Abstract
Managing type 1 diabetes in infants and very young children poses unique challenges due to their low insulin requirements, high insulin sensitivity, and rapidly changing metabolic needs. Standard insulin formulations (U100) may prove inadequate for this age group, especially when utilizing continuous subcutaneous insulin infusion or automated insulin delivery (AID) systems.
This article presents our clinical experience with diluted insulin (U10) in very young children using the AID system CamAPS FX, along with a literature review, highlighting its potential benefits, such as reduced incidence of hypoglycemia and rates of technical malfunctions. We also discuss key practical considerations for implementing insulin dilution in clinical practice, including the establishment of safety protocols, caregivers and healthcare professionals training, and the importance of accurate preparation and labeling of diluted formulations to mitigate potential serious dosing errors.
Introduction
The standard insulin concentration in current practice is 100 international units (IU)/mL (U100). 1 The move to standardized insulin concentration worldwide was a major accomplishment, allowing the use of uniform, volumetric insulin syringes and reducing dosing errors. However, this one-size-fits-all approach to insulin concentration may not be easily applicable in the management of diabetes in infants and toddlers who have low insulin requirements.2,3 The use of diluted insulin via continuous subcutaneous insulin infusion (CSII) is recognized as an option to overcome the challenges associated with the delivery of minimum insulin dosages, allowing for more precise dosing. 4 At present, there are no definitive guidelines on the use of diluted insulin, although dilution is a strategy discussed for the management of young children in the ISPAD guidelines.1,2
Insulin can be diluted with a diluting medium obtained from the manufacturer, or if this is not available, normal saline may be an alternative. 1 When using the specific diluting medium, diluted insulin maintains stability in terms of purity, potency, antimicrobial safety, and pharmacokinetics5,6 (Table 1).
Summary of Studies Reporting Use of Diluted Insulin in Children.

Search strategy for the articles: PubMed and Google Scholar online databases were comprehensively searched for peer-reviewed articles. The search was limited to English-language publications, with no restrictions on publication date or geographic location. Relevant articles were identified using combinations of keywords, including “diluted insulin,” “insulin dilution,” “10 U/ml insulin,” “insulin concentration,” “type 1 diabetes,” “diabetes in infancy.”
Abbreviations: T1D, type 1 diabetes; AID, automated insulin delivery; TIR, time in range; TBR, time below range; TDD, total daily dose; CSII, continuous subcutaneous insulin infusion; DKA, diabetic ketoacidosis; GV, glucose variability.
Several case reports, including our own, have highlighted that using diluted insulin in CSII is associated with lower hypoglycemia incidence, reduced absorption variability and better HbA1c compared to U100 insulin.7-13 (Table 1). In addition, clinical studies comparing diluted insulin (U20) with standard insulin in children younger than 7 years, using an automated insulin delivery (AID) system, showed reduced inter-individual variability in time to peak insulin action and reduced nocturnal hypoglycemia with diluted insulin,14-16 thus supporting diluted insulin use in children with a very low total daily dose (TDD). The main randomized trial comparing AID with diluted (U20) versus standard-strength insulin over three weeks in 24 children aged 1-7 years showed no significant differences in glycemic outcomes between the two groups. 17 However, in that trial average TDD was around 18 IU, and few children were younger than 3 years, which limits the generalizability of the findings to younger children or situations where TDD is less than 10 IU.
Despite these potential benefits, dosing errors with diluted insulin can occur and may have serious consequences. The greatest risk is insulin overdose, which can be life-threatening. Therefore, utmost care must be taken when diluting insulin, and proper training should be provided to ensure its safe implementation.
In this article, we share our practical experience of using U10 diluted insulin (10 IU/mL) in the AID system CamAPS FX in very young children. We also discuss key points related to its efficacy, safety, and the education required for both healthcare professionals (HCPs) and parents/caregivers.
Why to Consider Insulin Dilution in Very Young Children?
Low Insulin Requirements
The management of type 1 diabetes (T1D) in infants and young children presents unique challenges due to their high insulin sensitivity, rapidly changing insulin needs, low TDD, variable eating and exercise patterns, and limited ability to communicate symptoms of hypoglycemia.2,3 Insulin requirements often decline during the honeymoon period, which adds further complexity to diabetes management.2,18 CSII and AID systems are the recommended approaches to achieving satisfactory glycemic targets in this age group.2,19
Limitation With Current CSII and AID Systems
As highlighted in the ISPAD guidelines,1,2 the low TDD required in children with a body weight below 10 kg demands special consideration when using commercially available insulin pumps and insulin preparations. The guidelines suggest considering the dilution of U100 insulin or using an intermittent basal rate of 0 IU/hour for limited periods (e.g., every other hour).1,2
A few case reports highlight the issues associated with insulin delivery suspensions and high glucose variability when using AID systems in children with a very low TDD.7,15 These issues include frequent cannula occlusions, air bubbles in the tubing, and infusion sets lasting barely 2 days. Such complications can lead to pump malfunctions, causing stress for parents/caregivers and affecting the achievement of desirable glycemic outcomes. Notably, switching to diluted insulin has been associated with a reduced incidence of pump malfunctions, and longer duration of infusion circuits.7,15
CamAPS FX delivers a patency bolus of 0.04 IU (displayed as 0.2 IU on Glooko) approximately every 90 min, when basal insulin delivery is automatically suspended by the system to prevent the tubing from becoming blocked. 20 In our experience, a patency bolus can cause hypoglycemia in a child with a very low TDD using standard-strength insulin.
