Abstract

Introduction
On August 15, 2025, we will celebrate the 75th anniversary of the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) at the National Institutes of Health (NIH). This symposium is a collection of six manuscripts1-6 highlighting the development and clinical translation of the artificial pancreas (AP)—a landmark achievement in the treatment of diabetes, to a significant extent due to the critical support of NIDDK and the Institute’s Special Diabetes Program—a statutory funding program that began in 1998, focusing on basic, translational, and clinical research for the prevention and cure of type 1 diabetes (T1D). Over two decades, the AP grew from a disputed idea in 2005, to the standard of care for T1D today, improving the lives of those who struggle daily with this condition. The symposium is also part of a series of publications on a variety of relevant topics highlighting NIDDK-supported research contributions as the Institute celebrates its 75th anniversary. All manuscripts are written by those who had prominent contributions to the science, engineering, and clinical testing of the AP technology. Here is their story:
On a cold and cloudy December day in 2005, physicians, scientists, and engineers working on diabetes technologies gathered in the Lister Hill Auditorium at the National Institutes of Health in Bethesda, Maryland. This was the first workshop, “Obstacles and Opportunities on the Road to Artificial Pancreas: Closing the Loop,” organized by NIDDK and the Juvenile Diabetes Research Foundation (JDRF, now Breakthrough T1D) in collaboration with the Food and Drug Administration (FDA). In the previous 30 years, the treatment of T1D had been a technology-rich clinical endeavor with increasingly significant engineering components. In the 1970s, the first trials of continuous subcutaneous insulin delivery (CSII) using insulin pumps were reported,7,8 and by 2005, CSII has established its presence in the treatment of T1D. Research was also underway in the 1990s on designing automation algorithms for insulin delivery, anticipating the development of suitably accurate sensors and pumps, laying the groundwork for what was to become the core algorithm in modern commercial devices—model predictive control (MPC). 9 The progress of an AP suitable for outpatient was enabled by the introduction of continuous glucose monitoring (CGM).10,11 In 2004-2005, first attempts to automate glucose control using CGM and CSII were made.12,13 Thus, the focus of this first NIH/JDRF workshop was on the viability of an AP system using subcutaneous glucose sensing via CGM and CSII. The room was divided on this subject, with opinions ranging from “impossible” to “not too difficult”—and, as expected, the truth lied between these two extremes. Nevertheless, the workshop achieved its objective: a movement was initiated toward solving the challenges of automated insulin delivery (AID), and in 2006, a Request for Applications was issued initiating the JDRF Artificial Pancreas Project. The second FDA-NIH-JDRF workshop, “Towards an Artificial Pancreas,” was held in July 2008, followed by solicitations issued by the NIDDK to support research developing new AP technologies.
This collection of papers1-6 commemorates the academic and industrial research and development supported by NIDDK, Breakthrough T1D, and the Helmsley Charitable Trust, which drove the creation and validation for clinical use of several viable AP systems. A Supplement on Development & Future of AID Systems, published recently in Diabetes Technology and Therapeutics, detailed the private sector developments during this same period of time, highlighting the pioneering contributions of a number of companies to the AP field. 14
The Following 10 Years
A number of studies documented the benefits of CGM15-17 and outlined its future as a component of an AP system.18-21 The long way toward the contemporary interoperable user-friendly closed-loop AP began in 2007 with the introduction of a universal research platform—the APS—developed at the University of California, Santa Barbara (UCSB). 22 The APS enabled automated communication between a variety of CGM devices, insulin pumps, and control algorithms, and powered the first inpatient clinical trials.23,24 In April 2007, a major milestone was achieved with the APS enabling the first closed-loop human subject trial in the United States at UCSB. 25 In 2008, a computer simulator of the human metabolic system in T1D developed at the universities in Padova, Italy and Virginia, USA, and was accepted by the FDA as a substitute to animal trials in the testing of AID algorithms—this unprecedented event accelerated the AP development by years. 26 Outpatient studies took an AP system to a camp setting, where children with T1D were observed overnight, comparing AP to sensor-augmented pump therapy. 27 This same system was then taken to patients’ homes, achieving similar results. 28 However, these first inpatient and outpatient studies used a system that was based on laptop computers, still cumbersome and unsuitable for routine outpatient use.
