Abstract
Background:
Current diabetes screening methods are complex, inefficient, and inconvenient, requiring resource-intensive blood sampling. With the increasing prevalence of underdiagnosed type 2 diabetes mellitus (T2DM) worldwide, particularly in low-resource settings and underserved populations, affordable and sustainable mass-screening tools are crucial.
Methods:
The accuracy and safety of the miniaturized near-infrared (NIR), full-spectrum spectroscopy Glyconics-DS System in detecting T2DM risk status was assessed by pooling data from two independent pilot studies: ANODE01 and ANODE02. Rapid NIR assessments of glycated nail keratin in 60 repeated spectral readings of fingernails from individuals with or without T2DM focused on detecting dichotomized diabetes risk status (glycated hemoglobin [HbA1c] <6.5%) based on chemometric prediction models, clinical specificity/sensitivity, and true/false positive outcomes. An HbA1c point-of-care assay served as an internal control.
Results:
Over 12 000 NIR spectral readings were collected in a female-dominant (58.5%), mostly non-smoking (80.0%), diverse cohort of 200 participants (n = 100 with/n = 100 without T2D). The selected chemometrics prediction model on a diagnostic HbA1c cut-off of 6.5% showed a specificity of 92.9% (95% confidence interval [CI] = 88.5-97.4) and a sensitivity of 34.2% (95% CI = 23.4-45.1), with 71.5% concordance. Chemometric predictions were consistent and reproducible with no relevant impact of anthropometric variables, concomitant conditions/medications, smoking status, and number of spectral assessments/nail or hand dominance on NIR assessment. No adverse events or suspected de novo T2D cases were reported.
Conclusions:
This pooled analysis of two independent studies demonstrates the clinical feasibility and high specificity of rapid NIR spectral assessment of T2DM risk, with potential for screening, early detection, and sustainable management across health care settings.
Introduction
Diabetes mellitus (DM) represents a significant health care burden, currently estimated to affect over 830 million of adults worldwide.1,2 Between 2021 and 2045, the greatest increase is predicted in middle- (21.1%) vs low- (11.9%) and high-income (12.2%) countries. 3 Correspondingly, DM-related health care expenditure is estimated to climb from US$966 to US$1054 billion.3,4 Type 2 diabetes mellitus (T2DM) accounts for 90–95% of DM cases, 5 and is a leading cause of cardiovascular mortality and morbidity with reductions in quality of life and expectancy. 6 Race/ethnicity are known risk factors for T2D, with individuals of color at greater risk than white populations, alongside lower education, poverty, and metropolitan area.5,6
Despite its widespread prevalence, T2DM is still vastly underdiagnosed, particularly in low- and middle-income countries where there is often insufficient access to health care and/or lowered capacity in existing health systems. 4 Socio-economic inequalities across the spectrum of care still exist, with low individual socio-economic status and area deprivation—representing individuals with the greatest need—being the least likely to receive appropriate screening and care.7,8 Undiagnosed T2DM can lead to treatment delays, increasing the risk of poor health outcomes, including reduced quality of life, micro-/macrovascular complications caused by uncontrolled blood pressure, lipids, prolonged hyperglycemia, and increased mortality.8,9
Screening can allow early interventions and improve long-term health outcomes and health care costs.10,11 However, current methods are complex, inefficient, inconvenient, and require resource-intensive, invasive blood sampling. 11 While screening programs have been established in many high-income countries, 12 there is high variability in diagnosis rates in those from minority backgrounds 13 or with lower socio-economic status, 14 representing significant health care and economic disparities. Thus, there is a critical need for affordable, rapid, and precise mass-screening tools to combat clinical inertia in identifying undiagnosed T2DM among diverse populations. 12 Such tools should have high specificity to accurately exclude those at low risk, allowing precious clinical resources to be allocated to those requiring further investigation.
