Abstract
The development of automated insulin delivery systems has seen tremendous improvements from individual components to interoperable system combinations of devices and new drugs besides insulin. The components have become progressively smaller, more accurate, and more user friendly. This article summarizes the history of the artificial pancreas from the earliest concepts to fully functional systems to research into further improvements in the future. The authors include many of the developers of this technology who received research support from the National Institute of Diabetes and Digestive and Kidney Diseases at various stages to develop these systems.
Introduction
This article is part of a collection of publications to commemorate the 75th Anniversary of the NIDDK.
Stages of automated insulin delivery (AID) span the 1960s to the present day. 1 In this article, we discuss eight important periods in the development of AID systems. These include (1) the feasibility of automating insulin delivery including the first advanced insulin infusion system using a control loop (ADICOL) studies in Cambridge and studies at Medtronic; (2) the first US closed-loop trial—the early days—from zero to one; (3) real-time CGM and the artificial pancreas system (APS): the Juvenile Diabetes Research Foundation (JDRF) CGM study and first inpatient trials with the APS continuous glucose monitoring study and first inpatient trials with the APS; (4) the artificial Pancreas in a diabetes summer camp; (5) wearable artificial pancreas: Diabetes Assistants (DiAs) used in a number of clinical trials at ten clinical centers in the United States and Europe; (6) the artificial pancreas in children; (7) the AP in type 2 diabetes; and (8) multi-hormone AP and adjuvants. These sections cover a variety of progressively complex and progressively effective technologies developed by pioneers in AID technology.
Feasibility of Automating Insulin Delivery: First Advanced Insulin Infusion Using a Control Loop (ADICOL) Studies in Cambridge and Studies at Medtronic
1University of Cambridge, Cambridge, UK
2Harvard Medical School, Harvard University, Boston, MA, USA
3Boston Children’s Hospital, Boston, MA, USA
The concept of closed-loop insulin delivery was proposed in the 1960s by Kadish, 2 followed a decade later by the work of Albisser, 3 and Pfeiffer. 4 This led to the development of the fridge-size Biostator device. 5 This early work envisioned intravenous glucose sensing and intravenous/intraperitoneal insulin delivery culminating in the development and clinical testing of a fully implantable system using a long-term implantable glucose sensor and an implantable insulin pump filled with U400 insulin (Medtronic MiniMed, CA, USA). 6
Smaller external insulin pumps became available in the 1980s, with rapid acting insulin becoming available in the 1990s. The first continuous glucose monitor system (CGMS, Medtronic MiniMed), was released in 1999. 7 Expectations for a fully automated closed loop system with subcutaneous glucose sensing and subcutaneous insulin delivery (SC-SC) were high as Medtronic-MiniMed researchers aimed to develop a system that would not require any input from the user while achieving glucose outcomes comparable to those of healthy individuals. 8 However, progress toward these goals proved to be slow due to inaccuracies in the commercial continuous glucose monitor (CGM) system, 9 and the goals were revised to focus on gradual, incremental, improvements in outcomes.
Using the incremental approach, the Advanced Insulin Infusion Using a Control Loop (ADICOL) project 10 shifted its focus to the SC-SC (subcutaneous glucose sensing and subcutaneous insulin) approach, aiming to address the high-cost, invasiveness, and high complication rates associated with implantable systems. Model predictive control (MPC) was used to link the CGM system to an insulin pump. The MPC algorithm, as introduced in the ADICOL project, used a metabolic model of the glucose regulatory system to predict future glucose levels given the known history of insulin delivery, carbohydrate consumption, and other factors such as exercise. From the predicted values, an optimal insulin delivery rate was calculated at each sample interval.
Likewise, Medtronic-Minimed (Medtronic, Northridge, California) shifted its focus from developing its fully implanted system, to the development of a SC-SC system. A 2006 feasibility study showed their CGMS (continuous glucose monitor system) and insulin pump could be linked to a proportional integral derivative (PID) algorithm 11 to create a fully automated system. 6 The fully automated system resulted in acceptable nighttime control, but peak post prandial glucose levels were higher than desired and nadir levels were lower than desired. This prompting the group to refocus their efforts to the incremental approach. This led to a subsequent study that showed the meal response could be improved by having the user administer a pre-meal insulin bolus. This approach is now referred to as hybrid closed loop. 12
First US Closed-Loop Trial—The Early Days—From Zero to One
1School of Engineering, Brown University, Providence, RI, USA
2Harvard John A. Paulson School of Engineering and Applied Sciences, Harvard University, Boston, MA, USA
The first US (United States) based Closed-Loop artificial pancreas came to fruition, thanks to the input of a few clinicians at Sansum Diabetes and a few engineers at University of California at Santa Barbara as well as the hard work of subjects, the generosity of grants from the NIDDK/NIH (National Institutes of Health) and JDRF (Juvenile Diabetes Research Foundation) and industry. Lois Jovanovic was a visionary clinician scientist, with worldwide impact, whose early work included understanding insulin requirements during pregnancy/labor/delivery in women with type 1 diabetes using the Biostator, a 60 kg device created in 1976 that utilized automated intravenous glucose measurements and insulin delivery.5,13 Francis J Doyle III, a professor of Chemical Engineering, had explored type 1 diabetes as a control problem to be solved.14,15 Eyal Dassau was a researcher (Chemical Engineer) with expertise in design/ system engineering and risk analysis. Howard Zisser, a clinical researcher, conducted many early diabetes device trials.16-18
Construction of the new APSs built on previous run-to-run control algorithm study, 16 in which they used daily prandial glucose data to optimize basal rates and insulin to carb ratios. The APS would narrow the control cycle from 24 hours to five minutes.
The priority of Drs. Zisser, Doyle, and Dassau was to build a modular safe system to use as a testbed for closed loop trails for our group as well as the JDRF APS consortium. The system needed to be a single input=glucose and single output=insulin delivery. The glucose data needed to be freed from the patient’s pocket. Dexcom (Dexcom, San Diego, California) agreed to allow us to import glucose data from their receiver every five minutes. Once they had that data on a laptop, the three developers realized that they could send the data with geolocation to the study team. This freedom of the glucose data has led users and caregivers to view their data in many places including their wrist. Insulet validated six essential pump commands the team identified. The only thing that was missing was the brains of the system, in this case a model predictive control algorithm.19-21
It was essential that the clinical team could see the data displayed in real time. On the study day, the first subject arrived fasting. They were connected to the APS, which assumed insulin pump control. Figure 1 presents the in silico dynamic simulator used for closed-loop control with an artificial beta cell with hardware-in-the-loop. Everyone in the room could see the control moves. After a 25 g carbohydrate (CHO) meal, the study was paused by Dr. Zisser, because it appeared the system was delivering an excess of insulin. The subject was monitored for several hours after the end of the study and went home safely. As in many experiments, not everything went as planned, but the results of this one trial allowed the team to optimize insulin-on-board feedback for future trials, to design inhaled and intraperitoneal AP systems22,23 and share this system to many clinical and industry partners around the world. Echoes of this system still ring in current commercial AP systems.