In 2009 JAMA wrote “Artificial pancreas may soon be a reality,” 29 and by 2010, the Artificial Pancreas became a global research topic. In May 2012, a Diabetes Outlook published in Nature 30 highlighted the AP, and then Science featured the same topic. 31 These Nature 30 and Science 31 reviews featured a photo of a smart phone presenting a dual traffic-light display—the face of the first portable AP platform—the Diabetes Assistant developed at the University of Virginia in 2011. Diabetes Assistant was built using an Android smart phone as a computational hub, and included graphical user interface designed for the patient 32 and Web-based remote monitoring. 33 Diabetes Assistant was used in a number of clinical trials at ten centers in the United States and Europe.34-42
In 2014, NIDDK issued a program announcement titled “Advanced Clinical Trials to test Artificial Pancreas Device Systems in Type 1 Diabetes (UC4).” Increasing research activity was directed toward designing innovative control algorithms that could integrate CGMs and pumps, drawing on insights and expertise from the process dynamics and control field. 43 Studies supported by this initiative resulted in significant clinical translation—the roots of several contemporary AP systems can be traced to this NIDDK support. In July 2016, Diabetes Care published a symposium 44 dedicated to outpatient AP studies, 45 including trials at patient’s homes lasting a month or more37,38,45 and studies in young children. 39 The AP systems were pilot-tested, and were found effective, in pregnant women with T1D,46,47 and in people with type 2 diabetes on insulin therapy. 48 In 2016, a pivotal trial of the first commercial hybrid AID system was completed—the Medtronic 670G—which automatically modulates basal rates but not insulin boluses 49 —and the system was cleared by the FDA for clinical use. Between 2016 and 2018, the National Library of Medicine included more than 150 publications per year related to the engineering development and clinical testing of various closed-loop systems—a trend that confirmed the growing prominence of AP. Systematic reviews and meta-analyses were published by various journals, confirming the efficacy of closed-loop control for the insulin treatment of diabetes.50-53 The AP was coming of age.
State of the Art Today
A dream in 2005, closed-loop control of diabetes, also known as AID, or the AP, is now the gold-standard treatment for T1D, and is expanding to people with type 2 diabetes on insulin treatment. Several commercial systems are now available in the United States, including: Medtronic’s MiniMed 670G, 770G, and 780G, the latter adjusting automatically basal rate and administering correction boluses; t:slim X2 and Mobi with Control-IQ Technology by Tandem Diabetes Care, which automates both insulin basal rate and correction boluses; OmniPod 5 hybrid closed-loop system, Insulet Corporation; the iLet Bionic Pancreas system by Beta Bionics, CamAPS FX (CamDiab Ltd., Cambridge, UK)—a mobile app that is the first to be approved for use by pregnant women with T1D, and the twiist Automated Insulin Delivery (AID) system by Sequel. Other AP systems are available overseas, but are not cleared by the FDA in the United States, for example, Diabeloop DBLG1 (Diabeloop France) and Inreda AP—a bi-hormonal fully automated closed-loop system (Inreda Diabetic B.V., the Netherlands). The development and testing of most of these systems, for example, MiniMed 780G, Control-IQ, iLet, Cam APS FX, was supported by the NIDDK UC4 grants noted above, while NIDDK R01, R21, and DP3 grants supported developments at UCSB that led to the predictive algorithm powering the OmniPod 5. The major difference between AP configurations is the control algorithm, which directs insulin delivery based on CGM readings and other factors, such as insulin on board. For example, the MiniMed systems use proportional integral derivative algorithms, while most others use controllers based on the MPC algorithm, originally developed for the chemical process industry in the 1960s. 43 MiniMed 780G added a previously introduced fuzzy logic algorithm 27 responsible for the administration of correction boluses. Except for Inreda AP, which injects insulin and glucagon, all closed-loop systems inject insulin only, with the iLet potentially capable of injecting glucagon as well.
