Abstract
Background:
Evaluation of the performance of Cobas® Pulse (Roche Diagnostics GmbH, Mannheim, Germany), a new blood glucose (BG) monitoring system (BGMS; referred to here as BGMSA) intended for point-of-care testing using samples from patients in diverse clinical settings by intended point-of-care test operators.
Methods:
Arterial, capillary, venous, or heel stick whole blood (WB) samples from patients in non-critical and critical care settings were collected and analyzed using BGMSA and the Nova StatStrip® BGMS (Nova Biomedical, Waltham, MA; referred to here as BGMSB), and a hexokinase comparator (Cobas 6000 Analyzer Series; Roche Diagnostics GmbH). The blood glucose measurement accuracy was assessed by the Food and Drug Administration guidance criteria.
Results:
Two studies are presented. In the first, 2678 samples (622 arterial, 706 capillary, 1203 venous, 147 heel stick) were collected from 1577 patients in 14 US and three European sites. All accuracy criteria were met for arterial and venous samples considering all data combined. BGMSA showed better accuracy than BGMSB for arterial, venous, and heel stick, and similar results to BGMSB for capillary WB vs venous comparator. No endogenous interference from pO2, hematocrit, and sodium was identified. BGMSA was also accurate when analyzing contrived samples used to show accuracy over a wide range of glucose concentrations, alone and when combined with extreme hematocrit. In the second study, 117 capillary fingerstick samples collected at one US site were measured on both BGMSA and the comparator, and all accuracy criteria were met. No clinically significant medical risks were observed via Diabetes Technology Society Error Grids.
Conclusions:
BGMSA was effective for determining blood glucose in venous, arterial, neonatal arterial, neonatal heel stick, and capillary WB samples.
Introduction
Point-of-care (POC) blood glucose monitoring systems (BGMSs) are used to quantify whole blood glucose (BG) levels across a variety of health care settings. 1 A simple BGMS that requires a small volume of blood and generates quick and accurate results is beneficial for providers and patients. Stress-induced hyperglycemia is common in critically ill patients with and without pre-existing diabetes mellitus diagnoses 2 and is associated with longer hospital stays, increased complication rates, and higher mortality.3,4 Rapid BG assessment is important in controlling hyperglycemia and for avoiding hypoglycemia in critically ill patients and in neonates who may be at risk of neurologic impairment should hypoglycemia persist.5,6 Thus, frequent BG level monitoring is mandatory for many hospitalized patients. 7
Health care professionals rely on timely and accurate BG test results to make critical care decisions.8-10 BG fluctuations are associated with increased morbidity and mortality, especially in acutely ill patients and those with multiple medical comorbidities, as they often have extreme variations in blood chemistry and complex drug regimens that can affect BG measurement.11-14 Critically ill patients and neonates often exhibit abnormally low/high hematocrit values; 15 therefore, they require a more robust professional BGMS that is resilient to interference from endogenous and exogenous substances (compared with self-monitoring outpatients).
The US Food and Drug Administration (FDA) has separate criteria for evaluating performance and characteristics of BGMSs intended for home vs professional POC use. In 2020, they issued updated guidance for professional BGMS performance, outlining strict clinical evaluation accuracy requirements. 16
The Cobas® Pulse BGMS (Roche Diagnostics GmbH, Mannheim, Germany; referred to here as BGMSA) is a new POC BG testing system for use in professional health care environments, designed to report plasma-equivalent glucose concentration per the International Federation of Clinical Chemistry and Laboratory Medicine. 17 It incorporates an enzyme system (flavin adenine dinucleotide-dependent glucose dehydrogenase, which catalyzes oxidation of β-D-glucose in the blood sample) that minimizes the potential for cross-reacting substances, a glucose prediction algorithm that calculates glucose concentration in a sample based on a weighted combination of voltammetric and impedimetric current responses, and processes that compensate for the effects of potential interfering substances. 17 The goal of this international multicenter study was to evaluate its clinical performance using venous, arterial, capillary, and neonatal heel stick samples collected from critical care and non-critical care settings by intended POC operators.
Methods
Ethics
The protocol was reviewed and approved by Institutional Review Boards or Ethics Committees at each site. All participants/their legally authorized representatives provided written informed consent prior to undergoing study procedures.
