Abstract
The mainstay of type 1 diabetes (T1D) management in pregnancy is optimization of glucose levels in a tight range. Achieving euglycemia has been revolutionized by advances in diabetes technology, including the development of automated insulin delivery (AID) systems. A small but growing population of gravidas with T1D elects to pursue off-label use of AID systems in pregnancy, and their outcomes have been described in numerous observational cohorts. This review aims to aggregate data from all available observational studies examining glycemic, maternal, and neonatal outcomes associated with antenatal AID use. A total of 243 pregnancies managed antenatally with AID were described in 24 publications, with largely reassuring outcomes data. Time in range (TIR) with commercial AID systems was generally acceptable, with many patients reaching pregnancy target TIR > 70% by the third trimester. Time in range with open-source AID systems appeared even higher, although with the potential tradeoff of worse time below range (TBR). Clinically, there do not appear to be major differences in pregnancy outcomes between AID systems and other methods of insulin delivery, although this assumption is based largely on indirect comparisons with other population-level reports rather than direct comparisons within analytic observational cohorts. Clinical outcomes appear superior with open-source AID compared with commercial AID, although this should be interpreted with caution based on the small sample size of this subpopulation (n = 16) and potential confounding. The real-world evidence generated by these observational studies provides invaluable information for patients and providers seeking to improve outcomes for gravidas with T1D.
Introduction
As the prevalence of type 1 diabetes (T1D) in pregnancy rises, more individuals will be susceptible to the maternal and neonatal complications associated with this condition.1-4 Minimizing those complications by optimizing glucose levels is the mainstay of antenatal T1D management. Achieving euglycemia has been revolutionized by advances in technology, including continuous glucose monitors (CGMs) and insulin pumps. 5 Automated insulin delivery (AID) systems combine these two technologies, using data from CGM to automatically adjust the amount of insulin delivered via pumps. 6 These systems are now recommended by the American Diabetes Association (ADA) as the preferred method of insulin delivery for adults with T1D. 5 Multiple AID systems are commercially available and used by an increasing population of gravidas with T1D, despite not having been developed specifically for use in pregnancy. At the same time, a smaller but growing population has opted for antenatal use of open-source AID systems (also known as “hacked” or “do-it-yourself” [DIY] loops), which link CGM to pumps via unregulated open-source software that allows for the highly customizable algorithms for AID.7-9
Given the growing popularity of these systems, understanding outcomes associated with AID use in pregnancy is critically important for patients and providers. This review was undertaken to describe the impact of antenatal use of AID on glycemic, maternal, and neonatal outcomes from observational studies of pregnant individuals with T1D.
Methods
Embase, MEDLINE, and Web of Science were queried to identify observational studies published between January 2014 and November 2024 describing the use of AID by gravidas with T1D. Search terms are included in Supplemental Table 1. Reference lists of relevant publications were also searched to identify any additional publications.
After removing duplicates, a total of 168 unique records were identified (Supplemental Figure 1). On initial screening, 137 records were excluded, leaving 31 publications for detailed review. An additional four were excluded for describing only intrapartum and/or postpartum AID use. One publication was excluded for noncontinuous use of AID (48 hours at 20.7 weeks of pregnancy). 10 To avoid double-counting observations, another publication was excluded due to presumed overlap between the first publication (n = 52 from 2017 to 2022) and the second publication by the same group (n = 62 from 2017 to 2023, with identical inclusion criteria). 11 A similar pattern was seen with another group, with n = 6 individuals from the first publication explicitly included among the n = 21 in the second publication. 12 Because the first publication compared outcomes with and without AID use, it was included for the purposes of narrative review but excluded for the purposes of data aggregation. Ultimately, this narrative review included 25 observational studies examining clinical and/or glycemic outcomes with antenatal use of AID, with data aggregated from 24 observational studies.