While glycated hemoglobin (HbA1c) measurement is the current standard for diabetes diagnosis, newer technologies are in development using photoplethysmography from vascular signals in the fingertip 15 and spectroscopy of glycated proteins in the lip, skin,16,17 eye lenses,18,19 and fingernail clippings.19-22
Chemometric prediction models used in spectroscopy can be calibrated against spectral biomarkers including glycation of nail keratin, indicative of sustained elevated glycemia, and may have potential for swift and sustainable non-invasive mass-screening, diagnosis, or therapeutic monitoring.19-23 Using mathematical calibration of spectral data sets with known properties (“training sets”), predictions can be calculated for samples with new spectra through comparison with these “known (trained)” measurements. As such, the predictive power of a chemometric model may help to detect elevated and normal DM risk in diverse demographics through identification of absorption patterns associated with glycated keratin. 24 Real-time spectral acquisition and potential prediction may improve inertia needed for primary care.
The Glyconics-DS* system (“Glyconics-SX” when developed originally; Glyconics Ltd, UK) is a novel, miniaturized near-infrared (NIR) full-spectrum spectroscopy device intended for T2DM screening by assessing glycated keratin in the middle fingernail as an indicator of risk. Using chemometric models and mathematical calibration of data sets from thousands of spectral readings, this technology allows meaningful dichotomized cloud-based assessment of spectral data generated by a handheld device. 25 Optimization has focused on improving sensitivity and specificity, with a greater emphasis on high specificity due to the system’s intended-use for screening rather than diagnosis. Specificity (true negative rate) denotes how accurately a test measures the absence of a condition avoiding false positives (ie, proportion without T2DM testing negative on a screening test without misclassifying someone as having T2DM). In contrast, sensitivity (true positive rate), intended for diagnostics, measures how accurately a test detects a given condition avoiding false negatives (ie, proportion identified with T2DM who test positive during screening without missing someone with T2DM). 26
The ANODE program was designed to generate initial clinical evidence for the Glyconics-DS* system (spectrometer, cradle, and software for analysis) to identify and validate chemometric prediction algorithms for T2DM risk. We explored safety and accuracy by pooling data from two pilot studies of individuals with or without T2DM from different health care delivery settings: a specialized diabetes research center and a primary care center. The main objective was to demonstrate initial, high-quality spectral assessments indicative of accuracy in distinguishing individuals with or without T2DM, based on known and/or assumed clinical status, controlled by HbA1c measurement.
Methods
Trial Design and Participants
Two identical, independent, single-center, open-label, single-visit, cross-sectional studies were designed to retrieve repeated rapid NIR spectral assessments of glycated keratin of middle fingernails in individuals with or without known T2DM. ANODE01 (NCT05198895) was conducted at the Leicester Diabetes Centre, UK and ANODE02 (NCT05476016) was conducted at the Sant Martí Primary Health Center in Barcelona, Spain. The expected study duration was eight weeks. Both studies were conducted in accordance with the Declaration of Helsinki and Good Clinical Practice requirements, with approval of the protocols and informed consent by an Independent Ethics Committee.
Full inclusion and exclusion criteria are reported in the Supplementary Material and have been published previously. 25 Briefly, adults aged ≥18 years, with known or unknown T2DM status, and two undamaged, intact middle fingernails on both hands from visual assessment were included. Individuals with known medical conditions or structural, deviating, or visually detectable deviations in nail appearance that could impact spectral measurement, or taking systemic/topical medication known to modify the nail surface, structure, or HbA1c value accuracy were excluded. All participants provided written consent.
Trial Procedures and Randomization
Individuals were enrolled 1:1 based on their current status (with or without known T2DM). After confirmation of eligibility and visual fingernail assessment, up to 30 NIR spectral measurements per middle fingernail were taken on both hands (up to 60 measurements), lasting approximately 2 minutes. Participants were seated, with their finger placed on the cradle as instructed. Unique participant identifiers (pre-assigned quick response codes) were used to ensure measurements were recorded and labeled accurately.
After NIR spectral assessments, glycemic levels were confirmed using an HbA1c point-of-care assay with single-use test cartridges based on the manufacturer’s instructions. This served as an internal biomarker control for detecting potential underdiagnosed de novo diabetes and glycemia in both groups. The test was performed on any finger and hand and could be repeated twice (if required) for technical reasons.