In silico dynamic simulator for closed-loop control of artificial beta cell with hardware-in-the-loop.
Real-Time CGM and the APS: The JDRF CGM Study and First Inpatient Trials With the APS
1Harvard John A. Paulson School of Engineering and Applied Sciences, Harvard University, Boston, MA, USA
2Breakthrough T1D, New York, NY, USA
3Division of Pediatric Endocrinology, School of Medicine, Stanford University, Stanford, CA, USA
4School of Engineering, Brown University, Providence, RI, USA
5School of Medicine, Yale University, New Haven, CT, USA
JDRF-CGM Trials
In 2006, the JDRF-CGM Study Group conducted two clinical trials on real-time CGM devices. The primary trial involved 322 participants aged eight and older with baseline Hemoglobin A1c (HbA1c) levels over 7%, comparing CGM with standard fingerstick monitoring. The second trial included 129 individuals already meeting HbA1c targets (<7%), focusing on reducing biochemical hypoglycemia.
The primary trial showed significant reduction in HbA1c levels in adults over 25 years old using CGM, but not in younger age groups. A post-hoc analysis revealed that the frequency of CGM use, rather than age, was crucial for HbA1c reduction. Participants using CGM at least six days per week experienced over 0.5% in HbA1c reduction. The low HbA1c study demonstrated a 40% reduction in biochemical hypoglycemia with CGM use, while maintaining target HbA1c levels.
These trials highlighted the effectiveness of CGM in managing type 1 diabetes and the importance of consistent device use.24-26
The Innovation Brick Road: Clinical Translation of Closed-Loop Control, the Birth of the APS
A critical challenge to clinical testing was moving beyond straightforward computer simulations to addressing the complexity and uncertainty of actual devices interacting with the human body. In order to move beyond human-in-the-loop management of glycemia, it was essential to connect devices and algorithms with humans, and this void was filled by the APS. 27 The APS modular platform, developed by Dassau and team, allowed flexibility and integration of various sensors, pumps, and control algorithms, emphasizing clinical translation. This flexibility was crucial for testing different configurations.
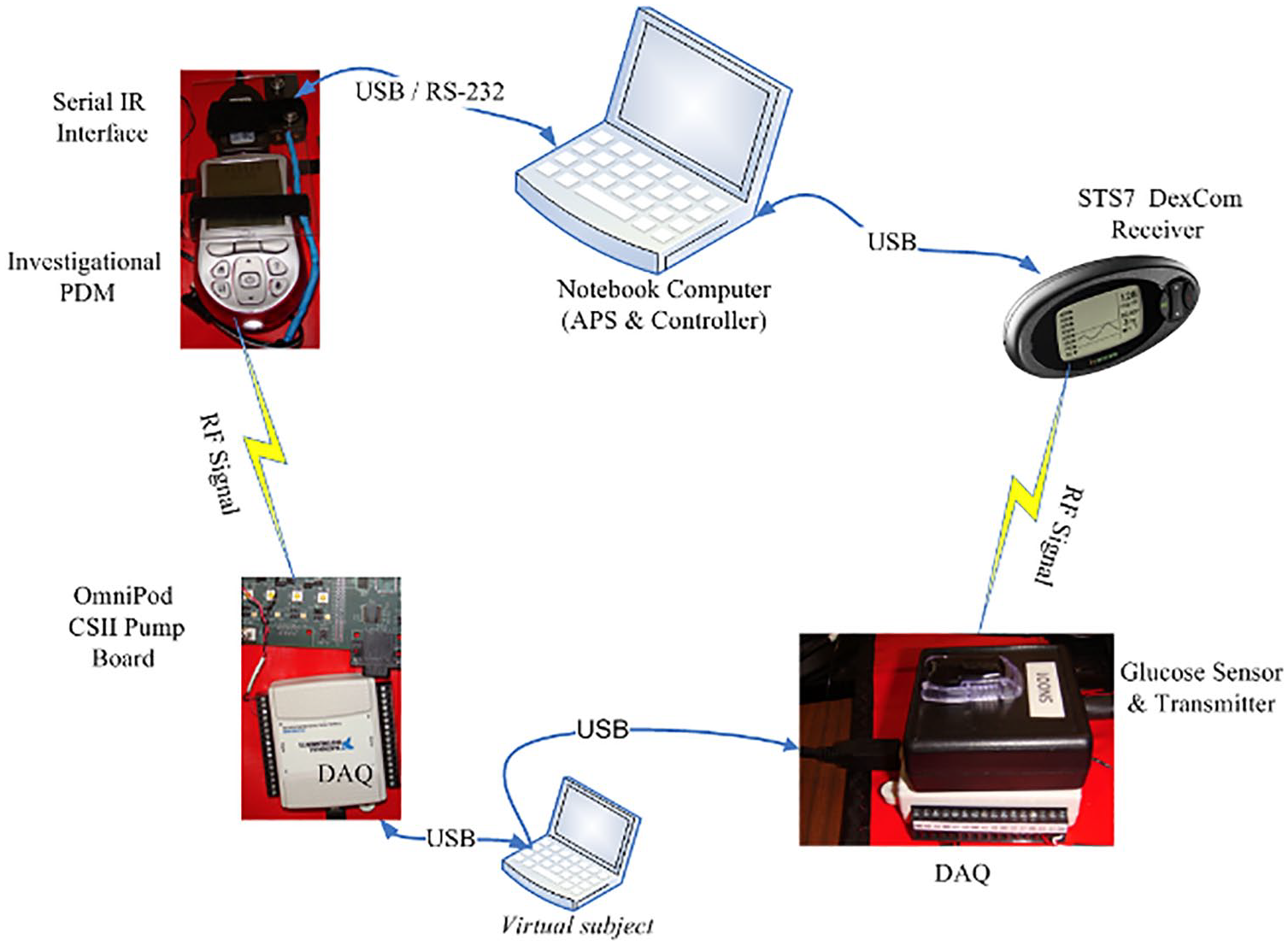
Omnipod communication was facilitated by limiting the number of commands, resulting in six core commands used in the APS and other systems to follow. Pump communication posed significant challenges, necessitating innovative solutions, such as using an activation personal data manager (PDM) to initiate the link with the POD and transfer control to the PDM connected to the APS.
The Dexcom receiver tool enabled direct data transfer, contributing to future communication protocols. This involved an engineering modification of the static transfer switch (STS) proprietary charging port, resulting from collaborative efforts. A similar strategy was used with the Abbott (Abbott Diabetes Care, Alameda, California) Navigator for glucose information transfer using an engineering cradle. Additional pumps, such as the Roche and Animas devices, were also integrated.
