and median ARD were high (mostly > 15%), which reflects a low level of accuracy of CGM results compared to their paired reference BG results. Up to now, there is a lack of published guidelines on the acceptable MARD for CGM in-hospital use. We based our MARD >15% cutoff on Bellido et al 15 which recommended to aim for in-hospital MARD <14% for CGM vs reference BG.
Notably, the included studies were designed to evaluate the accuracy of CGM results over the full glycemic range: hypoglycemia, normoglycemia, and hyperglycemia. The mean ARD and the median ARD are usually lowest for the normoglycemic range. Therefore, the full glycemic range mean or median ARD tends to show better accuracy than the hypoglycemic range.
Overall, the 465 hypoglycemic range CGM-reference BG pairs were only a very small percentage of the CGM-reference BG pairings in the included studies. The nine included studies19,27-34 spanned a range from 808 to 6648 CGM-reference BG pairings. Therefore, the small sample size of hypoglycemic CGM-reference BG pairings could have limited the power to detect adequate MARD.
For CBG, the included studies used point-of-care testing glucose meters that were approved for hospital use. For LabBG, the included studies reported that testing was performed in the hospital laboratory. Although the MARD for CGM-CBG pairings could be higher than the MARD for CGM-LabBG pairings, 35 our included studies likely did not provide enough data to assess this. This could be due to our focus on hypoglycemic CGM-reference BG pairings, and the limited sample size. Furthermore, the reference BG in the included studies (CBG or LabBG) differed from the YSI 2300 Stat Plus glucose analyzer, which is the usual method for the reference BG in the accuracy studies listed by the CGM manufacturers in their product monographs.10,11
The Pairing of CGM Result with Reference BG
The five studies that included the pairings of CGM result with a reference BG within five to 15 minutes (before or after) would be best interpreted in the context of the BG being in a steady state.
If the BG is in a state of change, then for the change in BG to show in the CGM interstitial glucose level, there is a known physiologic lag time. Different CGM brands and models have their own algorithms that yield variability in their respective reported CGM results’ lag time compared with reference BG. In a state of BG change, we would expect for a CGM result to be paired with a reference BG at a time preceding the CGM result. Two studies29,33 had a pairing method that incorporated a potential state of BG change (hence not in a steady state) by pairing the CGM result with a preceding reference BG. Wang et al 19 also performed a special “time offset analysis” for their data, which showed an “overall CGM sensor lag of 5 minutes”.
Limitations
Our search strategy would have missed literature that were only in abstract form, conference proceedings, and articles published in languages other than English or French. For our inclusion criteria, it is possible that the included studies had PLWD who had medical conditions for which the CGM manufacturers have not recommended CGM use, such as PLWD on dialysis. Another limitation is that data syntheses could not be performed because the included studies differed in the CGM brands/models, lacked granular data, and differed in their parameters for pairing CGM results with reference BG results.
Implications of Review Findings
The main implication of this systematic review is that for PLWD on non-critical care wards, the accuracy is rather low for CGM results in the hypoglycemic range, when compared with paired reference BG results (MARD >15%).
Current consensus guidelines recommend using CBG for the hypoglycemia range. 12 This needs to be clearly explained to the many PLWD who strongly prefer to keep their personal CGM on during their hospital stay. Furthermore, CGM use is not advised for PLWD with severe hypoglycemia (<40 mg/dL), severe hyperglycemia (>500 mg/dL), or during a period of rapid change in glucose levels. 12 CGMs used in the outpatient settings had overall MARD that were closer to 10%.10,11 The hypoglycemic range MARD was > 15% in these included inpatient studies. It is unclear why such discrepancies are present, and CGM manufacturers should further investigate these discrepancies before CGMs are approved for glucose monitoring in the hospital.
Conclusion
For PLWD admitted to non-critical care wards, the mean absolute relative differences between hypoglycemic range CGM results and paired reference BG results were high, at >15%, in eight included studies. Therefore, for in-hospital PLWD, CGM results cannot be relied upon to make therapeutic decisions regarding hypoglycemia. Currently, the successful in-hospital use of CGM was primarily supported by a hybrid model when CGMs were combined with CBG for validation and on demand calibration.1,12,15
Supplemental Material
sj-docx-1-dst-10.1177_19322968251412482 – Supplemental material for Systematic Review of Continuous Glucose Monitor Accuracy in the Hypoglycemia Range for Non-Critical Care Ward Hospitalized People Living With Diabetes
Supplemental material, sj-docx-1-dst-10.1177_19322968251412482 for Systematic Review of Continuous Glucose Monitor Accuracy in the Hypoglycemia Range for Non-Critical Care Ward Hospitalized People Living With Diabetes by Nicole Prince, Timothy Ramsay, Risa Shorr, Rémi Rabasa-Lhoret and Cathy J. Sun in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
ARD, absolute relative difference; BG, blood glucose; CBG, capillary blood glucose; CGM, continuous glucose monitor; LabBG, laboratory-measured blood glucose; MARD, mean absolute relative difference; med-ARD, median absolute relative difference; PLWD, people living with diabetes.
Authorship Contributions
Nicole Prince: Methodology, investigation, data curation, formal analysis, writing – reviewing and editing.
Timothy Ramsay: Methodology, formal analysis, writing – reviewing and editing.
Risa Shorr: Methodology, investigation, writing – reviewing and editing.
Rémi Rabasa-Lhoret: Conceptualization, formal analysis, writing – reviewing and editing.
Cathy J. Sun: Conceptualization, methodology, investigation, data curation, formal analysis, supervision, visualization, writing – original draft preparation, writing – reviewing and editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Prospero Registered Systematic Review
CRD420251117903
Supplemental Material
Supplemental material for this article is available online.