Patients and Clinical Settings
The first study included patients from diverse non-critical and critical care health care settings at 14 US sites, two Netherlands sites, and one Germany site. Fifteen contributed critical care samples and 12 contributed non-critical care samples. Samples were collected throughout a one-year period. Samples were drawn separately from clinical care, although often using a clinical care collection (eg, taking advantage of a clinical care heel stick, to avoid another puncture of the baby). Patients with and without diabetes aged ≥22 years for adults, 29 days to 21 years (up to but not including the 22nd birthday) for pediatrics, and from birth to 28 days for neonates were included. “Critical” denotes patients receiving intensive medical intervention/those in emergency departments deemed to be critically ill by the attending physician (neonatal, pediatric, medical, and surgical intensive care units, adult and pediatric general wards, newborn nurseries, emergency departments, operating rooms, outpatient clinics). The second study included non-critical patients with and without diabetes aged ≥22 years at one collection site, similar to a physician’s office setting. There were no subject exclusion criteria.
Products, Reagents, and Instruments
All sites received Cobas Pulse instruments, software v01.01.00, charging station, and reagents, including novel BG test strips (three lots) and quality control kits (one lot).
All sites also used Nova StatStrip® Glucose Meter (Nova Biomedical, Waltham, MA; referred to here as BGMSB) to measure BG from the same samples. It uses a glucose oxidase-based detection method, autocorrects for interference, and is the first and only device currently FDA-cleared for glucose measurement in venous, arterial, capillary, neonatal arterial, and neonatal heel stick whole blood (WB) samples in hospital and professional settings, and in critically ill hospitalized patients. 1 This was chosen as a predicate due to sample types and locations for use, which closely match those intended for BGMSA. i-STAT 1 blood analyzers (Abbott, Abbott Park, IL) were used to measure sodium, hematocrit, and pO2 in blood samples unless routine testing values were available from medical records.
Method Comparison
In the first study, arterial, venous, or heel stick WB samples were used to dose the systems, and to measure sodium, hematocrit, and pO2 on the i-STAT 1 blood analyzer (when necessary). Remaining blood (collected using Lithium heparin container tubes/syringes) was centrifuged, and the resulting plasma separated and frozen for transport. Frozen plasma specimens for analysis by the hexokinase (HK) comparator (GLUC3) were stored at ≤−15°C or lower until shipped on dry ice in a thermally insulated container for testing on the Roche Cobas 6000 Analyzer Series (Roche Diagnostics GmbH). Capillary samples were used to dose the systems directly from the fingertip. Based on FDA requirements during presubmission feedback, the result was compared with plasma from a separate venous sample obtained concomitantly, which was split, and measured by all three methods. In the second study, the same capillary sample was used to dose BGMSA and BGMSB and to prepare reference plasma.
For the first study, WB samples were tested using BGMSA by site-provided clinical staff who self-trained using materials (package insert, quick reference guide, user manual) provided, without any input from the study team. BGMSA users were self-trained in order for the study to meet the requirements for the CLIA waiver. Several sites used BGMSB routinely, and for those, training was waived. For others, in-person training was offered. For the second study, trained operators (operators that were instructed on the use of the meter specifically for the study) performed the measurements.
Results were captured electronically/by verified manual transfer if electronic data transfer was impossible. All measurements with BGMSB and the comparator were performed by trained users (routine users/users trained specifically for the studies).
Contrived Sample Studies
To supplement the multicenter evaluation, two bench studies were conducted to compare the performance of BGMSA with the HK comparator using samples with extreme glucose concentrations and wide hematocrit variations.
In the first, venous blood samples were altered to achieve <80 mg/dL and >300 mg/dL glucose. The BG concentrations were adjusted by spiking with a 20,000 mg/dL aqueous glucose solution. The volume added did not exceed 3% of the total sample volume. Hematocrit levels were not altered. Venous blood from healthy donors was contrived to produce eight samples with different target glucose concentrations (15, 30, 50, 65, 350, 400, 475, 550 mg/dL).
In the second, venous blood samples were altered to achieve glucose concentrations of 10 to 50 mg/dL and adjusted to hematocrit values of 40% to 65% to simulate the observed neonate range. Healthy donor venous blood was contrived to produce eight samples with different hematocrit combinations (40% and 65%) and glucose concentrations (15, 25, 35, 50 mg/dL).