In April 2007, a significant milestone for AID was reached with the first MPC clinical study with the APS presented in Figure 2, leading to further studies, including one in Israel. 28 Collaborations with researchers resulted in the first JDRF international AID study 29 and novel designs involving inhaled 22 and intraperitoneal insulin. 23 The APS acted as an incubator for academic research and early industry-led studies, enabling the development of novel control algorithms for insulin management from Fuzzy Logic to PID and MPC.30-34
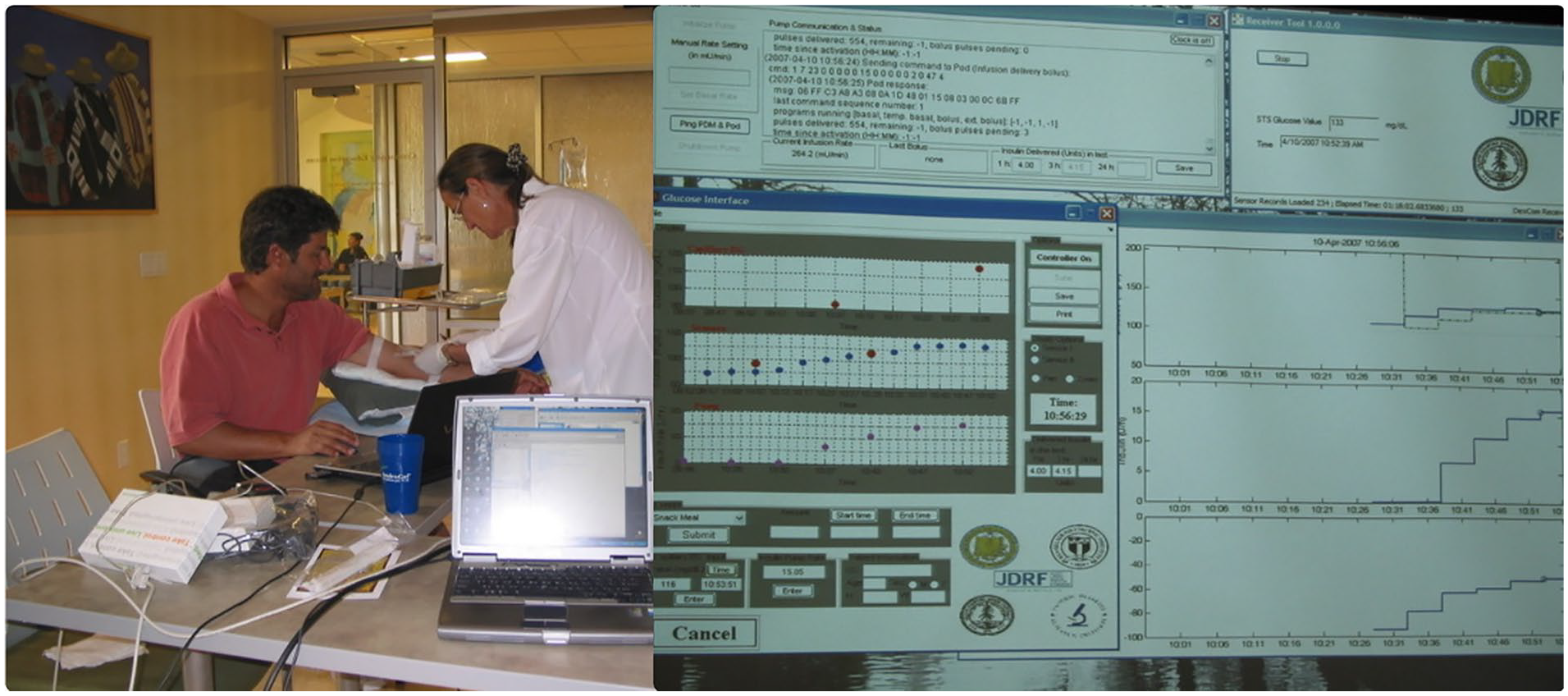
The first model predictive control (MPC) study using the Artificial Pancreas System (APS) on April 10, 2007, Santa Barbara, CA. On the top right, it displays APS screens, including communication linking the Omnipod, Dexcom STS-7, main APS user interface (UI), and the real-time control/prediction algorithm. On the left, APS is running on a laptop connected via cables to the personal data manager (PDM) and the Dexcom receiver, next to our first participant.
The Artificial Pancreas in a Diabetes Summer Camp
1Center for Diabetes Technology, The University of Virginia, Charlottesville, VA, USA
2Jesse Z and Sara Lea Shafer Institute for Endocrinology and Diabetes, National Center for Childhood Diabetes, Schneider Children’s Medical Center of Israel, Petah Tikva, Israel
3Faculty of Medicine, University Medical Center Ljubljana, University of Ljubljana, Ljubljana, Slovenia
4Harvard Medical School, Harvard University, Boston, MA, USA
5Beta Bionics, Inc, Irvine, CA, USA
Pediatric research on AID systems has been greatly assisted by testing in camp settings—which combines a means of assessing a large number of children and adolescents simultaneously and stress-testing systems in environments with intense physical activity and risk for hypoglycemia. Between vigorous childhood activity and the use of smart phones to run many prototype AID systems, it is not an exaggeration to say this approach to research has included broken phones and broken bones.
The use of camp settings initially progressed from simple remote-monitoring systems for overnight CGM surveillance and early recognition of hypoglycemia 35 to new AID systems used overnight to effectively prevent hypoglycemia.36,37 When AID systems that functioned around-the-clock were tested—both using both insulin alone and insulin plus glucagon—they performed exceptionally well in camp settings, with time in target range (TIR) increasing by 4% to 26% above sensor-augmented pump (SAP) times, while absolute time below glucose 70 mg/dL (TBR <70) was reduced up to 2%.37-40 These assessments were conducted across the pediatric age range, from children age five to eight years (Figure 3) through adolescents, as well as young camp counselors.

Pediatric AID studies at diabetes camps provide the opportunity to assess new technology approaches among groups of children—allowing acquisition of data collection in a more natural habitat.
While the majority of these studies occurred at diabetes summer camps (with study participants mixed in with other campers at meals and activities), other studies staged winter camps for trials that also assessed AID function in cold temperature and at high altitude for winter sports and downhill skiing. These studies also demonstrated superior glycemic management with AID compared to an SAP by 7% to 13% in absolute TIR.41-43
Finally, paralleling changes in the field overall, the first camp studies randomized campers to compare AID systems to SAP but eventually progressed to comparing AID systems with and without new features, including algorithms for initiation settings and adaptation.43-45 Overall, testing AID systems in pediatric camp settings has provided early, rapid assessment of new AID approaches—and has done so with an infinitely higher degree of fun than any other type of trial.