Glycemic outcomes of interest were metrics found in CGM reports, specifically mean sensor glucose, coefficient of variation (CV), glucose management index (GMI), and time spent in, above, and below range. Pregnancy-specific range was used, with time in range (TIR) defined as 63 to 140 mg/dL, time above range (TAR) as >140 mg/dL, and time below range (TBR) as <63 mg/dL, unless otherwise specified. Pregnancy goals included TIR > 70%, TAR < 25%, and TBR < 4%, per an expert consensus statement published in 2019. 13
Clinical outcomes of interest were maternal and neonatal outcomes, specifically cesarean delivery, preeclampsia, gestational age at delivery, preterm birth (PTB), birthweight, large for gestational age (LGA), neonatal hypoglycemia (NHG), diabetic ketoacidosis (DKA), and severe maternal hypoglycemia. These outcomes have been identified by an international Delphi consensus of key stakeholders as part of the core outcome set for studies of pregnant people with pregestational diabetes. 14 While many clinical outcomes have clear and widely accepted definitions (eg, cesarean delivery and DKA), others remain more ambiguous (eg, NHG and severe maternal hypoglycemia). For the purposes of this review, clinical outcomes were defined as per the original authors. Of note, other components of the core outcome set (eg, shoulder dystocia, stillbirth, and neonatal death) were not included in this review as they were not reported by the observational studies included herein.
Findings
A total of 243 pregnancies managed antenatally with AID were described in 24 publications (Supplemental Table 2), with 227 using commercial AID and 16 using open-source AID. Table 1 summarizes baseline characteristics of this population of young gravidas with long-standing but reasonably well-controlled diabetes. The characteristics of the open-source AID subgroup are notable, with preconception A1c of 6.0 and 0% incidence of microvascular complications. The characteristics of this population should be highlighted when considering the generalizability of these findings to other diabetes-in-pregnancy populations.
Baseline Characteristics of Pregnant Individuals With Type 1 Diabetes (T1D) Who Used Automated Insulin Delivery (AID) Systems During Pregnancy, by Type of AID System Utilized, Aggregated From 24 Publications.

Categorical data presented as % (n/N) and continuous data presented as mean ± SD (N).
Glycemic Outcomes
Aggregate data regarding glycemic outcomes are summarized in Table 2 and in Figure 1.