In both studies, each sample was used to dose BGMSA, then to prepare aliquots that were centrifuged within 5 minutes of dosing and subsequently tested with the HK comparator. Sample collection, contriving, and testing were performed at the Roche Diabetes Care Quality Laboratory following FDA Guidance. 16
Acceptance Criteria
Acceptance criteria from FDA Guidance were used to assess accuracy in the comparison of methods and contrived sample studies: 16 ≥95% of values must fall within ±12% of the comparator for samples with glucose concentrations ≥75 mg/dL and within ±12 mg/dL for samples with concentrations <75 mg/dL. ≥98% should be within ±15% of the comparator for samples with concentrations ≥75 mg/dL and within ±15 mg/dL for samples with concentrations <75 mg/dL.
Regression analyses were performed separately for each test-strip lot using ≥120 data pairs. Acceptance criteria were: |intercept| ≤10.0 mg/dL, slope (Passing-Bablok) 1.00 ± 0.1, correlation (Pearson) ≥0.9, and bias vs reference at three medical decision points (MDPs) (MDP1 60 [±10] mg/dL ≤12 mg/dL, MDP2 120 [±10] mg/dL ≤12.0%, and MDP3 250 [±25] mg/dL ≤12.0%).
Statistical Methods
The method comparison consisted of quantitative analyses (Passing-Bablok regression analysis) 18 and an assessment of accuracy. Analyses were performed separately by sample type. The Passing-Bablok regression analyses were performed using the bootstrap method for calculating confidence intervals for the slope and intercept (R Core Team [Package mcr Version 1.2.1, R Version 4.0.4], The R Foundation for Statistical Computing, Vienna, Austria). In the accuracy analysis, absolute and relative differences between BGMSA or the BGMSB and the HK comparator were calculated and compared with acceptance criteria.
Clinical significance of inaccuracies in BG measurements was assessed by plotting pairs of results obtained with BGMSA, or BGMSA and the HK comparator on a Diabetes Technology Society Error Grid. 19
Data and Resource Availability
This study was conducted in accordance with applicable regulations. There may be ethical, legal, or other restrictions on sharing the de-identified data set used for our analysis. Please send any data requests to
Results
Patients and Samples
The 2678 evaluable samples in the first study (622 arterial, 706 capillary, 1203 venous, 147 heel stick) were collected in diverse settings (Supplemental Table S1) by 127 POC test operators with diverse clinical training and experience (“intended users”: personnel that routinely perform routine POC testing at-site without additional study-specific training). Subjects had been administered 1031 unique medications (Preferred Names level in WHODrug) within 24 hours of blood draw (86 classes per ATC02 level in WHODrug). A total of 2293 unique diagnoses were observed (27 System Organ Classes). For the second study, 117 evaluable subjects had capillary blood collected in an outpatient setting and tested by a trained operator. Patient characteristics are presented in Table 1.
Baseline Characteristics of Patients Who Contributed Samples.

Values are n or n (%).
Abbreviations: EU, European Union; FDA, US Food and Drug Administration; N/A, not applicable.
Total unique patients (some patients contributed both arterial and venous samples).
Per FDA guidance, the cut-off from pediatric to adult patients is 22 years, which differs from EU guidance (18 years).
Two hundred and three sample results were excluded from the analysis due to different protocol deviations, including consenting issues, contamination due to improper priming of venous lines, and other pre-analytical handling issues that would compromise the integrity of the study results.
Hematocrit, pO2, and Sodium Concentration
Values varied widely for each sample type (Supplemental Tables S2-S4). No trends were identified with regard to observed bias in glucose results (difference between BGMSA and HK-based comparator) and any concentration of the three tested parameters. No endogenous interference was observed.
Accuracy
When compared with BGMSB, we showed that a consistently higher proportion of BG results from BGMSA were within ±12 mg/dL/12% or ±15 mg/dL/15% (breakpoint 75 mg/dL) of the values from the HK comparator for arterial vs arterial, venous vs venous, capillary vs capillary, and heel stick vs heel stick comparisons (Table 2). Results for BGMSA and BGMSB were similar for capillary vs venous samples.
Accuracy of BGMSA and BGMSB vs the Plasma-Based Hexokinase Comparator Method by Sample Type, Age Group, and Critical or Non-Critical Status a (Breakpoint 75 mg/dL).

Values are n/N (%), number of samples meeting criteria/total number of samples.
Abbreviations: BGMSA/B, blood glucose monitoring system A/B; EU, European Union; FDA, US Food and Drug Administration; HK, hexokinase.
Per FDA guidance, the cut-off from pediatric to adult patients is 22 years, which differs from EU guidance (18 years).