Multiple well respected peer reviewed medical journals embraced the AP topic. In the span of four years the New England Journal of Medicine, published seven clinical trials,54-60 indicating that AP technologies have firmly transitioned to the clinical practice of T1D. These papers featured several AP systems noted above: Control-IQ,54,55,57 the iLet, 58 Cam APX,59,60 and an open-source control algorithm. 56 The AP technologies made successful strides into insulin-using type 2 diabetes as well.61-63 Large volumes of papers reported outcomes of real-world AP use by thousands of people for extended periods of time,64-73 for example, 780G,64-67 Control-IQ,68-71 and OmniPod 5.72,73 Figure 1 presents the number of publications in PubMed per year resulting from a search on “artificial pancreas” or “closed loop control of diabetes.”
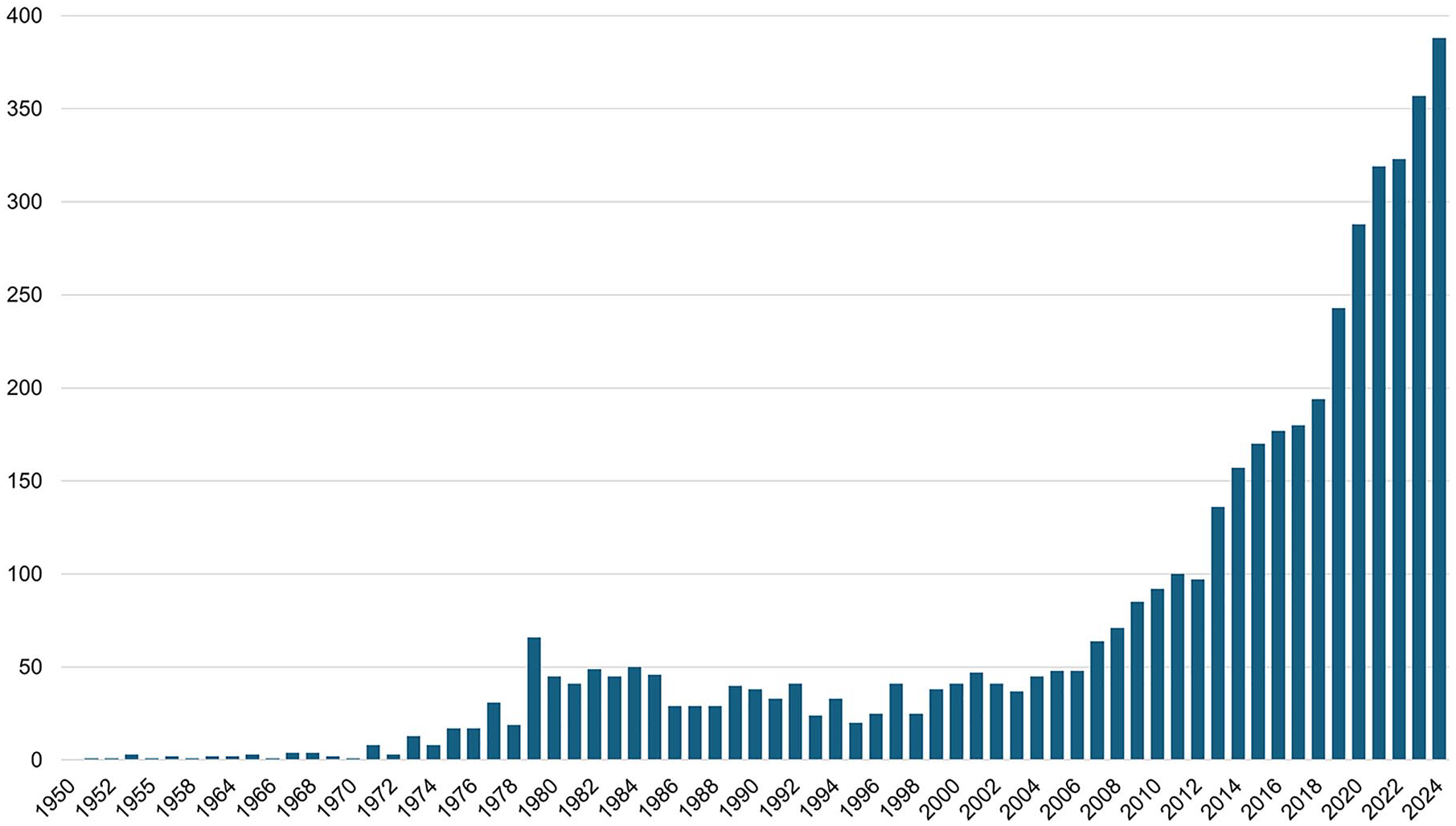
Number of publications in PubMed per year resulting from a search on “artificial pancreas” or “closed loop control of diabetes.”