Glycemic Metrics Among Individuals With Type 1 Diabetes (T1D) Who Used Automated Insulin Delivery (AID) Systems During Pregnancy, by Trimester of Pregnancy and by Type of AID System Utilized, Aggregated From 24 Publications.

All data presented as mean ± standard deviation (N). Time in range (TIR) defined as 63-140 mg/dL and time below range (TBR) defined as <63 mg/dL, except for N = 6 overall (n = 4 commercial AID and n = 2 open-source AID) from single study that defined TIR as 60-140 mg/dL and TBR as <60 mg/dL. 15 Time above range defined as 140 mg/dL.
The data above include findings from pregnant individuals with T1D using both early generation and modern AID systems, including the following: Diabeloop, Medtronic MiniMed 640G/670G/780G, Omnipod 5, open-source systems, Tandem Basal IQ/Control IQ, and Zone-MPC CLC-P.
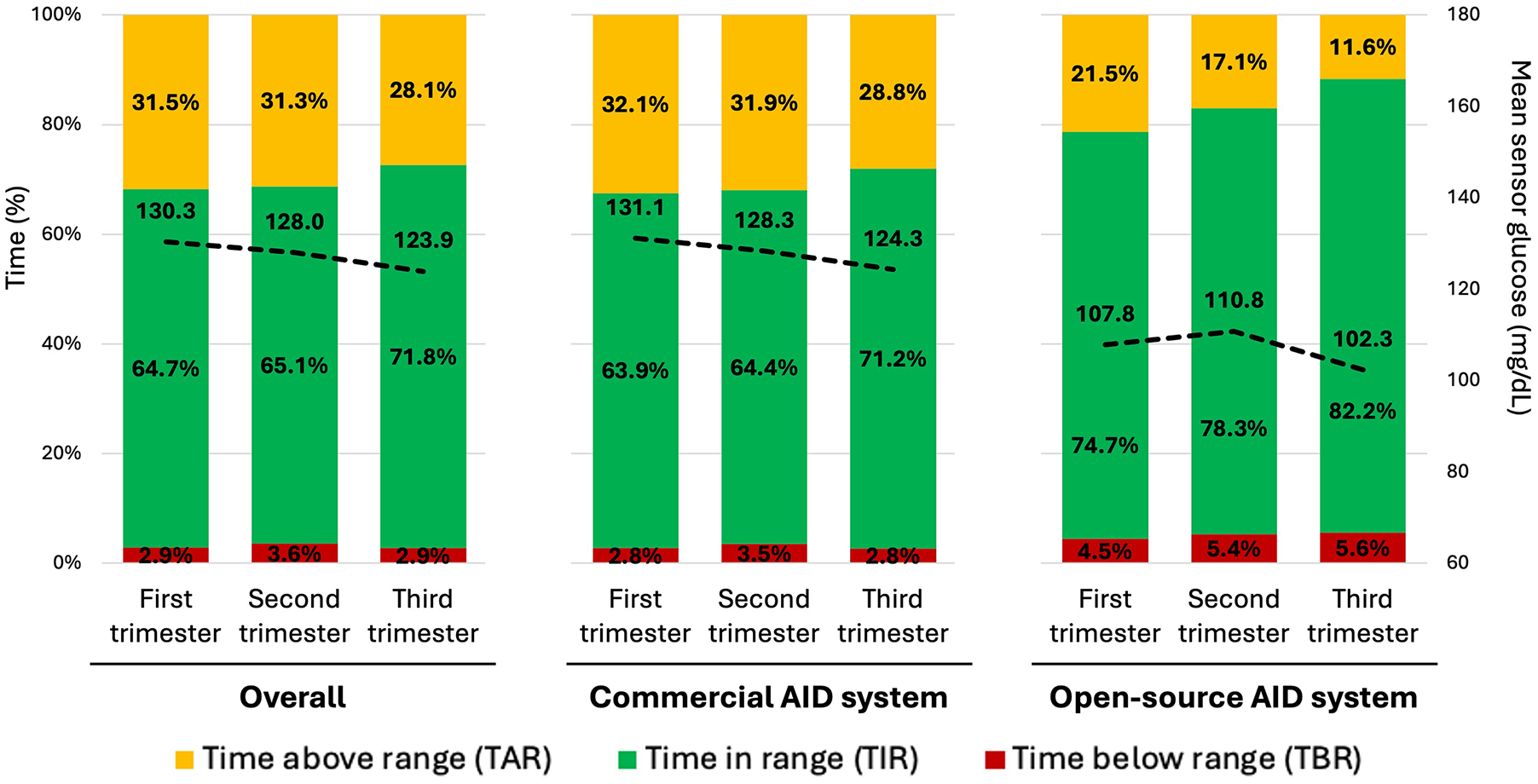
Glycemic metrics among individuals with type 1 diabetes (T1D) who used automated insulin delivery (AID) systems during pregnancy, by trimester of pregnancy and by type of AID system utilized, aggregated from 24 publications.
Descriptive studies of commercial AID systems
The first observational study describing a commercial AID system in pregnancy included three gravidas who used Medtronic MiniMed 670G for at least the latter half of pregnancy. 16 Because the CareLink software at the time did not allow for changes in target range from the default of 70 to 180 mg/dL, pregnancy-specific TIR, TAR, and TBR were not reported. Mean sensor glucose improved over gestation, from 162.8 mg/dL in the first trimester to 136.3 mg/dL in the third trimester. Of note, the target glucose in the 670G system can only be set as low as 120 mg/dL, and this early generation system is currently being phased out of clinical practice. The more modern Medtronic MiniMed 780G system allows for a glucose target as low as 100 mg/dL, and observational reports of its use in pregnancy have suggested superiority over the older system. For example, one report included six individuals using Medtronic MiniMed 780G from the preconception period through the postpartum period and demonstrated persistently high TIR throughout gestation (75%, 79%, and 83% in first, second, and third trimesters), with mean sensor glucose ranging from 113 mg/dL in the first trimester to 106 mg/dL in the third trimester. 17 These impressive TIR metrics were achieved without hypoglycemia, with TBR remaining ≤1% throughout pregnancy.