“Critical” denotes patients receiving intensive medical intervention or those in emergency departments deemed to be critical by the attending physician.
Whole blood samples were analyzed on BGMSA and BGMSB, whereas plasma derived from the same whole blood samples was analyzed by the HK comparator method.
All arterial samples were obtained from patients considered to be critical.
For arterial samples, most of the results from BGMSA and BGMSB were within ±12 mg/dL or ±12% of the HK comparator value, and within ±15 mg/dL or ±15% of the comparator (Table 2). The same trend (most of the results from BGMSA and BGMSB were within ±12 mg/dL or ±12% of the HK comparator value, and within ±15 mg/dL or ±15% of the comparator) was apparent for venous and heel stick samples.
The accuracy of BGMSA and BGMSB was lowest for capillary vs venous samples, as expected from a comparison of glucose concentrations between two differing sample matrix types. When the same sample type was tested on both systems in the second study, all results were within ±12 mg/dL or ±12% of the comparator value.
The accuracy of BGMSA was consistently higher than BGMSB across arterial and heel stick samples collected from neonates and across arterial, venous, and capillary (compared with capillary) from pediatric and adult patients (Table 2). The greatest difference was observed for neonatal arterial samples.
The accuracy of BGMSA was also consistently higher across arterial, venous, and heel stick samples from critical patients (Table 2).
Bias Plots
Bias plots demonstrated low systematic error with BGMSA for arterial, venous, and heel stick samples (Supplemental Figure S1). For BGMSA, the vast majority of results fell close to the horizontal line within the allowable total difference (ATD) zone (±12 mg/dL or ±12% of the comparator value). In comparison, bias plots for BGMSB showed a tendency to under-recover, with a higher proportion of results falling below the horizontal line, particularly those outside the ATD zone. In capillary vs venous analyses, there was larger scatter, but BGMSA was comparable to BGMSB.
Analysis of Separate Test-Strip Lots
Regression analyses showed good correlation between BG measurements for BGMSA and the HK comparator for all three test-strip lots when results were evaluated separately (Supplemental Table S5) or combined (Supplemental Table S6).
