Abstract
Background:
Ramadan fasting presents unique challenges for individuals with type 2 diabetes (T2D) due to alterations in diet and medication regimens. This study evaluates the effects of Ramadan fasting on glycemia by utilizing the glycemia risk index (GRI), which integrates both hypoglycemic and hyperglycemic risks into a unified metric, alongside continuous glucose monitoring (CGM) data. In addition, the study examines the correlation between GRI and diabetes therapy-related quality of life (DTR-QOL) to understand the broader impact on patient outcomes.
Methods:
An ambispective, one-group pre-post design was employed at a tertiary diabetes treatment center, involving 111 adults with T2D. Data were collected across three periods: one month before Ramadan, during, and one month after. Clinical, metabolic, and glycemic parameters were recorded. The CGM-based calculations included GRI, with its hypoglycemia component (CHypo) and hyperglycemia component (CHyper). The DTR-QOL was measured to evaluate therapy-related quality of life (QoL).
Results:
During Ramadan, GRI significantly decreased (median = 30.5) compared to before (35.2) and after (37.4; P < .001), indicating improved glycemic stability. Both CHypo and CHyper were significantly reduced during fasting. The %TIR70-180 increased from 42% before to 66% during (P < .001), accompanied by a notable decrease in glycemic variability. The DTR-QOL scores were high across all domains, reflecting a positive therapy-related QoL (scale score: 78.3 [interquartile range = 75.4-81.3]). No significant differences were observed across GRI zones.
Conclusions:
With tailored education and CGM-based monitoring, Ramadan fasting can improve glycemia in individuals with T2D, enhancing GRI and related glycometric parameters for safer, more stable glycemic patterns.
Keywords
Introduction
Ramadan fasting, a religious practice observed by millions of Muslims globally, entails abstinence from food, drink, and other activities from dawn to sunset each day for a month.1,2 While this practice carries profound spiritual and cultural significance, it poses unique challenges for individuals managing chronic illnesses, including type 2 diabetes (T2D). Although Islamic law exempts such individuals from fasting, many choose to fast, motivated by personal beliefs and communal traditions. 3 Fasting during Ramadan disrupts typical dietary, medication, and activity patterns, potentially destabilizing glycemic control. 4 Prolonged fasting hours elevate the risk of hypoglycemia, particularly for individuals on insulin or sulfonylureas, while excessive calorie intake during Iftar, the post-sunset meal, can lead to hyperglycemia. These glycemic fluctuations, if unmanaged, increase the risk of acute complications.4 -6 These challenges are further compounded by variability in fasting durations and individual health conditions, underscoring the need for patient-specific care plans.
Despite these risks, Ramadan fasting can offer metabolic benefits when managed properly. Studies have documented improvements in body weight, lipid profiles, and blood pressure among well-controlled diabetes patients.7 -11 Reductions in fasting blood glucose and hemoglobin A1C (HbA1c) levels have been observed in individuals adhering to structured dietary and medication regimens during Ramadan. However, these benefits often depend on pre-fasting education, appropriate medication adjustments, and continuous glucose monitoring (CGM), highlighting the importance of a proactive and holistic approach to diabetes management. 12 Advanced CGM tools have transformed diabetes care by enabling real-time tracking of glucose fluctuations, offering invaluable insights into glycemic patterns during Ramadan.13,14 Traditional metrics such as time in range (%TIR70-180), HbA1c, and mean glucose levels provide valuable insights into glycemic control over time.