Although other studies have not described as globally high TIR as that report, many demonstrate a pattern of improving TIR across pregnancy. Among 21 pregnant users of Medtronic MiniMed 780G, mean TIR improved from 64% in the first trimester to 75.7% in the third trimester, with 81% of participants achieving TIR >70% by the third trimester. 18 A comparable pattern was seen in four users of Tandem Control IQ, with improvement in mean TIR from 64.2% in the first trimester to 76.9% in the third trimester, with 100% achieving TIR >70% by the third trimester. 19 Similarly, mean TIR increased from 69.8% in the first trimester to 78.5% in the third trimester among four pregnant users of commercial AID systems (n = 2 Omnipod 5, n = 2 Tandem Control IQ) described in a recent observational cohort. 15 The most dramatic improvement in TIR was described in a case report of one gravida who used Diabeloop DBL-hu and experienced an improvement in TIR from 28.8% in the first trimester to 80.3% in the late third trimester while maintaining TBR <3% throughout pregnancy. 20
However, not all studies report high third trimester TIR and many report TIR below the pregnancy goal of >70%. For example, the largest observational study of Medtronic AID use included 62 patients using the 640G, 670G, or 780G systems, with mean TIR 66.8% in the third trimester, with <50% of the population reaching TIR >70% by the third trimester. 21 A smaller retrospective cohort of eight patients using Tandem AID systems (ie, Basal IQ or Control IQ) reported mean TIR 67.9% throughout pregnancy, 22 and the first report of Omnipod 5 in pregnancy included 17 pregnant users whose mean TIR never reached >70%. 23 In fact, in this study, TIR remained relatively unchanged throughout gestation, ranging from 65% at 12 weeks, to 58% at 20 weeks, to 59% at 32 weeks.
Even in the absence of at-goal TIR, many descriptive cohorts report low TBR and low CV with AID use. In three separate observational studies of Medtronic MiniMed 780G (n = 6, n = 8, and n = 13), TIR never exceeded 70%, but TBR remained <3% and CV remained ≤33% throughout gestation.24-26
Comparative studies of commercial AID systems
Multiple case reports have compared glycemic metrics in the same pregnant individual before and after initiation of AID (at 13, 16, 23, and 34 weeks).27-30 In each of these cases, initiation of AID was followed by improvement in glycemic metrics, including reduction in TAR from 50.2% before to 35.1% after; 27 reduction in TBR from 5% before to 2% after; 28 improvement in TIR from 62% before to 70% after; 29 and improvement in CV from 39% before to 30% after. 30
The largest within-pregnancy comparison of glycemic outcomes before and after AID was published by a group that developed a pregnancy-specific AID system using a zone model predictive controller (CLC-P) and reported on the outcomes in 10 individuals who started CLC-P in the second trimester. 31 Glycemic outcomes during CLC-P use were compared with the one-week to two-week run-in phase prior to CLC-P use. The CLC-P use was associated with statistically significant improvements in TIR (64.5% before vs 78.6% after, P = .002), TAR (29.8% before vs 19.7% after, P = .037), and TBR (3.7% before vs 1.6% after, P = .033). The vast majority (90%) were able to achieve the recommended TIR >70% with CLC-P use. Notably, there were two episodes of DKA related to infusion set occlusions with CLC-P use, neither of which required hospital admission. This system is unique in its ability to target a glucose value as low as 80 mg/dL, which is particularly useful for the pregnant T1D population, and which may explain the very positive glycemic outcomes associated with CLC-P use in pregnancy.
Two additional observational studies provide between-pregnancy comparisons of commercial AID versus other methods of insulin delivery. The first compared glycemic outcomes in a first pregnancy managed with sensor-augmented pump (SAP) with those in a subsequent pregnancy managed with Medtronic MiniMed 780G, within the same six individuals. 12 This direct comparison found that AID was associated with significantly improved second trimester TIR (78.6% AID vs 69.1% SAP, P = .045) and second trimester TAR (16.6% AID vs 26.6% with SAP, P = .045). A larger study compared 53 users of multiple daily injections (MDI) with 59 users of AID (Diabeloop, Medtronic MiniMed 780G, or Tandem Control IQ). 32 Although there were no statistically significant differences in first, second, or third trimester TIR or TAR between the two groups, second and third trimester TBR were significantly lower in the AID group (2% AID vs 3% MDI in second trimester, P < .05; 1% AID vs 3% MDI in third trimester, P < .05). A similar pattern was seen with second and third trimester CV (29.1% AID vs 31.9% MDI in second trimester, P < .05; 27.2% AID vs 29.1% MDI in third trimester, P < .05).