Assessment of Risk: Diabetes Technology Society Error Grids
Risk assessment was performed using Diabetes Technology Society Error Grids (Figure 1), including pairs of measurements from 2678 samples. For BGMSA results relative to HK comparator, 99.0% of arterial, 99.3% of venous, 99.3% of heel stick, and 91.8% of capillary vs venous BG measurements were within risk zone A of the Error Grid (no risk). No arterial, venous, or heel stick results were in zones C (moderate risk), D (high risk), or E (extreme risk), whereas for capillary vs venous, 0.6% of results were in zone C and none were in zone D or E. For the capillary vs capillary comparison, 99.7% of measurements were within zone A and 0.3% were within zone B. BGMSB grids are shown in Supplemental Figure S2. Both instruments showed similar risk profiles.
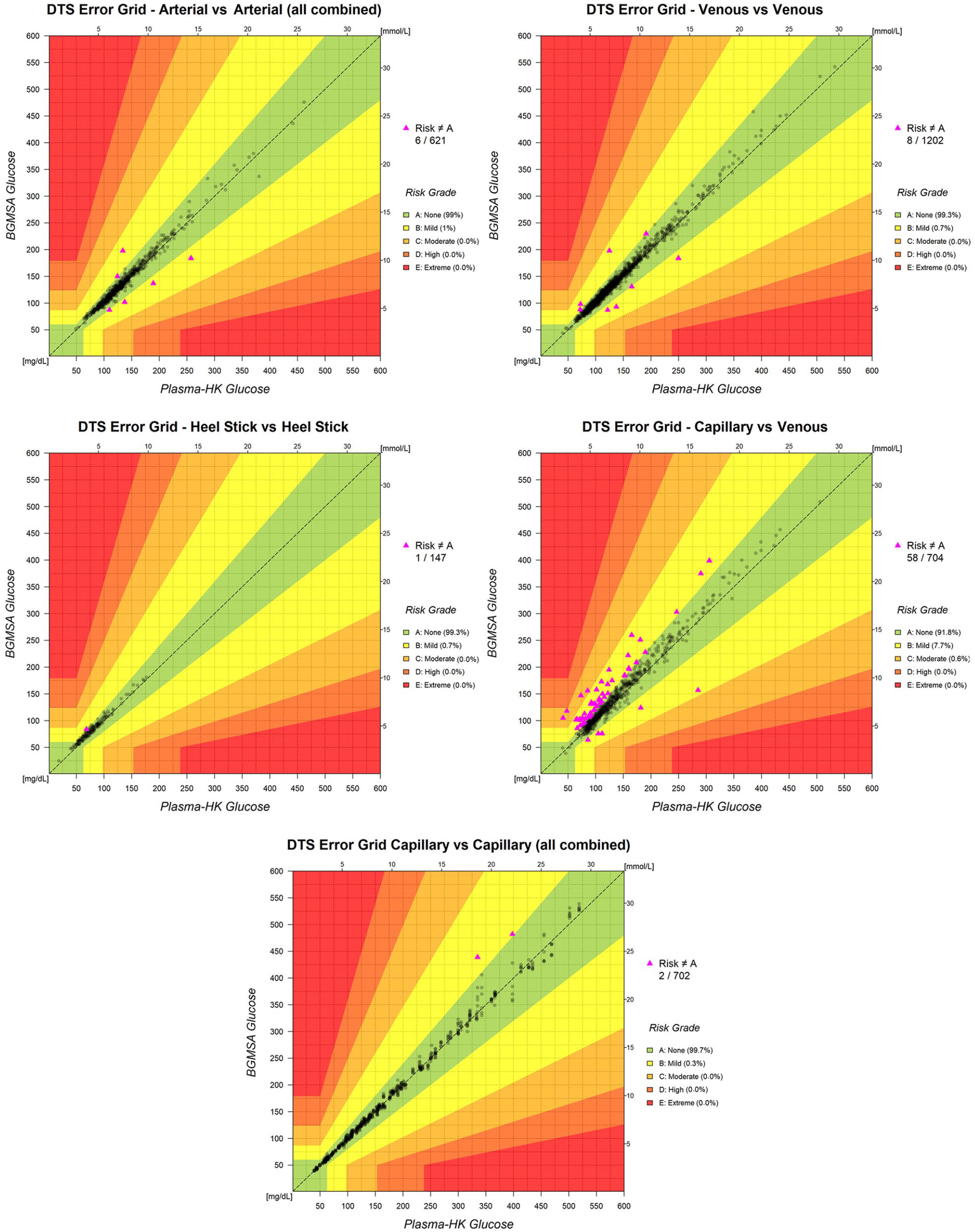
DTS Error Grid for the BGMSA vs plasma-based hexokinase comparator test results for arterial, venous, heel stick, and capillary blood samples, and for capillary to capillary. DTS Error Grid risk class: (A) None; (B) Mild; (C) Moderate; (D) High; (E) Extreme.
Contrived Sample Studies
Venous blood was obtained from 13 donors and used in the two contrived sample studies. In the first, samples were altered to achieve concentrations <80 mg/dL and >300 mg/dL. All contrived samples with concentrations <75 mg/dL had biases <12 mg/dL and all samples with concentrations ≥75 mg/dL had biases <12% (Table 3). The bias between BGMSA and the HK comparator method is shown in Supplemental Figure S3. In the second study, venous blood samples were altered to achieve glucose concentrations 10 to 50 mg/dL and adjusted to hematocrit values of 40% to 65% to simulate the range observed in neonates. All samples tested had biases <12 mg/dL (Table 3), and no significant trends were observed across the range of concentrations tested. The mean bias between BGMSA and the HK comparator was −0.56 mg/dL for high hematocrit samples and 0.25 mg/dL for low samples (Supplemental Figure S4).
Samples Contrived to Simulate Wide Variation in Glucose Concentrations and to Simulate Neonate Samples With Both Wide Variation in Glucose Concentrations and Hematocrit Values (BGMSA vs Plasma-Based Hexokinase Comparator Method).