Study Endpoints
The primary endpoint was to determine accuracy in detecting dichotomized diabetes risk status (known, clinically diagnosed T2DM vs no known DM) using established cut-off values: HbA1c < or ≥6.5% (48 mmol/mol).
Secondary endpoints included assessment of specificity and sensitivity for chemometric model outcomes (%); calculation of negative and positive predictive values (NPV, PPV) and area under the curve (AUC), with the axis defined by false positive and negative rates for the model; and median (interquartile range [IQR]) HbA1c values in those with/without T2DM as measured by the comparator. The NPV conveys the proportion of individuals with a negative result from a screening test who probably do not have T2DM. This measures the test’s ability to identify individuals without T2DM correctly among all negative results. In contrast, PPV refers to the proportion of individuals testing positive using a diagnostic test who probably have T2DM. This indicates a test’s accuracy in identifying true cases of T2DM among all positive results.
Safety endpoints included adverse events (AEs), adverse device effects (ADEs), serious adverse events (SAEs), serious adverse device effects (SADEs), and all potential cases of suspected de novo DM reported during assessment. Safety outcomes were recorded and listed descriptively.
Exploratory endpoints (not reported) also assessed other HbA1c cut-off values with categorization based on predictions being in one of three HbA1c level classes: <5.7% (39 mmol/mol), <6.0% (42 mmol/mol), or <6.5% (48 mmol/mol) corresponding to normal, prediabetes, and T2DM thresholds defined in current guidelines.11,27 This included categorical and continuous variable assessment over the entire glycemic spectra.
Study Assessments and Chemometric Modeling
The NIR-spectroscopy using the Glyconics-DS system and control point-of-care HbA1c tests using QuoTest finger prick tests were conducted as outlined in the study flow (Supplementary Figure S1). Chemometrics modeling used up to six predictive models that employed partial least squares-discriminant analysis. Models were trained with a data distribution ratio of 80:20 for training and validation, respectively, to determine their potential robustness and insensitivity to baseline patient characteristics (conducted separately to ensure no overlapping between data sets). Validation was completed using seven-fold cross-validation.