Wearable Artificial Pancreas: DiA Used in a Number of Clinical Trials at Ten Clinical Centers in the US and Europe
1Center for Diabetes Technology, The University of Virginia, Charlottesville, VA, USA
2Division of Pediatric Endocrinology, School of Medicine, Stanford University, Stanford, CA, USA
3Division of Endocrinology, The University of Virginia, Charlottesville, VA, USA
4Institute of Functional Genomics, Montpellier University Hospital, CNRS, INSERM, University of Montpellier, Montpellier, France
5Barbara Davis Center for Diabetes, Anschutz Medical Campus, University of Colorado, Aurora, CO, USA
6College of Medicine and Science, Mayo Clinic, Rochester, MN, USA
7Icahn School of Medicine at Mount Sinai, New York, NY, USA
Following its initial validation in hospital settings, the next logical step was to expose AID to increased variability in behaviors and metabolic fluctuations: clinical studies therefore needed to increase in length from days to weeks and be used in free-living conditions, forcing AID platforms to transition from computer-based prototypes to on-body systems. 46 The introduction of the first smartphones in 2007 (iOS) and 2008 (AndroidOS) offered a unique opportunity to leverage existing integrated connectivity to satellite devices (CGM and insulin pump) via standard protocols (eg, Bluetooth Low Energy [BLE]), computation power, battery life, and customizable user interfaces.
The first such system was proposed by researchers at the University of Virginia and used in multi-days supervised studies in 2011. 30 The Diabetes Assistant (DiAs) is a smartphone-based, modular, portable device that operates on a commercial phone, with wireless communication with satellite devices is presented in Figure 4. These devices include insulin pumps, CGMs, activity trackers, and any medical device using a standard wireless protocol like Bluetooth or BLE. 47 In addition, DiAs uses the smartphone’s Wi-Fi or cell data connection to stream the device data to a dedicated remote monitoring server, allowing real-time device specific monitoring by, and automated alerts to, the study team. 48 Finally, its modular architecture allows for different control modules and user interfaces to be easily swapped, enabling different AID or expert-decision support algorithms to be tested in human clinical trials. 49 Originally using a specifically modified Android operating system to protect operations, DiAs was filed with the United States Food and Drug Administration (FDA) in 2011 and received its first investigational device exemption in 2012.

The Diabetes Assistant (DiAs), the first mobile glucose control platform.
DiAs has since been used in over 20 clinical trials, on four continents,50,51 from supervised hotel studies to the ski slopes,41,52 for up to six months per user. 53 It has implemented six different families of AID algorithms50,53-57 and was integral in the eventual adoption of at least two commercial AID systems.58,59 Over the years, other smartphone-based systems have also supported AID research, such as the Oregon Health & Science University (OHSU) platform, 60 the interoperable artificial pancreas system (iAPS), 61 or do-it-yourself (DIY) community developments. 62
Since 2012, DiAs has continued to evolve, keeping pace with AndroidOS (using an unmodified commercial version since 2024), adding ancillary satellite devices (eg, pedometer and heart rate sensors), and a cloud-based computation and monitoring architecture, enabling secured bidirectional communications between the clinical team and the user, as well as the cloud computation capacity and the on-body algorithm. DiAs are used in feasibility studies to this day.
The Artificial Pancreas in Children
1Division of Pediatric Endocrinology, School of Medicine, Stanford University, Stanford, CA, USA
2Division of Endocrinology, Department of Pediatrics, University of California, San Francisco, San Francisco, CA, USA
3School of Medicine, Yale University, New Haven, CT, USA
4Barbara Davis Center for Diabetes, Anschutz Medical Campus, University of Colorado, Aurora, CO, USA
5Anschutz Medical Campus, University of Colorado, Aurora, CO, USA
6Center for Diabetes Technology, The University of Virginia, Charlottesville, VA, USA
AID is possible because of two main components: (1) CGM and (2) algorithms that constantly modulate insulin pump delivery. Early CGM development that led to Abbott Diabetes Care, 63 Medtronic, 64 and Dexcom 65 CGMS, and algorithm development at academic institutions (see other chapters) all began with NIDDK’s support. NIDDK/NIH provided support for translational research units conducting inpatient studies, early stage and established investigators, collaborative groups, and coordinating centers that allowed feasibility/pilot studies, and pivotal trials for AID in children. We focus on these studies.
Early inpatient studies: Initial studies requiring frequent venous blood glucose values were conducted in research units with algorithms on bedside computers (Figure 5a). An early study in 2008 determined that glycemic outcomes improved more with hybrid AID than with full AID. 12 Studies in 66 2014 and 2016 67 assessed the benefit of AID for missed meal boluses in adolescents. Algorithm development in 2009-2013 focused on predictive pump suspension to minimize nocturnal hypoglycemia,68,69 improve overall nighttime glycemia,70-72 and day and night hybrid AID with meals and exercise for up to two days.73,74 In 2013, an inpatient multicenter randomized clinical trial (RCT) showed that three days of hybrid AID within a week of type 1 diagnosis rapidly improved glycemic control, 75 but did not improve c-peptide levels. 76 Figures 5 and 6 present early AID systems.