Descriptive studies of open-source AID systems
The first report of open-source AID system in pregnancy described TIR >70% throughout pregnancy in one gravida (74%, 76%, and 77% in first, second, and third trimesters). 33 High TIR throughout pregnancy has been persistently demonstrated in other observational cohorts of open-source AID use (eg, 82%-89% in one report of two gravidas; 15 79.6%-85.2% in another report of two gravidas; 34 and ≥80% in three of the five gravidas in the largest report to date). 35
Notably, many studies demonstrated higher-than-recommended TBR with open-source AID. For example, an observational cohort of four gravidas demonstrated persistently high TIR (72.7%-82.5%) but TBR >4% throughout pregnancy (4.9%, 6.1%, and 7.4% in first, second, and third trimesters). 36 A similar glycemic pattern was demonstrated in another report of one open-source AID user, whose improvement in TIR (63% prior to conception to 72% in third trimester) was accompanied by worsening TBR (3% prior to conception to 7% in third trimester). 37 Despite these findings, none of the publications reported any episodes of severe maternal hypoglycemia, typically defined as symptomatic and requiring third-party assistance.
Maternal and Neonatal Outcomes
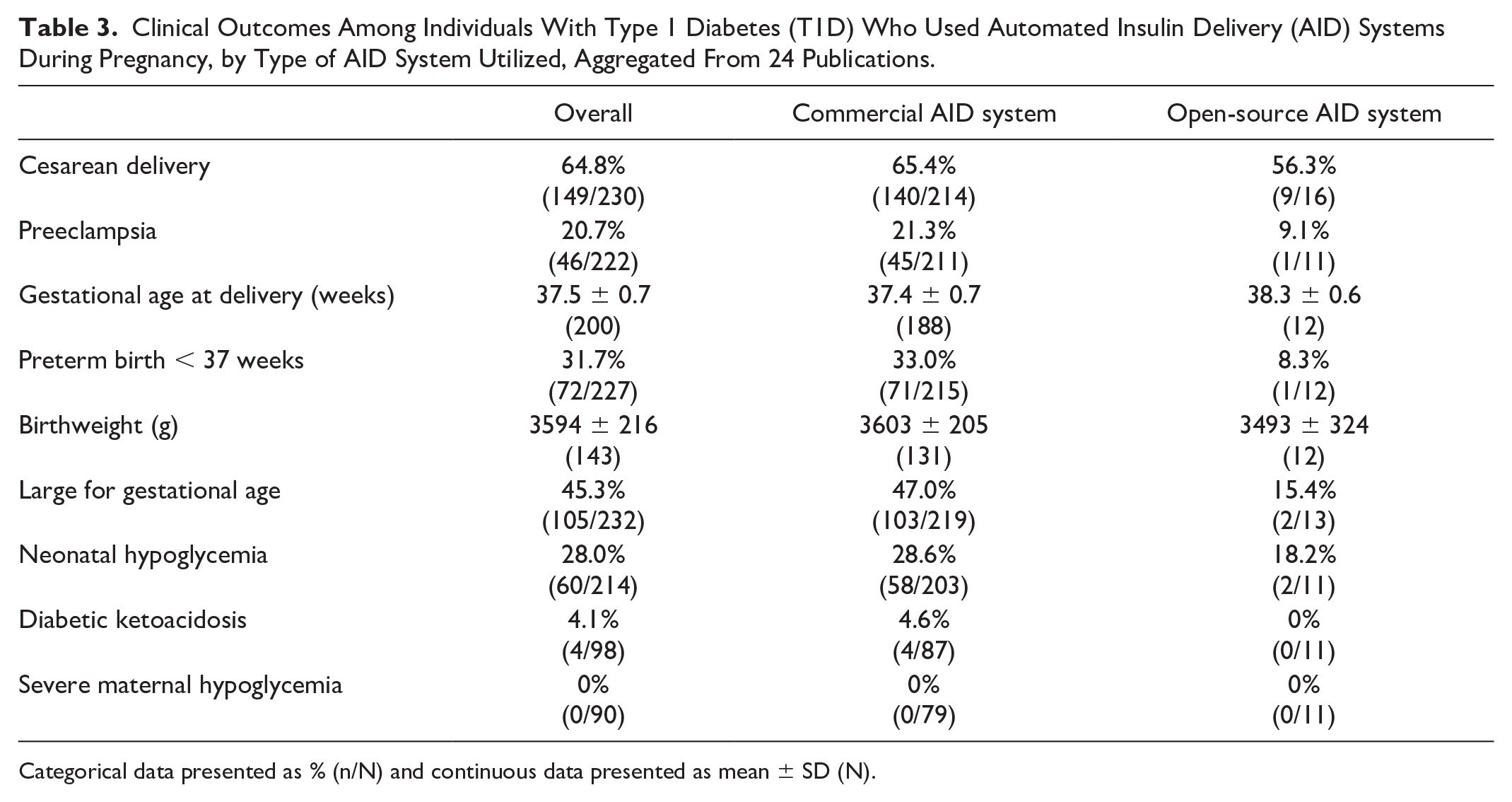
There is considerable inconsistency in the reporting of clinical outcomes in observational studies examining AID in pregnancy. For example, of the 24 studies included here, more than one-third failed to report preeclampsia as an outcome. Preeclampsia is found at a high incidence in gravidas with T1D, has short- and long-term sequelae for both mothers and babies,38,39 and contributes to considerable medical, psychosocial, and economic burden.40-43 Unfortunately, the absence of information regarding this critically important outcome is not unique, and many of the outcomes identified as the core outcome set for studies of T1D in pregnancy are not mentioned in the included studies. 14 Aggregate data regarding available pregnancy outcomes are summarized in Table 3 and Figure 2.