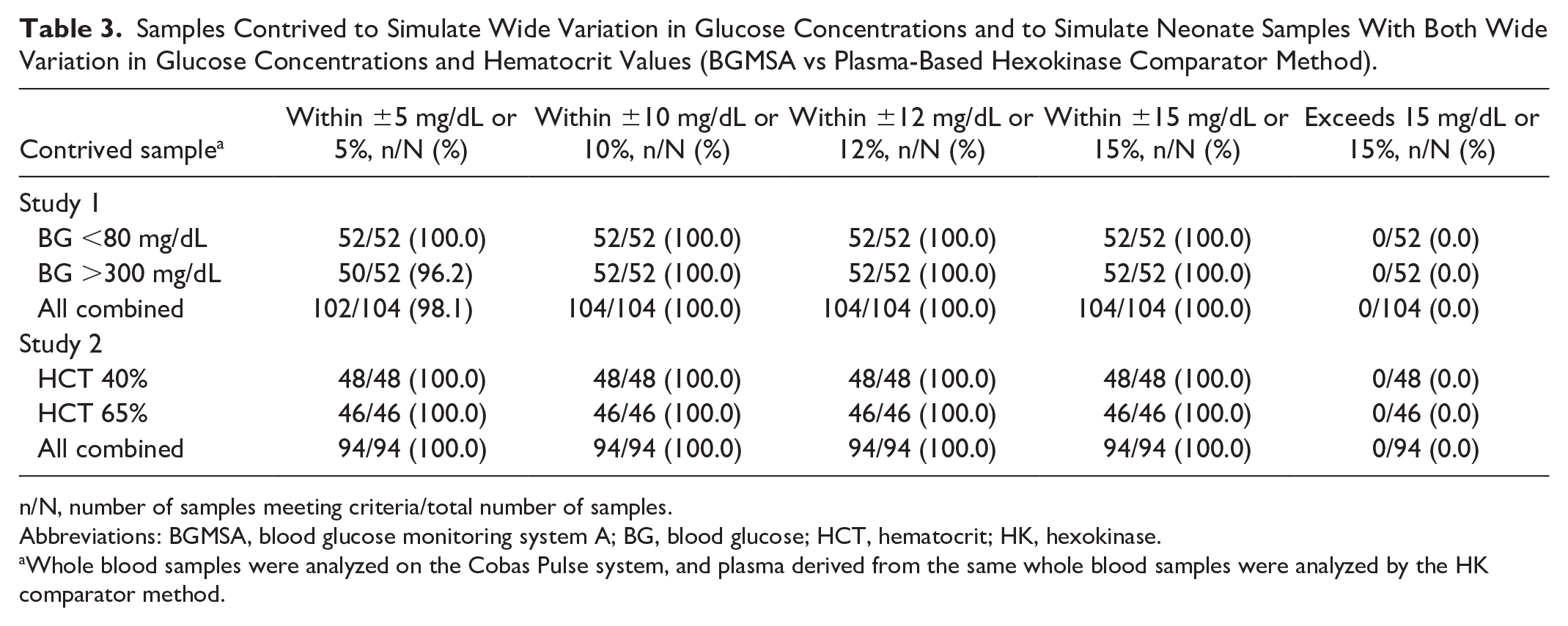
n/N, number of samples meeting criteria/total number of samples.
Abbreviations: BGMSA, blood glucose monitoring system A; BG, blood glucose; HCT, hematocrit; HK, hexokinase.
Whole blood samples were analyzed on the Cobas Pulse system, and plasma derived from the same whole blood samples were analyzed by the HK comparator method.
Discussion
We demonstrated the accuracy of BGMSA for BG measurement in WB samples. Strengths include the range of sample types and the diverse population of adult, pediatric, and neonatal outpatients and inpatients, including a substantial proportion of critically ill patients treated in intensive care or emergency department settings. Simultaneous analysis of each sample was performed using three methods. A wide range of self-trained POC operators participated in the first study to represent a worst-case, real-world environment in which expert operators may not be available to train others. The study is additionally strengthened by performing comparator testing in one central lab to ensure consistent measurements.
It should be noted that both instruments were compared to the same reference in this study. BGMSB was cleared in successive submissions, using different instruments as reference (including YSI 2300 STAT Plus [YSI Incorporated, Yellow Springs, Ohio] and Cobas Modular P 800 [Roche Diagnostics GmbH]).
Accuracy analysis shows that consistent results were obtained with BGMSA. For arterial vs arterial and venous vs venous samples, the performance of BGMSA exceeded that of BGMSB when testing the same samples.
For neonatal heel stick samples, <98% (96.6%) were within ±15 mg/dL or 15% bias of the comparator; several factors may have contributed to this wider variation (requirement for a larger volume of blood to allow for HK comparator testing; excessive squeezing of the heel, which may result in contamination of the sample with interstitial fluid; slow blood flow that can result in clotting during collection and cause sample heterogeneity20,21). However, results obtained with BGMSA were in closer agreement with the comparator than those obtained with the same samples using BGMSB.