Preclinical training and optimization of the models used in ANODE01/02 have been reported previously 25 and are shown in Figure 1. In this clinical analysis, data from the two identically designed pilot studies were pooled as planned, and optimal spectral parameters were assessed for training and to evaluate overall performance in terms of specificity and sensitivity. Further details on chemometric model training and spectral regions for glycated protein analysis are listed in the Supplementary Material.
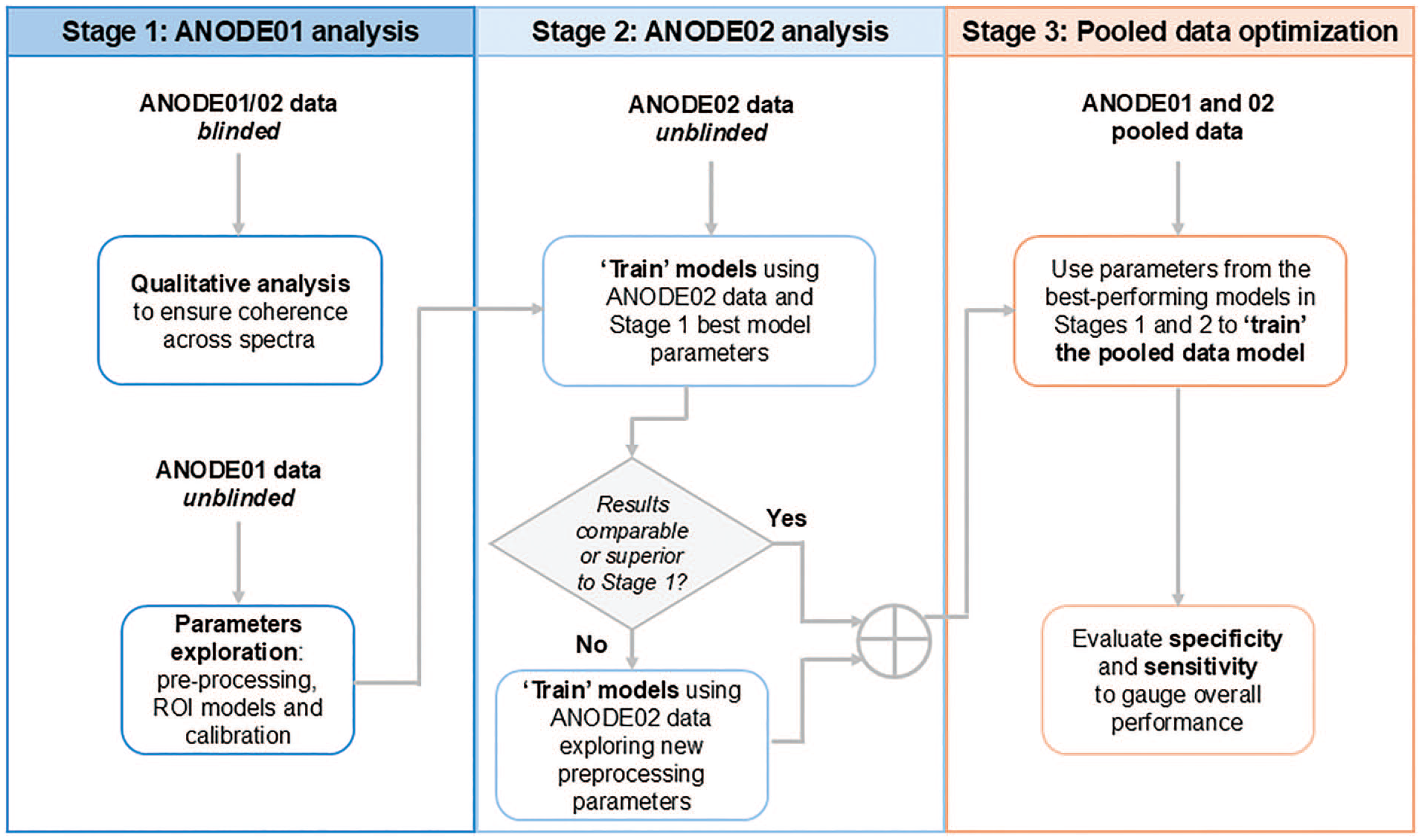
Optimization process for staged data analysis and preclinical validation of the algorithm in the ANODE01 and 02 studies.
Statistical Analysis
All analyses were conducted on the full analysis set (all enrolled participants irrespective of fulfilling other endpoint criteria).
Primary endpoint
Accuracy in detecting DM status (known and clinically diagnosed T2DM vs no known DM) was based on dichotomized outcomes as per the chemometric model using conventional HbA1c measurement as an internal control (< or ≥6.5% for without DM or with T2DM, respectively) compared using McNemar’s test. Accuracy was defined as the overall probability that a participant was correctly classified by the model output to either have (yes) or not to have (no) DM. Dichotomized diabetes risk status (HbA1c < or ≥ 6.5%) was assessed based on chemometric prediction models and the relationship between clinical specificity and sensitivity, true and false positive outcomes. Specific details relating to secondary and exploratory endpoints are included in the Supplementary Material.
Primary and secondary analyses were repeated using the non-dominant hand. All statistical tests were two-sided and performed at an overall significance level of .05.
Safety endpoints
The safety analysis set included all participants who received at least one assessment of their glycemic status (NIR or HbA1c test). This set was used to describe demographic and baseline characteristics, safety parameters, and concomitant medications. Events were reported by subtype (AE/ADE, SAE/SADE), system organ class, and preferred term, according to the Medical Dictionary for Regulatory Activities version 23.0.
Results
Baseline Characteristics
Between August and November 2022, 200 individuals with (n = 100) or without (n = 100) T2DM were assessed in the two studies with data from over 12 000 NIR spectral readings. Demographics showed a female-dominant (58.5%), mostly non-smoking (80.0%) diverse cohort of participants, with a median age of 58 years (range = 19–94), body mass index of 27.3 kg/m2 (range = 17–44), and median HbA1c (%) of 7.0 (IQR = 6.3%-7.8%) and 5.4 (IQR: 5.2–5.6) for those with and without known T2DM, respectively (Table 1).