Licensing
Despite the commercial availability of AID systems, some young children may not meet their licensing criteria, due to age, weight or TDD. 21 This means some children could be using an AID system off-license regardless of standard or diluted insulin.(Table 2)
Minimum Age, Weight and Total Daily Dose Of Commercially Available Automated Insulin Delivery Systems.
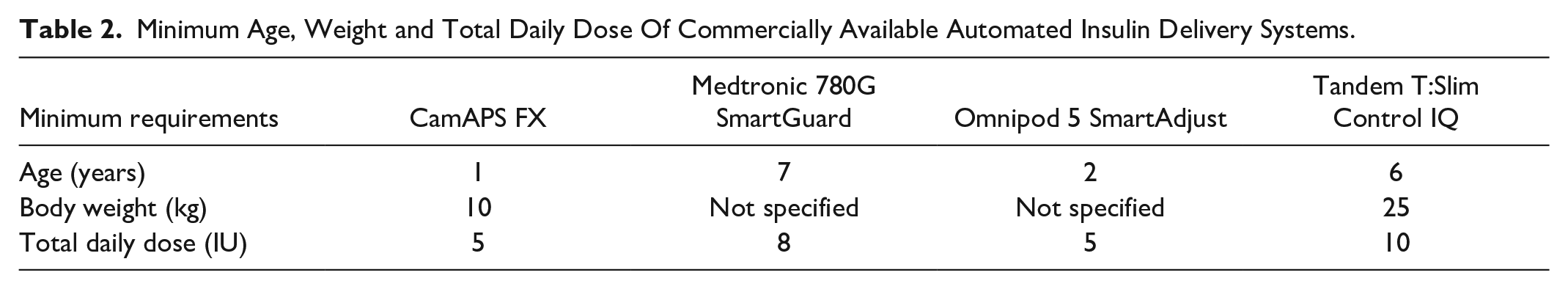
In our center, we have been using diluted insulin primarily with the CamAPS FX system. However, other groups have reported diluting with other AID systems.10,13
How to Safely Implement Diluting Insulin?
Setting
In our tertiary pediatric diabetes center, we typically start diluted insulin with an AID system when children are admitted as inpatients following T1D diagnosis. However, this approach is also feasible in the outpatient setting. The pros and cons of these approaches are reviewed in Table 3.
Pros and Cons of Starting Insulin Dilution in Different Settings.

Safety Considerations
Labels on pumps
It is essential to highlight that diluted insulin is not a standard-strength formulation. Diluted insulin is filled into regular reservoirs; therefore it appears to be “standard” insulin. To prevent any confusion and potential dosing errors, we label the pump and specify “U10 DILUTED INSULIN (10 IU/mL)” prominently. This ensures that if the child is hospitalized or interacts with HCPs, the pump will be immediately recognized as containing diluted insulin.
Flag on hospital records
We also add alerts to the children’s hospital records. This is to inform any HCP who encounters the child that they are using diluted insulin.
Care plan
As part of our education package, we provide families with a care plan which clearly displays “U10 DILUTED INSULIN IN INSULIN PUMP” on the first page. We recommend that families bring this care plan with them whenever seeking medical advice, ensuring that the HCP is aware of the non-standard concentration of insulin in the pump.
Out of hours (OOH) support
Our diabetes team participates in an OOH pediatric diabetes consortium, a regional service where diabetes teams rotate coverage. Since children using diluted insulin may not follow standard diabetes management protocols, we instruct these families to contact our pediatric diabetes consultant on call for specialized advice in urgent situations.
Insulin pens at home
Depending on the child’s TDD and insulin sensitivity factor, they may not be able to receive a suitable insulin dose via insulin pen to correct hyperglycemia or ketones. For example, 0.5 IU of U100 rapid-acting insulin may equate to a large percentage of their TDD. Despite this, we would always provide a stock of rapid-acting and long-acting insulin pens for families to have at home for administration if required, such as in the event of a pump failure. In this situation, families are instructed to contact the diabetes team for advice on insulin doses. If an insulin pen correction is not safe to be given, families are advised to attend the nearby hospital for further management of insulin therapy.
Practical Considerations
Guideline
A “Non-standard insulin concentrations: Usage in clinical practice” guideline was written by our pediatric diabetes educators and consultants with input and approval from pharmacy and the hospital Trust document’s body. The guideline includes: (1) the rationale for diluting insulin, (2) expectations for those involved in the care of the child (e.g., inpatient ward nurses), (3) instructions for preparing the correct diluted insulin solution, (4) guidance on interpreting insulin pump settings using U10 preparations, (5) safety considerations, and (6) practical considerations (Appendix 1, Table S1).