As expected, accuracy was lowest for the capillary vs venous comparison for BGMSA and BGMSB; however, accuracy of the system and device were similar. For arterial vs arterial, venous vs venous, and heel stick vs heel stick WB comparisons, an aliquot of the same sample was tested by each method, whereas for the comparison of capillary vs venous samples, capillary samples were used to dose the systems directly from the fingertip. The results were compared with plasma from a separate venous sample obtained concomitantly that was split and measured by all three methods. The use of different sample types explains, in part, the greater divergence of results from the comparator. Capillary blood generally has a higher glucose concentration than venous, and the magnitude of the difference varies with the patient’s prandial status,22,23 with higher capillary values in the fed vs fasting state.24,25 Venous samples are also potentially subject to contamination with intravenous fluids when collected via central line.26-28 Unsurprisingly, when comparing capillary to capillary directly, BGMSA met the accuracy criteria.
The BGMSA was consistent: there was little variability in BG measurements within and between test-strip lots, with consistently low bias. Bias plots showed that the system had low error across arterial, venous, heel stick, and capillary samples (when compared directly with capillary). In comparison, bias plots for BGMSB showed a tendency to under-recover, with a higher proportion of results falling below the horizontal line, particularly those that fell outside the ATD zone.
The comprehensive assessment of clinical risk showed that there are few discrepancies between results obtained with BGMSA and the HK comparator, and that most of these discrepancies pose either no risk or only a mild risk to patients. The DTS Error Grid was developed to provide a “modern, easy-to-use” method for “rigorous evaluation of point accuracy in glucose monitoring devices” for developers of such devices, as well as for clinical researchers and regulatory bodies. 19 The developers intend the DTS Error Grid to be used in the “development and certification of current generation BGMSs and continuous GMSs, as well as future technologies for measuring glucose.” 19
The contrived sample studies provide insight into how BGMSA performs across a wide range of BG concentrations, alone or in combination with a wide range of hematocrit values. All samples were within ±12 mg/dL or ±12% of the comparator method. These results show that the performance of the system is not affected by high hematocrit or low BG concentrations, demonstrating suitability for testing samples from neonates, which have characteristically high hematocrit values and low BG concentrations.9,29,30
This study has some limitations. The population was predominantly white and non-Hispanic, and clinical populations may be more diverse. It is unlikely that the accuracy of a BGMS varies by race, so this limitation is unlikely to be clinically significant.31-33 The study included a large number of patients overall, with high representation of critically ill patients. However, within certain subgroups the number of samples was comparatively low, for example, those from pediatric patients in general, and the number of samples from non-critically ill neonates in particular. Finally, the capillary vs capillary study only included non-critically ill subjects.
Conclusions
This study demonstrates that, when used as intended by health care professionals in hospital and clinic settings, BGMSA is highly effective for BG determination in arterial, venous, and capillary WB samples from adult and pediatric populations, and in heel stick and arterial samples from neonates. When compared using the same split samples, BGMSA was generally more accurate than BGMSB, which is currently the only FDA-cleared device for BG measurement in critically ill patients.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251325873 – Supplemental material for Multicenter Evaluation of a New Strip-Based Blood Glucose System for Point-of-Care Testing in Critical and Non-Critical Care Settings
Supplemental material, sj-docx-1-dst-10.1177_19322968251325873 for Multicenter Evaluation of a New Strip-Based Blood Glucose System for Point-of-Care Testing in Critical and Non-Critical Care Settings by Michael Goodman, Sven Bercker, Nancy Breitenbeck, Cristina Canada-Vilalta, Daisy D. Canepa, William A. Clarke, Christopher W. Farnsworth, Edward O. Ganser, Nienke Geerts, Barbara Goldsmith, Gary Headden, Debra Hoppensteadt, Birgit Klapperich, Ryan Matika, Gabrielle Miles, Sagori Mukhopadhyay, James H. Nichols, Rebecca M. O’Dell, Zubaid Rafique, Andre Schuetzenmeister, Robbert Slingerland, Ann F. Stankiewicz, Jonathan R. Swanson, Nam K. Tran, Alan H. B. Wu and Brad S. Karon in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors acknowledge Michael Marquant (Scientific Lead, Electrochemical Measuring Methods, Roche Diagnostics GmbH) and the sustained collaborative effort and dedication of the entire Roche POC Hospital Glucose Development Team in the realization of this project; along with Simon Gessler (Development Lead, Roche Diagnostics GmbH) and Michelle McLean (Medical Affairs Lead, Roche Molecular Systems, Inc., Indianapolis, IN) for help in proposing content, facilitating the writing process, and providing feedback. Research support in the form of third-party medical writing assistance for this manuscript, furnished by Blair Jarvis, MSc, on behalf of Nucleus Global, an Inizio Company, was provided by Roche Diagnostics GmbH.