Demographics and Baseline Characteristics.

Data are median (range), n (%), or median (IQR).
Abbreviations: BMI, body mass index; HbA1c, glycated hemoglobin; IQR, interquartile range; T2DM, type 2 diabetes mellitus.
Chemometric Predictions
The current chemometrics prediction model was shown to have high predictive accuracy for the diagnostic HbA1c cut-off level of 6.5% and a lower cut-off of 6.0%. Using HbA1c cut-offs of 6.5% and 6.0%, the most robust model and algorithm correctly identified a large proportion of participants without T2DM (n = 100) (Figure 2), demonstrating high specificity of 92.9% (95% confidence interval [CI] = 88.5-97.4) and 81.0 (95% CI = 73.4-88.5), respectively. The corresponding NPVs were 71.1% (95% CI = 64.2-78.0) and 69.7% (95% CI = 61.5%-77.8%), respectively (Table 2). The corresponding AUC values for the model at HbA1c cut-off values of 6.5% and 6.0% were 0.79 and 0.80, respectively (Figure 2).
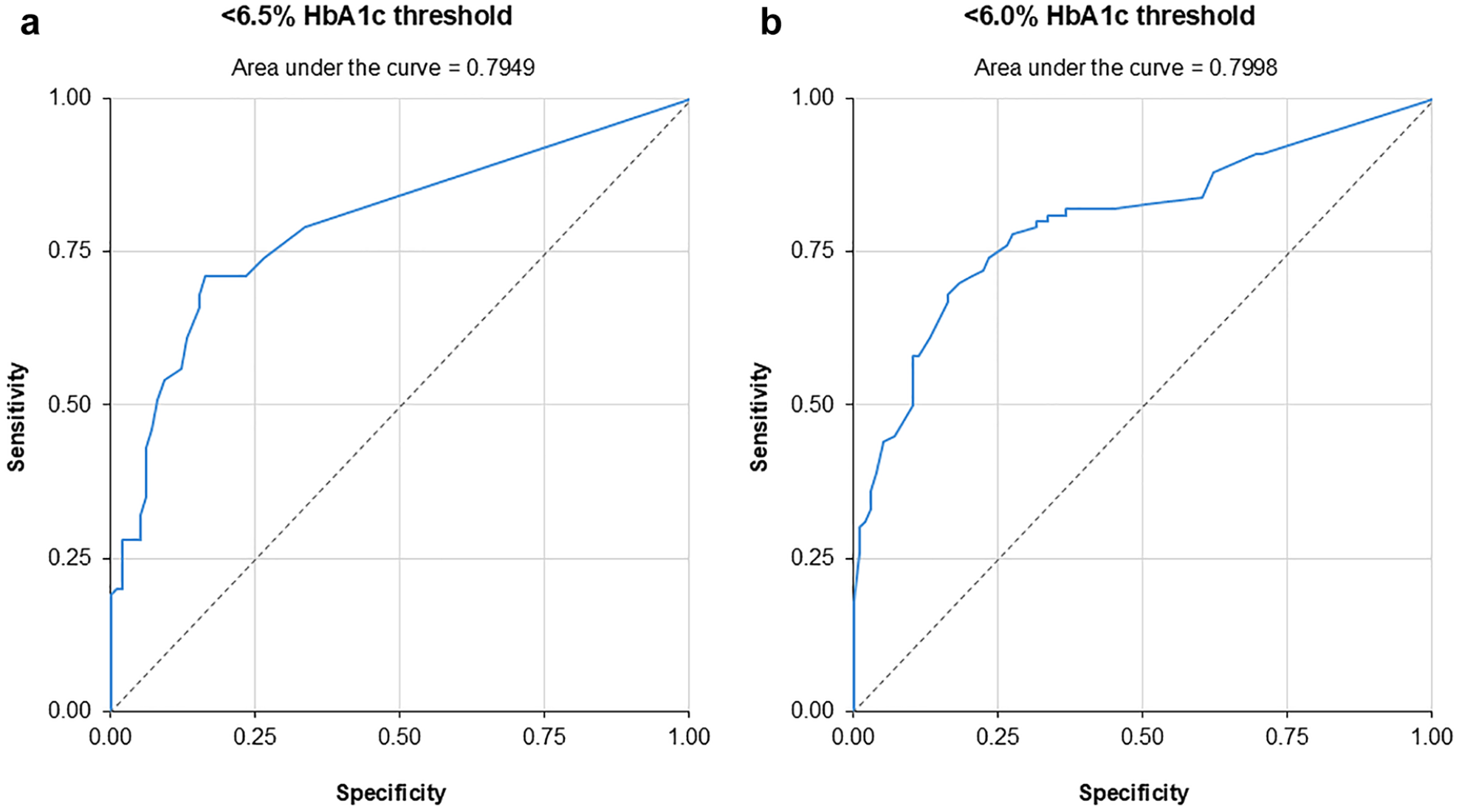
ROC analysis for (a) <6.5% and (b) <6.0% HbA1c cut-off values. (a) <6.5% HbA1c threshold. (b) <6.0% HbA1c threshold.