Clinical Outcomes Among Individuals With Type 1 Diabetes (T1D) Who Used Automated Insulin Delivery (AID) Systems During Pregnancy, by Type of AID System Utilized, Aggregated From 24 Publications.
Categorical data presented as % (n/N) and continuous data presented as mean ± SD (N).
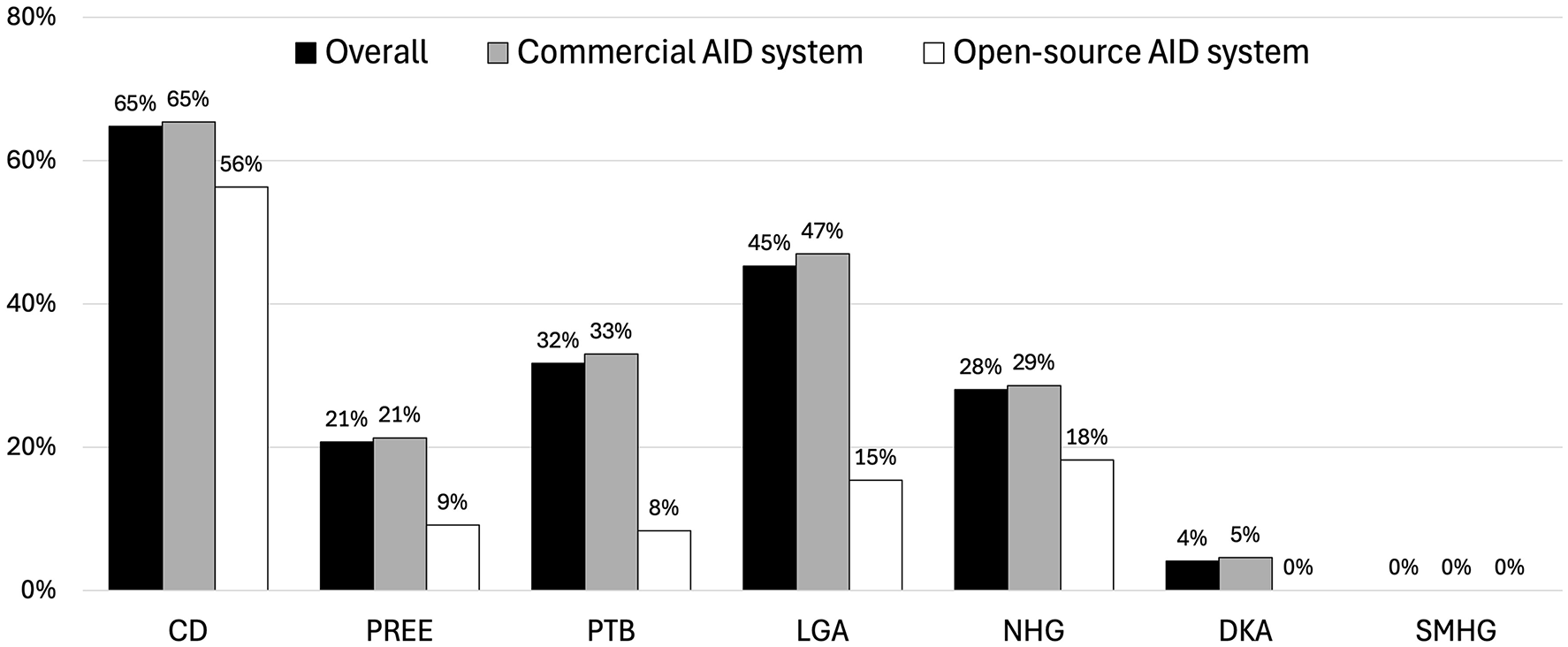
Clinical outcomes among individuals with type 1 diabetes (T1D) who used automated insulin delivery (AID) systems during pregnancy, by type of AID system utilized, aggregated from 24 publications.