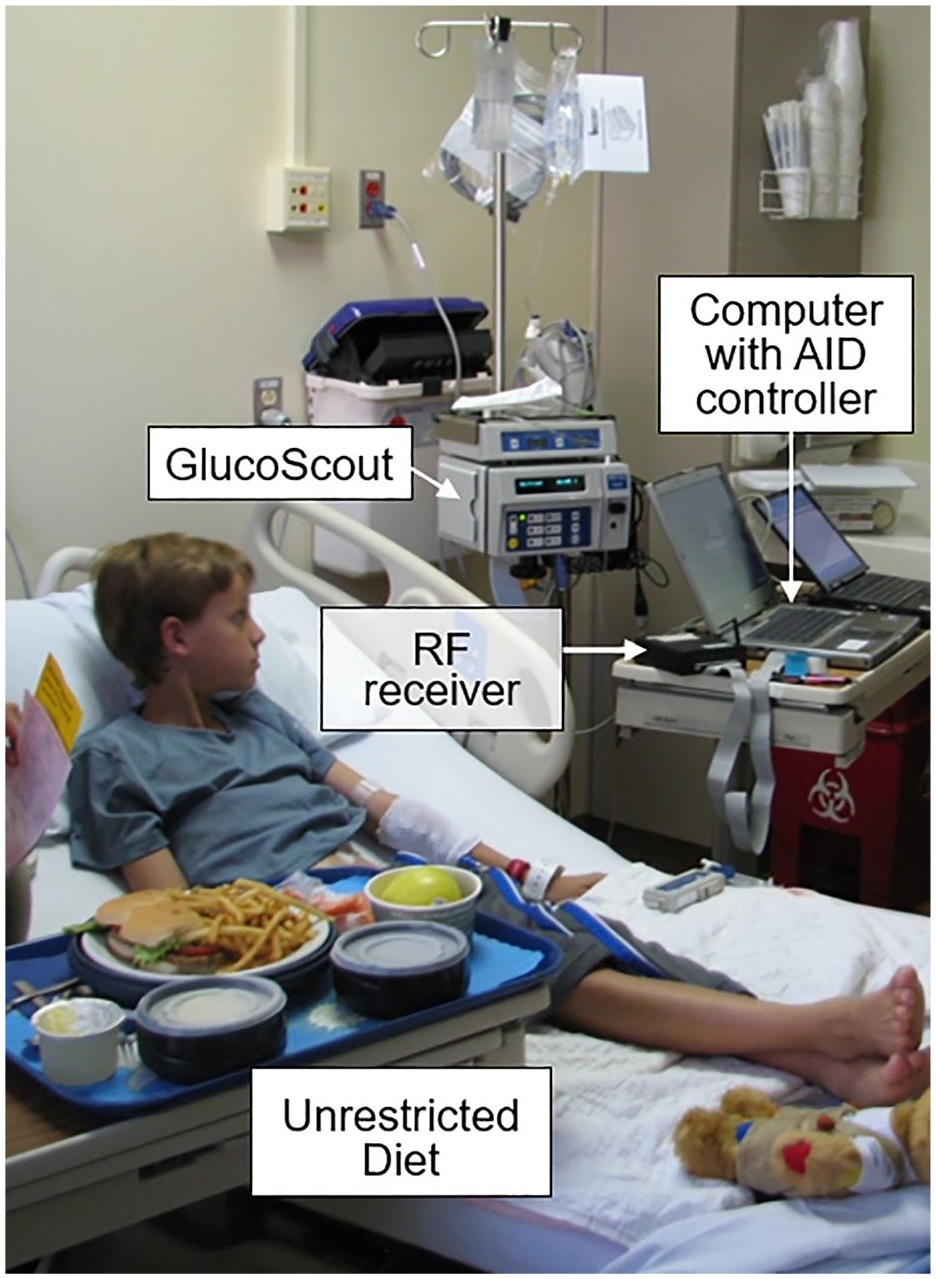
An early inpatient AP system.
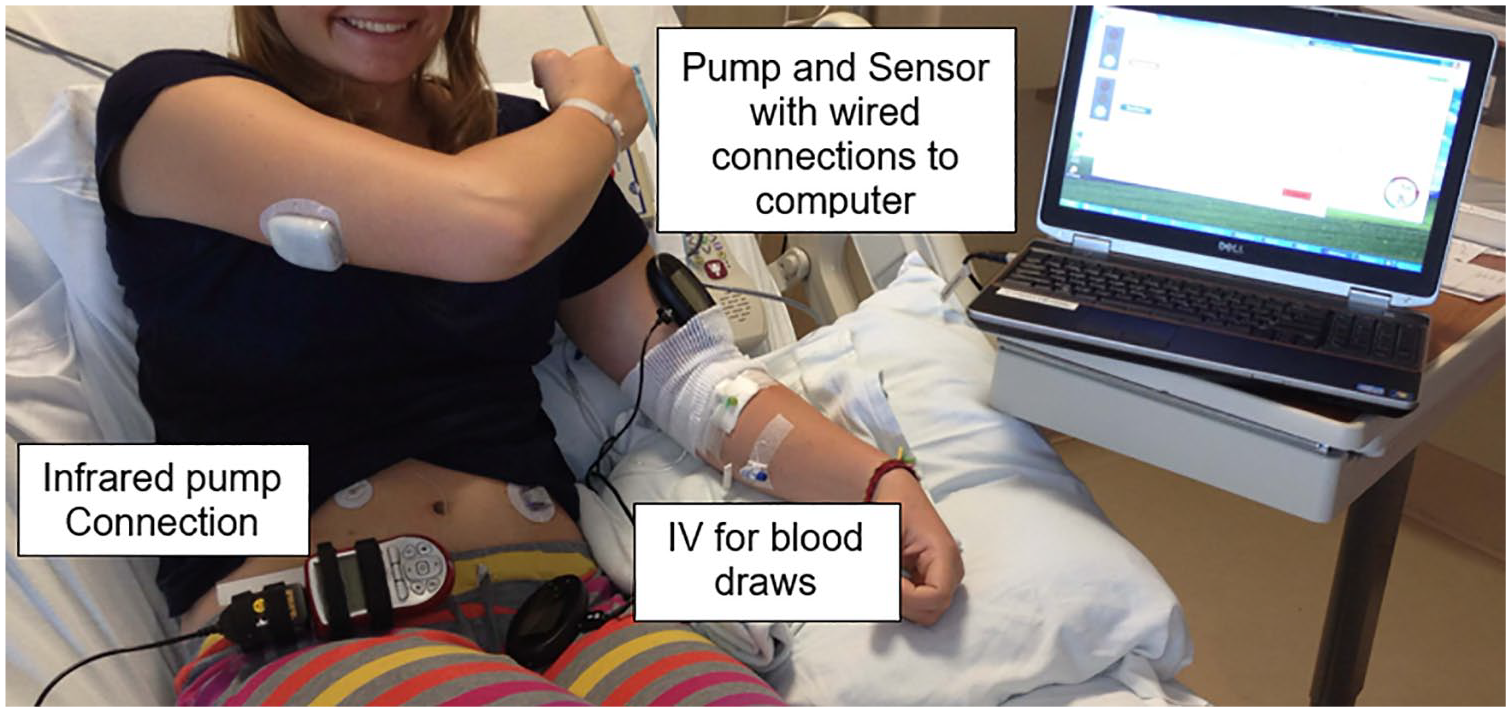
An early automated insulin delivery system.