Abbreviations
ATD, allowable total difference; BG, blood glucose; FDA, US Food and Drug Administration; HK, hexokinase; MDP, medical decision point; POC, point-of-care; WB, whole blood.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors received third-party writing assistance from Roche; funding from Roche for study supplies. M.G. received grants or contracts from the National Institutes of Health and the Department of Defense; payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing, or educational events from Johnson & Johnson and Grifols; and participation on a Data Safety Monitoring Board or Advisory Board for the TRAIT study. S.B. received payment for expert testimony from Berlin District Court, Public Prosecutor’s Office Dresden, and Arbitration Centre of the Saxon State Medical Association; support for attending meetings and/or travel from bioMérieux (investigator meeting); participation on a Data Safety Monitoring Board or Advisory Board for DSMB for Hemotune, Switzerland; is Vice Chair of local ethics board. N.B. has no other conflicts of interest. C.C.-V. is an employee of Roche. D.D.C. is an employee of and a shareholder in Roche. W.A.C. received grants or contracts from Thermo Fisher Scientific, Radiometer, Becton Dickenson, and Werfen; consultancy fees from Thermo Fisher Scientific, Saladax Biomedical, Roche Diagnostics, Radiometer, Werfen, and Truvian. C.W.F. received grants or contracts from Abbott Laboratories, Cepheid, Roche Diagnostics, Sebia, Beckman Coutler, The Binding Site, BlueJay Diagnostics, and bioMérieux; consultancy fees from Abbott, Roche, Werfen, and Cytovale; payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing, or educational events from Werfen, BD, Abbott, and Roche. E.O.G. is an employee of Roche Diabetes Care, Inc. N.G. received provision of study materials and payment for labor (inclusion, sampling handling, etc.) to the institution from Roche for investigations. B.G. has no other conflicts of interest. G.H. has no other conflicts of interest. D.H. received grants or contracts from Sysmex America, Inc., Glycan Therapeutics, and SBIR/NIH. B.K. is an employee of Roche. R.M. has no other conflicts of interest. G.M. is an employee of Roche. S.M. received funding from Roche for study enrollment and procedures. J.H.N. received grants or contracts to institution for support of study from Roche Diagnostics; research support from Werfen, Abbott Laboratories, and Siemens; consultancy fees from Werfen and Abbott Laboratories for Scientific Advisory Boards; payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing, or educational events from Roche, Werfen, Abbott Laboratories, BD, Thermo Fisher Scientific, Physician’s World/Alexion, and Siemens for speaker honoraria; support for attending meetings and/or travel from the Association of Diagnostic Lab Medicine (ADLM), Clinical and Laboratory Standards Institute (CLSI), and College of American Pathologists (CAP); leadership or fiduciary role in other board, society, committee, or advocacy group for CAP, CLSI, Journal of Applied Laboratory Medicine, and ADLM; is President of CLSI; and Deputy Editor of the Journal of Applied Laboratory Medicine. R.M.O. is an employee of Roche. Z.R. is an Advisory Board Member—renal (personal fees) from Vifor Pharma, Inc. and an Advisory Board Member—cardio renal (personal fees) from AstraZeneca, Inc. A.S. is an employee of Roche Diagnostics GmbH. R.S. received lab funding from Roche for investigations and serves on a Roche Advisory Board. A.F.S. is part of the Roche employee program. J.R.S. received speaker honoraria from Prolacta Bioscience, Inc. N.K.T. serves on the Roche Advisory Board; in a Roche consulting role; and their institution, UC Davis, is a Roche Diagnostic Center of Excellence. A.H.B.W. received funding from Roche; Scientific Advisory Boards for Werfen, Babson, Truvian, and RCE. B.S.K. serves on a Roche Advisory Board.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Roche Diagnostics. The Cobas Pulse system is approved for clinical use in Europe (CE Mark-accepting countries). The system is not yet available in the United States, but it is FDA-cleared for 510K and CLIA waived for use with arterial and heel stick whole blood samples. All product names and trademarks are the property of their respective owners.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