In contrast, the glycemia risk index (GRI) goes a step further by integrating the risks of hypoglycemia component (CHypo) and hyperglycemia component (CHyper) into a single, unified score, delivering a comprehensive assessment of overall glycemic safety.15 -17 Although these metrics collectively enhance our understanding of the impact of Ramadan fasting on glycemic health, their application in Ramadan-specific studies remains limited. By quantifying the severity and risk of deviations from optimal glucose ranges, GRI provides clinicians with deeper insights that go beyond average glucose exposure, drawing attention to dangerous fluctuations that traditional metrics often overlook.13,17,18 Incorporating GRI into diabetes management enables a more comprehensive risk assessment, supporting personalized treatment adjustments to minimize glucose extremes and reduce the likelihood of diabetes-related adverse outcomes.17,19 -21
The role of GRI and its components in Ramadan is particularly important given the unique challenges posed by fasting. The CHypo and CHyper, together, provide a balanced assessment of both extremes, which is essential for evaluating the overall safety of fasting for individuals with T2D. The current study aims to bridge critical gaps in understanding by analyzing the impact of Ramadan fasting on GRI, CGM metrics, and quality of life (QoL) among individuals with T2D who fast. By leveraging real-world data from CGM, it seeks to provide actionable insights into personalized diabetes management strategies. Ultimately, the findings will support safer fasting practices and inform health care providers on optimizing care for patients observing Ramadan.
Methods
Study Design
We conducted this ambispective, one-group pre-post study at a tertiary diabetes treatment center in Prince Sultan Military Medical City (PSMMC), Riyadh, Saudi Arabia, across three specific periods: one month prior to Ramadan, during Ramadan, and one month after Ramadan in 2024. The study protocol received approval from our Research and Ethics Committee (IRB approval no. 1394). It was conducted in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines 22 and complied with the 1964 Helsinki Declaration and its subsequent amendments. 23
Eligibility Criteria
The study recruited individuals diagnosed with T2D, aged 30 to 70 years, who were managed with non-intensive insulin regimens and demonstrated both the willingness and ability to fast during Ramadan 2024. Eligibility was determined using the newly established International Diabetes Federation Diabetes and Ramadan (IDF-DAR) risk stratification tool,24 -26 which helped identify participants with no absolute contraindications for fasting. The final determination of eligibility was made by the treating physician based on clinical judgment. Moreover, candidates were required to have utilized a flash continuous glucose monitoring (FGM) system, specifically the FreeStyle Libre 2 with real-time alerts, for a minimum of three months prior to enrollment.
Participants were excluded if they had unstable or poorly controlled diabetes and accepted the decision not to fast, as determined by the treating physician’s assessment. In addition, participants were excluded if they were expected to transition to intensive insulin therapy during Ramadan, were classified as “high risk” for fasting according to the IDF-DAR guidelines,24 -26 were scheduled for surgical procedures during the fasting period, or were unwilling to commit to the study.
Study Procedure and Data Collection
Retrospective data were collected during the month leading up to Ramadan, while prospective data were gathered throughout and following the Ramadan period.
All participants routinely attend a structured educational program as part of the standard care provided at the center, at least two weeks prior to Ramadan. This program is conducted onsite and involves individual consultations with health care providers. It is designed to educate individuals with diabetes on appropriate dietary practices, exercise routines, meal timing, and the management of antidiabetic medications during Ramadan. In addition, the program offers guidance on fasting protocols, specifically addressing circumstances under which fasting should be avoided due to hyperglycemia or hypoglycemia, in accordance with the recommendations of the DAR guidelines. Individuals classified in any risk strata of the IDF-DAR guidelines were advised to break their fast and seek immediate medical attention if they experienced any episodes of hypoglycemia or symptomatic hyperglycemia during Ramadan.24,27
Before enrolling in the study, participants were informed of their unconditional right to withdraw or break their fast at any time without the need to provide a reason or prior notice. All participants received comprehensive information regarding their roles in the study and provided signed informed consent forms prior to enrollment.
At baseline, demographic and clinical data were meticulously collected. This included information on age, sex, body mass index (BMI), medical history, duration of diabetes, and current treatment regimens. We gathered data on CGM system usage and metabolic control by downloading and analyzing information from participants’ devices using the LibreView platform.