Pooled Specificity, Sensitivity, and Other Model Outputs.

Participants included individuals without and with known T2DM status.
Abbreviations: CI, confidence interval; DM, diabetes mellitus; HbA1c, glycated hemoglobin; NPV, positive predictive value; PPV, positive predictive value.
The chosen model, as expected, had reasonable to good sensitivity in correctly identifying those known to have T2DM (HbA1c cut-off 6.5%: 34.2% [95% CI = 23.4-45.1]; HbA1c cut-off 6.0%: 61.1% [95% CI = 51.2-70.9]). There was also good concordance between sensitivity and specificity (71.5% each for both HbA1c cut-offs).
The model showed consistent and reproducible chemometric predictions with no relevant impact of tested anthropometric variables, race, ethnicity, concomitant conditions or medications, smoking, and number of spectral assessments per nail or dominance of the assessed hand. In addition, it was demonstrated to be robust against variables affecting the quality of glycation in the nail, including the presence of microcirculatory or rheumatoid conditions, aged keratin in those ≥65 years, and hormonal fluctuations in post-menopausal women.
Safety
Overall, in both studies, there were no adverse NIR device–related adverse safety reports or incidents or suspected de novo diabetes cases for over 12 000 spectral measurements (6000 in each study).
Discussion
This exploratory clinical investigation confirmed that our chemometric prediction model, chosen based on preclinical validation, was able to assess spectral data accurately to correlate between spectral patterns and dichotomized diabetes risk status along the glycemic spectrum, using HbA1c cut-offs of clinical importance (6.5% and 6.0%). The upper cut-off reflects the HbA1c threshold at which subsequent clinical diagnostics are indicated, while the lower cut-off represents the threshold for prediabetes or no immediate risk of T2DM (normal) in accordance with current international diabetes associations’ definitions.11,27 Importantly, the system was able to detect when T2DM was not present with very high specificity and good probability.
The preliminary results from our pooled analysis indicate high specificity and safe clinical implementation of non-invasive NIR technology with the potential for simplifying large-scale screening for T2DM and allowing early detection and management of T2DM risk in diverse populations. Individuals with T2DM were included in these studies to train the algorithm to understand the dimensions of the glycemic spectrum. The IQR of baseline HbA1c values in those with and without T2DM was narrow, with some individuals presenting with levels close to or below the diagnostic threshold of 6.5% as outlined in treatment guidelines.11,27 These results suggest that this model is accurate at different glycemic levels and can differentiate between values close to this important threshold.
In this pooled analysis, the best-performing prediction algorithm was trained in a known population along a wide glycemic spectrum in individuals with or without diabetes. However, as the intended-use population for this mass-screening system will be individuals with unknown diabetes status, the best prediction algorithm was based on the highest-possible specificity and NPV. Therefore, as sensitivity and specificity are inversely related, values for sensitivity were relatively low in this pooled analysis of individuals with known glycemia. Real-world sensitivity and the true rate of detection of previously undiagnosed cases (those not testing negative) among the intended-use population will only be defined outside controlled clinical investigations.