Outpatient studies using computers, tablets or cell phones: Once venous glucose values were not required, longer at-home unmonitored outpatient studies were done.77-80 With algorithms on phones and tablets connected to cellular networks closed loop systems allowed remote monitoring at diabetes camps and hotels with exercise challenges.38,41,81-87
AID algorithms integrated with insulin pumps: The MiniMed 530G, which was approved by the FDA in 2013, was the first system in the United States to incorporate a feature Threshold Suspend automation, which could automatically halt insulin delivery for up to two hours if sensor glucose values reached a preset low threshold. 88 The Medtronic 670G was the first commercial hybrid AID pump approved for people over 14 years old in 2016, for children seven to 13 in 2018, and two to six years old in 2020. In 2019, the Tandem pump with Control-IQ and its precursor algorithm were tested in children six to 18 years old at ski camps.41,42 Pivotal RCTs of Control-IQ were completed in 14 to 71 year olds in 2019),89,90 six to 13 year olds in 2020, 91 and in two to six year olds in 2023. 92 A three-month OmniPod 5 AID safety study in six to 70 year olds was completed in 2021. 93 The 780G with autocorrection was evaluated in adolescents 94 in a randomized pivotal trial with two to 17 year olds, 95 and in a head-to-head comparison with the 670G. 96 A pivotal RCT of the insulin-only bionic pancreas was completed in participants six to 79 years old in 2022.97,98
Benefits to youth using current AID systems include meeting glycemic goals in the real world99-101 and thereby dramatically reducing the risk of long-term complications, allowing better sleep quality 102 and improvements in neurocognitive and neuroimaging changes associated with hyperglycemia. 103 AID systems are standard of care (“A” level evidence) for children and adolescents with type 1 diabetes,104,105 but there are still major issues to access based on socioeconomic status. 106
The AP in Type 2 Diabetes
1Institute of Metabolic Science-Metabolic Research Laboratories, University of Cambridge, Cambridge, UK
2Center for Diabetes Technology, The University of Virginia, Charlottesville, VA, USA
3University of Cambridge, Cambridge, UK
Outpatient Fully Closed-Loop Approach
The use of automated insulin delivery systems in people with type 2 diabetes (T2D) is limited despite this population having two times higher day-to-day variability in insulin requirements than adults with type 1 diabetes as presented in Figure 7. 107 There is growing evidence from randomized controlled trials that automated insulin delivery systems are safe and effective in this population. In a single-center crossover trial involving 26 people with T2D, CamAPS HX fully-automated closed-loop system (no meal announcements) was safe and associated with increased time in target range (70-180 mg/dL) compared with standard insulin therapy (66% vs 32%) over a period of eight weeks. 108 Fully closed-loop reduced time above range and mean glucose without increasing time in hypoglycemia. Similar benefits were observed when CamAPS HX was used in a more vulnerable population with T2D with end-stage renal disease requiring dialysis. 109 The benefit of a fully closed-loop approach in this population is that it removes the need for carbohydrate counting education and health care professional input for optimization. Larger multicenter trials are required to provide evidence to support widespread adoption and reimbursement.
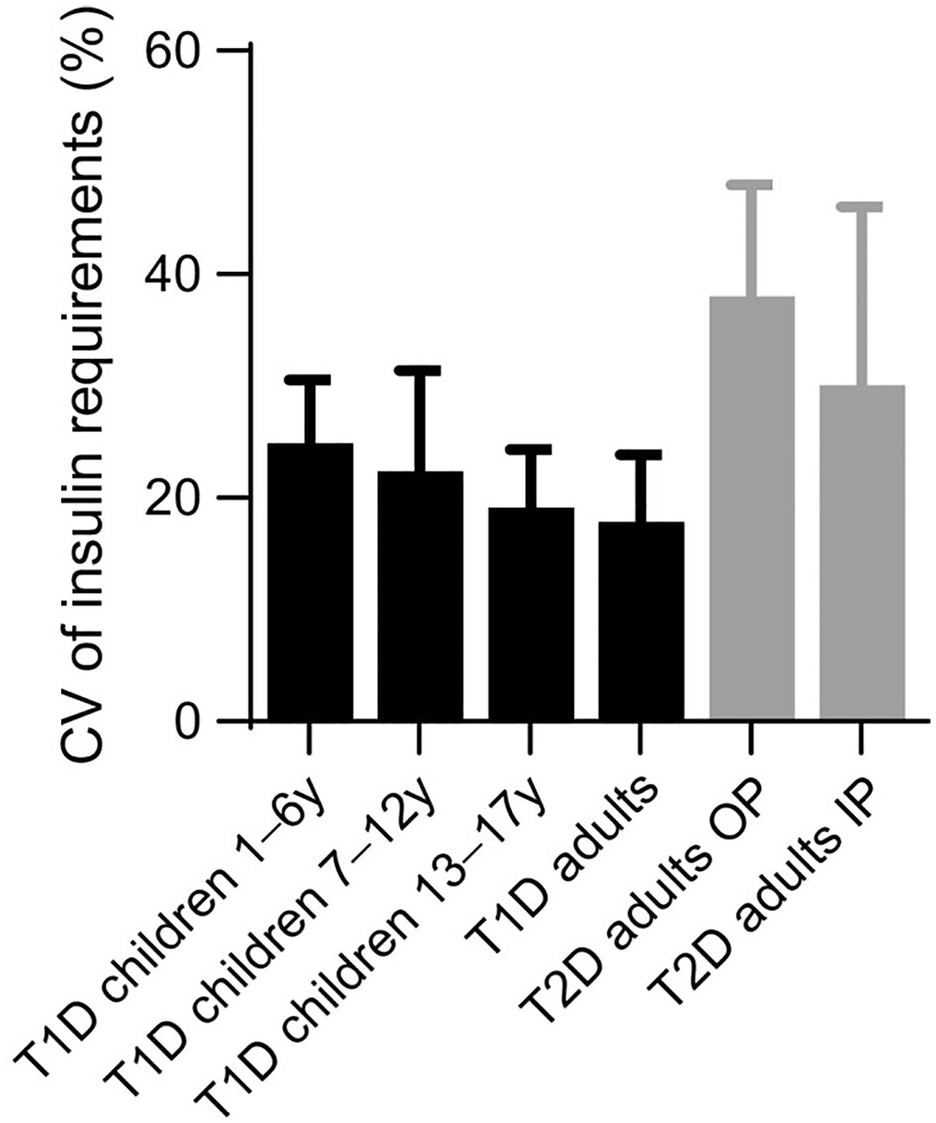
Variability of day-to-day insulin requirements as determined by closed-loop insulin delivery across different populations with diabetes. Data presented are mean with standard deviation. Reproduced from Boughton and Hovorka 110 under the CC-BY 4.0 license (http://creativecommons.org/licenses/by/4.0/).
Outpatient Hybrid Closed-Loop Approach
In a multicenter crossover trial involving 17 adults with T2D using insulin pumps in France, the DBLG1 hybrid closed-loop system improved time in range compared to insulin pump and sensor (76% vs 61%). 111 There was less time with glucose above target during hybrid closed-loop and mean glucose was lower. Improved glucose outcomes were achieved without any increase in hypoglycemia.