Pump Setup
In order to program the pump so it can deliver U10 insulin, the settings need to be tenfold greater than the desired amount so this can achieve U10 delivery (Appendix 1, Table S2).
Practical Equipment
A child may need to start on an insulin pump before a new pump can be delivered. Therefore, having a spare pump readily available is essential. Maintaining a supply of infusion sets (the preferable option in this age group is steel cannulas) and reservoirs for the pump, and glucose sensors is also required. The supplies for diluting insulin are also required: U100 insulin syringes, IV syringes (various sizes), U100 rapid-acting insulin and diluting medium.
Care Plan
To provide the required training and education of parents/caregivers, we have developed a care plan, which covers: (1) management of hypoglycemia and hyperglycemia; (2) troubleshooting potential issues with the pump or CamAPS FX app; (3) instructions for using the pump without the CamAPS FX app (manual mode); (4) guidance on inserting glucose sensors; (5) information about pump settings; (6) Personalized Sick Day Rules; and (7) how to revert to subcutaneous injections, in the event of pump failure.
Other Considerations
Some centers can organize hospital pharmacies to make up the diluted insulin preparation in advance. This is not something we could offer for our families. Therefore, we need to teach parents to dilute the insulin when required, usually every two to three days.
Family Support
Conversation With Parents/Caregivers
Current AID systems are only licensed with U100 insulin; therefore, using diluted insulin means operating the system off-license, which carries potential risks. If parents prepare the diluted insulin solution incorrectly, it could lead to hypo- or hyperglycemia. Thus it is important to have a conversation with parents about the clinical benefits of using diluted insulin over the standard concentration and the associated risks. This conversation is typically conducted using a multidisciplinary team approach, with the consultant leading the conversation and one of the diabetes educators present to explain the practical implications of diluting insulin. We discuss why, in our view, it is clinically beneficial for the child to use diluted insulin rather than standard concentration, presenting the management options in a balanced manner so that parents can make an informed choice.
Intensity of Training for Parents/Caregivers
Given the potential risks of incorrectly diluting insulin, we strongly encourage that at least two adults in the child’s life receive our training. Most frequently, this is the child’s parents. We always discuss the importance of completing the diluting process in pairs, both for the professionals delivering the training and the family members receiving the training. We also discuss the need for both parents to alternate roles: one leading the diluting process while the other shadows. This approach ensures that both individuals become equally skilled and confident in the process.
Role of Educator
Skills
It is important that the educators involved in diluting insulin already have good experience in managing very young children with T1D. Although we need to deliver a standard package of education, it is important for educators to be agile in their delivery of the information to meet the diverse learning styles and speeds of parents/caregivers. Educators must possess a solid general knowledge of CSII and AID systems.
Time
Since we typically start children on diluted insulin as an inpatient, this process can be time-intensive for the educator team. We aim to have two educators working together to deliver education and practical sessions. This collaborative approach helps reduce risk of mistakes and oversights. Recognizing that education and practical sessions can be overwhelming for parents, we usually schedule several daily sessions of 1–2 hours each over a week, to allow parents to digest the information received.
In-Hospital OOH Support
While the child is an inpatient and just started on an AID system with diluted insulin, a backup plan is required for overnight support in case issues arise that could disrupt insulin delivery. This could involve, but is not limited to, infusion set failure, technical issues with the insulin pump/AID, and pump reservoir unexpectedly empty.
Conclusions
Insulin dilution can be a beneficial strategy for managing very young children with diabetes. Guidelines support its use, particularly in cases where very small doses are required.
It offers improved accuracy, and control of blood glucose levels. However, it requires careful preparation and monitoring to prevent dose errors and ensure safety. The decision to use insulin dilution should be made on an individual basis, as part of a shared decision-making process with caregivers. The approach used may vary across pediatric diabetes centers.
The experience at our center supports its efficacy but also highlights the need for extensive education/training of staff and caregivers.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251322183 – Supplemental material for How to Safely Use Diluted Insulin in an Automated Insulin Delivery System in Very Young Children: An Educator Perspective
Supplemental material, sj-docx-1-dst-10.1177_19322968251322183 for How to Safely Use Diluted Insulin in an Automated Insulin Delivery System in Very Young Children: An Educator Perspective by Eilidh Nicol, Jennifer Ashford, Beatrice Prampolini and M. Loredana Marcovecchio in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
We would like to acknowledge past members of the diabetes team involved in diluting insulin without whom our experience and progress would not have been possible: Julia Fuchs, Sara Hartnell, Helen Hysted, Cliodhna Myles and Rachel Williams. We would also like to thank our current diabetes team for their ongoing help and support: Nazia Bhatti, Lisa Cooper, Benjamin Fisher, Stephen Forster, Emile Hendriks, Jenny Lee, Natalie Schillaci, Ajay Thankamony and Sandra Walton-Betancourth.
Abbreviations
AID, automated insulin delivery; CSII, continuous subcutaneous insulin infusion; DKA, diabetic ketoacidosis; GV, glucose variability; IU, international units; MDI, multiple daily injections; OOH, out of hours; T1D, Type 1 diabetes; TDD, total daily dose; TBR, time below range; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