Clusters of publications addressed important subtopics of the general AP theme, such as use of AID in pregnancy complicated by T1D74-78; ultra-fast insulin analogues attempting to minimize postprandial glucose excursions with hybrid AP systems, which resulted in mixed success79-82; use of AID in various type of exercise83-86; and the efficacy of AP in people with history of severe hypoglycemia and hypoglycemia unawareness.87-89 International forums crafted consensus statements on the clinical use of closed-loop control.90-92
Overall, while detailed comparisons of results across studies are not statistically plausible due to differences in sensors used, experimental setup, and lack of randomization, it became evident that (1) all contemporary AP systems perform significantly better than both multiple daily injections or sensor-augmented insulin pump therapy, and (2) the first-generation AP systems achieve, on average, strikingly similar results, for example, time in the target range of 70 to 180 mg/dL (TIR) of ~70-75 percent and rate of hypoglycemia (below 70 mg/dL) in the order of 2% to 3%. The “ceiling effect” of TIR is an important observation pointing at the limitations of the current hybrid closed-loop devices: both the physiology of the subcutaneous route of insulin delivery and factors stemming from the behavior of AP users present challenges that need to (and will be) addressed in the near future.
The Next Frontier
While predictions are often inaccurate, in this section, we will attempt to forecast what is coming next to the AP field, based on current major research trends. In 2023, the Fifth Artificial Pancreas Workshop: Enabling Full Automation, Access, and Adoption was held at the NIH Campus in Bethesda, Maryland. 93 The Workshop outlined several major trends in the development of AP systems: (1) data management, integration, and cybersecurity; (2) adaptation of AID through the lifespan; (3) approaches to fully automated closed-loop control, and (4) novel artificial intelligence (AI) strategies to develop smarter, personalized, diabetes management systems. 93 Overall, the next frontier of AP development is abundantly clear: the quest is for a fully automated system that would not require input from its user to maintain optimal glucose control. This is in contrast to virtually all contemporary AID systems, which are “hybrid,” in that the user is expected to announce meals and initiate meal boluses or anticipate physical activity and attenuate insulin delivery in advance. While the most advanced hybrid AID systems have consistently shown improvements in overnight control, all studies to date point out that achieving optimal control during the day is still a challenge. This is a direct result of behavioral disturbances typically occurring during the day, for example, meals or exercise that contribute to unexpected glycemic fluctuations. Moreover, the current speed of insulin action, even with modern rapid-acting analogues, is still too slow to enable adequate AID response—it is simply too late to automatically inject insulin when a CGM indicates a postprandial glucose excursion or to discontinue insulin delivery when exercise is detected. This motivates the development of a fully automated closed-loop (FCL) system that does not require meal or exercise announcements. Interestingly, one of the first published closed-loop trials in the United States employed an FCL system, which showed promise, albeit using small (30 g carbohydrate) unannounced meals. 24 While the task is clear, the routes to achieving FCL are different and can be classified as follows: (1) ultra-fast insulin attempting to compensate for both the time difference between glucose absorption during meals and the onset of insulin action, and the attenuation of insulin action during exercise; (2) dual insulin plus glucagon control typically increasing insulin doses around meals and counteracting potential insulin-induced hypoglycemia with automated glucagon injection; (3) use of adjuvant hormones, for example, amylin, GLP-1 receptor agonists, SGLT-2 inhibitors, which are known to mitigate postprandial meal excursions, or additional signals, such as motion sensing or heart rate, reacting fast to physical activity; and (4) new control algorithms trying to anticipate the timing of the next meal or exercise based on historical data for each user. The latter is a route to adaptive personalized AP systems, which are certainly on the list of upcoming developments. Currently, some algorithms are “trainable” adapting control parameters based on the experience of their users.94,95 In one study using adaptation of the algorithm’s “aggressiveness” during periods of uncertainty, a remarkable 88% TIR was achieved, which remains one of the highest recorded in a closed-loop study. 95