Detection of glycated keratin was not impacted by DM-related complications, concomitant conditions/medications, or other variables including smoking and therefore assumed contamination of fingernails with tar. Of note, differences in ethnicity did not impact detection of glycated keratin on fingernails, a stable non-pigmented tissue, 28 which is important given that most optical devices fail in accuracy due to skin color or ethnicity impacting the methodology used, including blood-based HbA1c. The implementation of quality control models to ensure high-quality spectra has been successfully demonstrated in nails. 29 No adverse safety reports or incidents, or suspected de novo diabetes cases, were reported.
These initial findings support further development of NIR technology and the Glyconics-DS system, as a reusable, portable, non-invasive device with potential for use in primary care and community settings, to reduce barriers to diabetes screening within and outside of traditional health care settings and improve access to timely interventions. The potential utility for opportunistic screening within hard-to-access health care settings should be highlighted, particularly compared with conventional blood testing. It is known that HbA1c has lower clinical sensitivity at the diagnostic threshold and that age, race, ethnicity, and clinical conditions that alter erythrocytes or hemoglobin levels can affect HbA1c independent of glycemic level. Also, HbA1c testing is inaccessible, resource-intense, and expensive in most regions. 12 Furthermore, as the suggested NIR system with rapid cloud-based analytics does not generate hazardous waste or rather, necessitate the use of any disposables (ie, plastics, tubes, or cartridges for analytics or calibration), it is likely to offer a sustainable and accessible, low-cost alternative for mass screening in any environment.
In ANODE01 and ANODE02, the clinical feasibility and usability of the non-invasive NIR methodology were confirmed for the first time in clinical investigations, independent of the health care delivery setting or cohorts assessed for DM risk. The present pooled analysis was used to validate the chosen algorithm for which the detailed conceptual, preclinical chemometric model performance, and technical results have been published elsewhere. 25 The key results from this analysis contributed to the pivotal performance evaluation study (ANODE03: NCT06261944) investigating the final system using the model from our pooled analysis for the diagnostic cut-off, which has proven insensitive to clinical variables. In addition, the preferred hand (and fingernail) will be used based on observed no difference between hands, or hand dominance, in the current study. Three measurements will be employed, shown to be sufficient per person, initiated by an app with the operator only initiating serial screening tests. Additional algorithmic data will be generated in the “DIRTY” substudy to identify and mitigate the risk of spectral contamination, to ensure exclusion of suboptimal data, and to establish the foundation for predictive modeling in spectroscopy-based screening. Furthermore, a change to the internal standard methodology (blood sampling for HbA1c testing) will enable correction for anemia and hemoglobinopathies, and a modified cradle will be validated with user requirements. Furthermore, test sets from an independent cohort, essential for validating model performance, will be included in subsequent studies to verify the selected prediction algorithm in the intended-use population.
Study strengths include the robust pre-planned design of two identical studies for use in a pooled analysis, diverse participant cohort, and wide age range (and varying nail plate thickness which typically increases with age), with known/unknown T2DM status to optimize identification of a robust prediction algorithm. The accuracy and high level of specificity in detecting when T2DM was not present was also key, in contrast to conventional HbA1c measurement which can vary in certain conditions, especially in diverse populations. Also, from a sustainability perspective, no consumables are required even for calibration, with rapid, affordable, portable, and accurate testing, comparable to any invasive methodology. Limitations include that the presence of nail abnormalities, discoloration, or other visible changes currently renders the use of this technology largely impractical, underscoring the need for clear nail health for accurate and timely early screening using this method. However, use of any hand/fingernail removes the risk of inaccuracy provided there is at least one healthy nail for assessment. Analysis of pooled data from both pilot studies highlighted the known compromise between sensitivity and specificity across the models because optimization of specificity and high NPV were among pre-specified selection criteria for the most optimal analytical prediction algorithm. We also acknowledge that people with long-term T2DM and sustained hyperglycemia, who used to display the higher end of glycemic spectrum, present with a different type of keratin glycation pattern compared with pre- or early stages. The ANODE03 study will assess the genuine accuracy of the system in the intended-use population among those with unknown DM risk. Only real-world studies, known to enroll more health-conscious individuals, will be able to demonstrate the true sensitivity in detecting undiagnosed cases of diabetes outside systematic clinical investigations in apparently healthy people.