Non-randomized before and after studies of hybrid closed-loop systems including Control-IQ and Omnipod 5 have been undertaken in adults with T2D, but efficacy cannot be determined because of the absence of a control group.112,113 Real-world observational data has also been reported for people with T2D using Control-IQ.114,115
Inpatient Fully Closed-Loop Approach
Automated insulin delivery systems have also been deployed in the inpatient setting to address the challenge of glucose management during an acute admission for people with T2D. In a multinational parallel design trial involving 136 people with T2D, a fully-automated closed-loop system was safe and associated with increased time in target glucose range (100-180 mg/dL) compared with standard insulin therapy (66% vs 42%) during the hospital admission. 116 Less time was spent above target and mean glucose was lower in the group using the fully closed-loop system. Importantly there was no difference in time spent in hypoglycemia between groups. Similar results were observed in a study involving inpatients with T2D receiving nutritional support with parenteral and/or enteral nutrition. 117 Implementation of inpatient closed loop requires overcoming several practical challenges including procurement, integration, and reimbursement. 118
Multi-Hormone AP and Adjuvants
1Department of Biomedical Engineering, McGill University, Montreal, QC, Canada
2Harvard Medical School, Harvard University, Boston, MA, USA
3Beta Bionics, Inc, Irvine, CA, USA
4UAB Diabetes Technology Program, The University of Alabama, Birmingham, Birmingham, AL, USA
5Division of Endocrinology & Metabolism, School of Medicine, Indiana University, Indianapolis, IN, USA
6Center for Diabetes and Metabolic Diseases, School of Medicine, Indiana University, Indianapolis, IN, USA
Although automated insulin delivery (AID) has become the standard of care for managing type 1 diabetes (T1D), only 30% to 50% of people with T1D are able to achieve the recommended glycemic targets (A1c <7% or time in range >70%).89,93,96,97,119,120 This disappointing outcome is primarily due to the mismatch between the slow absorption of subcutaneous insulin 121 and rapid absorption of carbohydrate, hindering the ability of the systems to counteract variations in postprandial glucose levels. 122 Moreover, these systems do not address the rising prevalence of obesity in T1D, 123 with its associated increased cardiovascular risk. Closed-loop systems that deliver other hormones or that are augmented with adjuvant therapies have been proposed to address these limitations.
The absorption of subcutaneous glucagon is faster than that of insulin, 121 and thus delivering glucagon is more effective in preventing hypoglycemia than suspending insulin. Therefore, bihormonal insulin-and-glucagon systems can deliver insulin more aggressively to correct hyperglycemia and achieve lower glucose levels than insulin-alone systems, while employing glucagon to mitigate against hypoglycemia.38,124-126 Bihormonal insulin-and-glucagon systems are currently under development.126,127
Pramlintide is an amylin analogue that improves postprandial glycemia by delaying gastric emptying to better match insulin absorption, suppressing nutrient-derived endogenous glucagon secretion, and reducing carbohydrate intake through induction of satiety. 128 Although several short-term studies have shown that bihormonal insulin-and-pramlintide systems may outperform insulin-alone systems, 129 the development of these bihormonal systems hinges on the availability of insulin-pramlintide co-formulations. Several co-formulations are under development. 130
Sodium glucose cotransport inhibitors (SGLTi) have been shown to improve glycemic outcomes, reduce insulin requirement in people with T1D and also have established cardio-renal safety in type 2 diabetes. SGLT2i use along with AID has been shown to reduce hyperglycemia without increasing hypoglycemia.131-133 However, increased risk of diabetic ketoacidosis (DKA) has led to failure to receive approval by the FDA for use in T1D. Potential use of lower dosages of SGLT2i and 134 use of continuous ketone monitoring may be potential remedies for the risk of DKA and remain to be tested in clinical trials.
Long-acting glucagon-like peptide 1 receptor analogs (GLP-1RAs) have demonstrated excellent glycemic, weight, and cardio-renal benefits in those with and without type 2 diabetes. These agents are not approved for T1D; however, off-label use in T1D is increasing135,136 and real-world studies have shown promising results.137,138 A few randomized trials are ongoing to assess the benefits and risks of these agents as an adjuvant to AID.
Footnotes
Acknowledgements
The authors thank Alessandra T. Ayers and Cindy N. Ho for their careful editorial assistance.
Abbreviations
ADICOL, advanced insulin infusion using a control loop; AID, automated insulin delivery; APS, artificial pancreas system; BLE, Bluetooth low energy; CGM, continuous glucose monitoring; CGMS, continuous glucose monitor system; CV, coefficient of variation; DiAs, Diabetes Assistant; DIY, do-it-yourself; DKA, diabetic ketoacidosis; FDA, Food and Drug Administration; GLP-RAs, glucagon-like peptide 1 receptor analogs; HbA1c, hemoglobin A1c; iAPS, interoperable artificial pancreas system; IP, inpatients; IV, intravenous; JDRF, Juvenile Diabetes Research Foundation; MPC, model predictive control; NIDDK, National Institute of Diabetes and Digestive and Kidney Diseases; NIH, National Institutes of Health; OHSU, Oregon Health Sciences University; OP, outpatients; PDM, personal data manager; PID, proportional integral derivative; RCT, randomized controlled trial; RF, radiofrequency; SAP, sensor-augmented pump; SC-SC, subcutaneous glucose sensing and subcutaneous insulin; SGLTi, sodium glucose cotransport inhibitors; STS, static transfer switch; T1D, type 1 diabetes; T2D, type 2 diabetes; TBR, time below range; TIR, time in range; UI, user interface; US, United States.