We can, however, anticipate a brighter future for truly personal FCL using the vast amounts of data available now. Looking back at the beginning of the AP project 20 years ago, data were sparse. The CGM was in infancy and data sets including both glucose readings and insulin delivery were generally unavailable, with one notable exception: elaborate studies of the human metabolic system, typically involving tracers containing labeled glucose, have been done, allowing the creation of comprehensive mathematical models of insulin-glucose dynamics.96-98 Consequently, to date, most AP control algorithms are model-based using models of the metabolic system, simplified enough to be tractable in real time, to calculate control actions. 43 With the accumulation of large data sets and the increasing ability of contemporary CGM/CSII systems to track an individual across long periods of time, “model-free” data-driven controllers are emerging based on techniques that fall in the domain of data science, such as machine learning (ML) and AI. Both ML and AI can remedy certain shortcomings of the current AID systems, as elaborated by a recent review of data science methods available to diabetes research and clinical practice. 99 These methods range from detection of glycemic patterns and prediction of high-risk events, to the first AID system based entirely on ML and a deep neural network, which opened the field for a new class of purely data-driven control algorithms.100,101 By design, ML and AI are capable of real-time learning, adaptation, and problem-solving. While CGM data are still relatively sparse (one year of five-minute sampled CGM = approximately 100 000 points) compared with the volume of data required to train typical Large Language Models (hundreds of GBs to TBs), as more data are accumulated across AP users and time, we can expect that future AID developments will gradually transition from traditional equation (model)-driven control algorithms to smarter, personalized, trainable, fully automated ML, and generative AI implementations. This transition has already begun 101 and the movement toward the AI AP of the near future is likely inevitable.
About This Symposium
This symposium is a collection of six manuscripts highlighting the development and clinical translation of the AP idea,1-6 assembled in celebration of two decades of NIDDK and the Special Diabetes Program support to this technology. All manuscripts are written by scholars, engineers, and physicians, who had prominent contributions to the development and clinical translation of the AP. The collection includes a paper on the hardware components of the AP—CGM and insulin pumps, 1 as well as a paper on the metabolic models, in silico pre-clinical trials, and algorithms that integrated the components in what is now known as AP system. 2 The clinical testing and translation of the AP is reflected by two papers—one on early studies 3 and another on pivotal registration trials, which earned FDA clearance to the systems currently in use. 4 Finally, two papers discuss the challenges and opportunities to AP research 5 and the practical considerations for widespread AP use in the treatment of diabetes. 6 As pointed out in the beginning, the focus of this collection is more on academic than industry developments, and in particular highlights the contributions of NIDDK and organizations such as Breakthrough T1D to the creation of the AP.
Footnotes
Acknowledgements
As stated in the manuscript, the purpose of this symposium is to celebrate NIDDK’s 75th anniversary, to recognize the scientific input from NIDDK and its partners, and to acknowledge NIDDK, its partners and the Special Diabetes Program overseen by NIDDK, for their sustained long-term financial support toward the development and translational success of the AP project. The genesis for this symposium is credited to Drs. Guillermo Arreaza-Rubin of NIDDK. The authors thank Dr Thomas Eggerman and Neal Green (NIDDK) for the support in organizing this complex endeavor. This manuscript was not prepared under the auspices of the NIDDK and does not represent analyses or conclusions of the NIH or the NIDDK. FJD would like to acknowledge the stimulating and fruitful multi-decade collaborative endeavors with Eyal Dassau and Howard Zisser. BPK would like to acknowledge his long-standing collaborators Sue Brown and Marc Breton, as well as his outstanding colleagues at the UVA Center for Diabetes Technology.
Author Contributions
BPK proposed wrote the first draft of the manuscript. FJD revised the manuscript and added additional material.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FJD reports funding over the time course described in this editorial from the Showalter Foundation, Roche Diagnostics Corporation, the Juvenile Diabetes Research Foundation, the Helmsley Charitable Trust, and the National Institutes of Health (NIDDK). FJD also reports royalty payments from patents in a portfolio that have been licensed to Insulet, Roche, and Dexcom. BPK reports research grants handled by the University of Virginia from the National Institutes of Health, Novo Nordisk, Dexcom, and Tandem Diabetes Care. In addition, BPK has a number of patents with royalties paid to Dexcom and Novo Nordisk.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