Maternal and neonatal outcomes with commercial AID systems
Aggregate data from pregnant users of commercial AID demonstrate high incidence of known complications of T1D in pregnancy, including 65% cesarean delivery, 21% preeclampsia, 32% PTB, 47% LGA, and 28% NHG. While most publications included here are descriptive cohorts without comparison groups, many authors note that their reported rates of clinical complications with AID are comparable with those described in population-level cohorts of gravidas with T1D using other methods of insulin delivery. One observational cohort directly compared AID users with MDI users and found more excessive gestational weight gain with AID use (52% AID vs 25% MDI, P = .003) but otherwise no statistically significant differences in other pregnancy outcomes. 37
Maternal and neonatal outcomes with open-source AID systems
Although direct comparisons are not available, data aggregated in this review suggest that open-source AID may be associated with improved clinical outcomes when compared with commercial AID. For example, the incidence of preeclampsia, PTB, and LGA were all twofold to fourfold lower among open-source AID users compared with commercial AID users. While it is possible that this apparent effect is due to the flexibility and customization inherent to open-source AID, it is also possible that this effect is secondary to confounding factors, including publication bias and/or baseline differences in diabetes severity, degree of diabetes self-management skills, and/or comfort with advanced diabetes technology.
Conclusion
Multiple retrospective observational cohorts have examined the use of AID in pregnancy and provided promising data for positive glycemic and clinical outcomes. The TIR with commercial AID systems is generally acceptable, with many patients reaching pregnancy target >70% by the third trimester. Even those who do not reach this target are able to maintain low TBR and low CV throughout pregnancy. The TIR with open-source AID systems appears even better, although with the potential tradeoff of a worse TBR. Clinically, there do not appear to be major differences between AID systems and other methods of insulin delivery, based on one direct comparison in an analytic observational cohort and many indirect comparisons with other population-level reports. Notably, clinical outcomes appear superior with open-source AID compared with commercial AID, although this should be interpreted with caution based on the small sample size and potential confounding.
The impact of these observational studies is twofold. First, they have provided important preliminary data to support the development of randomized clinical trials of AID use in pregnancy. Second, they have generated real-world evidence, which has not traditionally been considered the gold standard of evidence but is increasingly recognized as more relevant to real-world decision-making.44,45
Larger observational cohorts are needed to better understand the impact of AID system use in pregnancy, especially with direct comparisons between different types of AID systems. Glycemic and clinical outcomes associated with AID in pregnancy are only expected to improve over time as patients and providers become more comfortable with these systems, learn how to optimize preexisting AID systems to perform for pregnancy, and ultimately develop pregnancy-specific algorithms for widespread use. New and upcoming technologies that hold particular promise for use in pregnancy include AID systems with pregnancy-specific settings (eg, CamAPS FX) and/or lower glucose targets (eg, Twiist pump with Loop algorithm, which provides the option of setting glucose target as low as 87 mg/dL). Future studies should also assess outcomes beyond the standard glycemic and clinical outcomes, including patient-reported diabetes distress, provider experience, and cost-effectiveness.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251327603 – Supplemental material for Impact of AID on Glycemic Profile and Maternal/Neonatal Outcomes in Pregnancy: A Review of the Evidence From Observational Studies
Supplemental material, sj-docx-1-dst-10.1177_19322968251327603 for Impact of AID on Glycemic Profile and Maternal/Neonatal Outcomes in Pregnancy: A Review of the Evidence From Observational Studies by Nasim C. Sobhani in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
AID, automated insulin delivery; CGM, continuous glucose monitor; DKA, diabetic ketoacidosis; LGA, large for gestational age; MDI, multiple daily injections; NHG, neonatal hypoglycemia; PTB, preterm birth; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