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: At the time of the research reported here, H.Z. was employed by Sansum Diabetes Research Institute. H.Z. was subsequently employed by Insulet, Google, and Supersapiens. He holds patents on artificial pancreas technology. H.Z. received product support from Insulet Corporation, Roche, and Dexcom during this research. The Juvenile Diabetes Research Foundation, the Helmsley Charitable Trust, and the National Institutes of Health (NIDDK) provided research funding. H.Z. has received royalty payments from patents in his portfolio that have been licensed to Insulet and Animas. R.H. has received speaker honoraria from Sandoz, Abbott Diabetes Care, and NovoNordisk, receiving consultancy fees from Abbott Diabetes Care, patents related to closed-loop, and is director at CamDiab. A.B. received research funding from Medtronic. T.B. served on advisory panels of Novo Nordisk, Sanofi, Eli Lilly, Boehringer, Medtronic, Abbott, and Indigo Diabetes. T.B. received honoraria for participating on the speaker’s bureau of Eli Lilly, Novo Nordisk, Medtronic, Abbott, Sanofi, Dexcom, Aventis, Astra Zeneca, and Roche. T.B.’s institution received research grant support from Abbott, Medtronic, Novo Nordisk, Sanofi, Novartis, Sandoz, and Zealand Pharma, Slovenian Research and Innovation Agency, the National Institutes of Health, and the European Union. C.K.B. has received consultancy fees from CamDiab, Speaker Honoraria from Ypsomed, and research support from Dexcom, Abbott Diabetes Care, YpsoMed, and CamDiab. M.D.B. report research funding through his institution from Dexcom, Tandem, and Novo Nordisk. M.D.B. reports honoraria/consulting activities with Boydsense, Roche, Sinocare, and Tandem. M.D.B. reports royalties managed by his institution from Dexcom, Lifescan, NovoNordisk, and Sanofi. B.A.B. has received research support from Medtronic Diabetes, Tandem, Insulet, Bigfoot, Dexcom, Abbott, Convatec, and Lilly. B.A.B. serves on the advisory boards for Medtronic, Tandem, Animas, Sanofi, Roche, Novo-Nordisk, Lilly, Convatec, BD, Profusa, and Glysense. S.A.B. has received research support to her institution from Dexcom, Insulet, Roche, Tandem, and Tolerion and has participated in a data management board for MannKind. D.R.C. has no disclosures relevant to this work. E.D. has received personal fees from Roche and Eli Lilly and Company; holds patents on artificial pancreas technology; and has received product support from Insulet Corporation, Tandem Diabetes Care, Roche, and Dexcom, Inc. The work presented in this article was performed as part of his academic appointment and is independent of his employment with Eli Lilly and Company. M.D.D. has received research support from Tandem, Dexcom, Medtronic. F.J.D. reports funding over the time course described in this article from the Showalter Foundation, Roche Diagnostics Corporation, the Juvenile Diabetes Research Foundation, the Helmsley Charitable Trust, and the National Institutes of Health (NIDDK). F.J.D. also reports royalty payments from patents in a portfolio that have been licensed to Insulet, Roche, and Dexcom. L.E. receives salary support through and NIDDK career development research grant. L.E.’s institution has received research support from Breakthrough T1D, Medtronic, Mannkind, and Abbott. She has served on the advisory board of Abbott, Diabetes Center Berne, Sequel, and Medtronic. She has received consulting fees from Jaeb and Tandem Diabetes Care, and has received honorarium fees from Med Learning Group (Sanofi-sponsored grant), Tandem Diabetes Care, Medtronic, and Insulet. C.F. reports receiving research support from Novo Nordisk, Dexcom, and Tandem Diabetes Care handled by the University of Virginia, and patent royalties from Novo Nordisk and Dexcom handled by the University of Virginia’s Licensing and Ventures Group. G.P.F. conducts research supported by Medtronic, Dexcom, Abbott, Tandem, Insulet, and Beta Bionics and has been a speaker/consultant/advisory board member for Medtronic, Dexcom, Abbott, Tandem, Insulet, Beta Bionics, Sequel, and Lilly. A.H. has acted as a consultant for Eli Lilly and Abbott Diabetes Care, and has received drugs, supplies, equipment, and other in-kind support from Tandem, Adocia, Dexcom, Eli Lilly, and Ypsomed. D.C.K. is a consultant for Afon, Embecta, GlucoTrack, Lifecare, Novo, Samsung, and Thirdwayv. B.K. reports research grants handled by the University of Virginia from the National Institutes of Health, Novo Nordisk, Dexcom, and Tandem Diabetes Care. In addition, B.K. has a number of patents with royalties paid to Dexcom and Novo Nordisk. A.J.K. is the Chief Executive Officer at Breakthrough T1D. C.J.L. has received research support from Tandem Diabetes, Dexcom, Abbott, and Insulet paid to my institution (within last three years). C.J.L. have served as a consultant for Tandem Diabetes and Dexcom (within the last three years). Y.C.K. is a consultant for Novo Nordisc, Tandem Diabetes Inc, Vertex pharmaceuticals. Y.C.K. has industry studies with compensation to the following employers: Medtronic, Tanem, Dexcom, Novo Nordisk, and Sanofi. Y.C.K. supported products from Dexcom, Tandem, and Insulet. D.M.M. has had research support from the NIDDK, NIH, Breakthrough T1D, and Helmsley Charitable Trust. His institution has had research support from DexCom Inc., Medtronic, Insulet, Bigfoot Biomedical, Tandem, and Roche; and he has consulted for Abbott, the Leona M. and Harry B. Helmsley Charitable Trust, Lifescan, Sanofi, Medtronic, Provention Bio, Kriya, Novo Nordisk, Eli Lilly, Insulet, Biospex, and Bayer. M.P. is an advisory board member for AstraZeneca, Eli Lilly, Mannkind, Medtronic Diabetes, Pfizer, Sanofi, DOMPE, LifeScan, Novo Nordisk, Insulet, ProventionBio, Merck, Ascensia, Bayer, embecta, and Tandem. M.P. has received consulting fee from Qulab Medical, and ProventionBio. M.P. has received research grants from Eli Lilly, Medtronic Diabetes, Novo Nordisk, Pfizer, Sanofi, DreaMed Diabetes, NG Solutions, DOMPE, Lumos, Gwave, OPKO, ProventionBio, AstraZeneca, and Omega Galil. M.P. owns stocks from DreaMed Diabetes and NG Solutions. E.R. declares consultant/speaker fees from A. Menarini Diagnostics, Abbott, Air Liquide SI, Astra-Zeneca, Becton-Dickinson, Boehringer-Ingelheim, Cellnovo, Dexcom Inc., Eli-Lilly, Hillo, Insulet Inc., Johnson & Johnson (Animas, LifeScan), Medtronic, Medirio, Novo-Nordisk, Roche, and Sanofi-Aventis and research support by Abbott, Dexcom Inc., Insulet Inc., Roche, and Tandem Diabetes Care. S.J.R. is an employee and stockholder of Beta Bionics, Inc. V.N.S.’s institution has received research support from Enable Bioscience, Zucara Therapeutics, Lilly, Cystic Fibrosis Foundation, Breakthrough T1D, and NIH. V.N.S. has received personal fees from Sanofi, Lilly, NovoNordisk, Dexcom, Tandem Diabetes Care, Insulet, Sequel Med Tech, Embecta, Genomelink, and Lumosfit for consulting, advising, or speaking; outside of this submitted work. G.M.S. was employed by Medtronic/MiniMed and was one of the original developers of the Medtronic/MiniMed closed-loop insulin delivery algorithm. G.M.S. currently works at Boston Children’s Hospital where he is developing closed-Loop insulin delivery algorithms for use in the Intensive Care Unit. G.M.S. has served as a consultant to Abbott Diabetes Care, Roche Diabetes Care, and Eli Lilly. R.P.W. conducts research funded by Medtronic, Dexcom, Abbott, Tandem Diabetes Care, Insulet, and Eli Lilly and has been a consultant for Dexcom and Tandem Diabetes Care and an advisory board member for Sequel Med Tech. S.A.W. has conducted clinical trials for Abbott Diabetes, JDRF/Breakthrough T1D, Medtronic, and Tandem; has received honoraria for consulting or speaking engagements from Abbott, Dexcom, Insulet, Medtronic, and Tandem; and has served on advisory boards for Zealand. He currently receives research support (to his institution) from Abbott Diabetes.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