Conclusions
The pooled results of our two independent studies indicate the clinical feasibility and high specificity of a novel NIR spectroscopy-based assessment of T2DM risk, with potential for easy and effective mass screening, allowing early detection of increased T2DM risk across any health care delivery setting. As an affordable, portable, reusable, non-invasive device, the Glyconics-DS system has the potential to reduce health inequalities and improve access to timely interventions to reliably determine when DM is not present or requires early intervention, preserving limited health care resources for those in need of further assessment.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251331069 – Supplemental material for Use of a Miniaturized Near-Infrared Spectroscopy Device for Type 2 Diabetes Mellitus Screening: Pooled Analysis of the Pilot ANODE01 and ANODE02 Studies
Supplemental material, sj-docx-1-dst-10.1177_19322968251331069 for Use of a Miniaturized Near-Infrared Spectroscopy Device for Type 2 Diabetes Mellitus Screening: Pooled Analysis of the Pilot ANODE01 and ANODE02 Studies by Pratik Choudhary, Päivi M. Paldánius, John E. Salter, Daniela Lazaro-Pacheco and Francesc Xavier Cos Claramunt in Journal of Diabetes Science and Technology
Supplemental Material
sj-png-2-dst-10.1177_19322968251331069 – Supplemental material for Use of a Miniaturized Near-Infrared Spectroscopy Device for Type 2 Diabetes Mellitus Screening: Pooled Analysis of the Pilot ANODE01 and ANODE02 Studies
Supplemental material, sj-png-2-dst-10.1177_19322968251331069 for Use of a Miniaturized Near-Infrared Spectroscopy Device for Type 2 Diabetes Mellitus Screening: Pooled Analysis of the Pilot ANODE01 and ANODE02 Studies by Pratik Choudhary, Päivi M. Paldánius, John E. Salter, Daniela Lazaro-Pacheco and Francesc Xavier Cos Claramunt in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
For ANODE01, the authors thank all personnel and participants who were involved in the study, including study coordinators Mikila A Walters and Harvinder Sian at Leicester Diabetes Centre and Vivienne Abbas for study management at Glyconics Ltd. For ANODE02, this research was supported by Center for Biomedical Research on Diabetes and Associated Metabolic Diseases (CIBERDEM; group CB15/00071) at the Instituto de Salud Carlos III, Barcelona, Spain. The authors thank all personnel and participants, including Francisco Cegri, Jordi Ingla, Xavier Peligros, and Bogdan Vlacho from CIBERDEM and Vivienne Abbas for study management at Glyconics Ltd. Medical writing support was provided by Suzanne Axford-Bryars BSc (Hons), of BMP Services (UK) Ltd, a contract writer working on behalf of Glyconics Ltd, in accordance with Good Publication Practice 2022 guidelines under the direction of the authors (![]() ).
).
Abbreviations
ADE, adverse device effect; AE, adverse event; AUC, area under the curve; CI, confidence interval; DM, diabetes mellitus; HbA1c, glycated hemoglobin; IQR, interquartile range; NIR, near-infrared; NPV, negative predictive value; PPV, positive predictive value; SADE, serious adverse device effect; SAE, serious adverse event; T2DM, type 2 diabetes mellitus.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PC: Medtronic, Novo Nordisk, Lilly, Sanofi, Abbot, Dexcom, Vertex, Roche, and Insulet; research support: Medtronic, Abbott, Dexcom, and Novo Nordisk. PMP: Consultant; Glyconics Ltd, Altimmune, Speaker’s Bureau; Novartis. JES: Consultant; Glyconics Ltd. DL-P: Employee; Glyconics Ltd. FXCC: AstraZeneca, Abbott, Boehringer Ingelheim, Lilly, Novartis, Novo Nordisk, and Menarini.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The ANODE01 and ANODE02 studies were sponsored by Glyconics Ltd and registered with ClinicalTrials.gov (ANODE01: NCT05198895; ANODE02: NCT05476016).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