We categorized the %TBR into specific glycemic levels. Very low glycemia was identified as %TBR<54, while low glycemia was categorized as %TBR54-69. The calculation for CHypo was performed using the formula: CHypo = [%TBR<54 + (0.8 × %TBR54-69)]. Conversely, CHyper was determined by categorizing %TAR, with high glycemia classified as %TAR181-250 and very high glycemia as TAR>250. The formula for CHyper was as follows: CHyper = [%TAR>250 + (0.5 × %TAR181-250)]. The GRI was calculated by integrating the CHypo and CHyper components using the following formula: GRI = [(3.0 × CHypo) + (1.6 × CHyper)].17,21 Recent findings have established that a 14-day period of CGM data is optimal for calculating GRI. 28
In addition to GRI, we analyzed several key glycometric parameters to further characterize participants’ metabolic control. These parameters included mean glucose levels (mg/dL), the Glucose Management Indicator (GMI%), the percentage of time in range (%TIR70-180), which represents glucose levels between 70 and 180 mg/dL, the percentage of time above range (%TAR>180) for glucose levels exceeding 180 mg/dL, and the percentage of time below range (%TBR<70) for glucose levels below 70 mg/dL. Glycemic variability (GV) was assessed using the coefficient of variation (CV%). Data on fasting breaks and hospital visits due to glycemic issues were collected through a review of participants’ electronic medical records and confirmed via patient interviews conducted during the post-Ramadan follow-up, specifically when administering the diabetes therapy-related quality of life (DTR-QOL) Questionnaire.
Diabetes Therapy-Related Quality of Life Questionnaire
The DTR-QOL questionnaire was employed after Ramadan to assess the influence of the prescribed therapy regimen on patients’ QoL. This validated instrument consists of 29 items distributed across four domains: Burden on Social and Daily Activities (BSDA), which comprises 13 items; Anxiety and Dissatisfaction with Treatment (ADT), containing eight items; Hypoglycemia (HG), which includes four items; and Satisfaction with Treatment (ST), featuring four items. The Arabic version of the DTR-QOL has been validated for use among patients with diabetes in Saudi Arabia, demonstrating strong relevance and clarity of items, as indicated by a content validity index of 0.938. 29 The response scale is a seven-point Likert scale (1: strongly agree; 7: strongly disagree). Each item’s score is reversed, such that a score of 7 represents the highest QoL, while a score of 1 indicates the lowest QoL. The total score is calculated as the mean score of all items and converted to a scale from 0 to 100, where the best response equals 100, and the worst response equals 0.29,30
Statistical Analysis
The analysis was performed using SPSS version 28.0 (SPSS Inc, Chicago, Illinois) and RStudio version R 4.3.0. The normality of the variables was assessed using the Kolmogorov-Smirnov test. Quantitative data that followed a normal distribution were presented as means with standard deviations (SD), while non-normally distributed data were reported as medians and interquartile ranges (IQR, 25th to 75th percentile). Categorical variables were expressed as frequencies and percentages. A repeated measures analysis of variance (ANOVA) using the Friedman test was conducted to assess glycometric parameter changes before, during, and after Ramadan. Pairwise comparisons were performed to investigate specific differences identified by the Friedman test. In addition, Spearman’s correlation coefficient (r) was calculated to determine correlations between quantitative variables, with correlation strength ranging from 0 to ±1. Statistical significance was established at P-values less than .05.
Results
Baseline Clinical and Metabolic Characteristics
A total of 149 patients were screened for the study (Supplemental Figure S1). The final cohort consisted of 111 participants, with a balanced distribution of gender: 55 females (49.5%) and 56 males (50.5%). The mean age of participants was 48.1 years (SD = 7.31). The mean BMI was 29.7 kg/m2 (SD 3.58 kg/m2), with a mean HbA1c of 8.22% (SD = 0.39%). Regarding comorbidities, dyslipidemia was the most prevalent condition, observed in 51 participants (45.9%), followed by hypertension in 38 participants (34.2%). Biguanides were the most commonly prescribed medication class, used by 100 participants (90.1%), while glucagon-like peptide 1 (GLP-1) receptor agonists were utilized by 58 participants (52.3%). The mean duration of diabetes among participants was 7.03 years (SD = 4.6). Table 1 details the baseline clinical and metabolic characteristics of the study participants.
Baseline Clinical and Metabolic Characteristics of Study Participants.

Comparative Analysis
Glycemia risk index and its components—hypoglycemia component and hyperglycemia component
The comparative analysis of metabolic and glycemic parameters across the three periods of Ramadan revealed significant changes, as summarized in Table 2 and Figure 1. The Chypo was recorded at a median of 0.8 (IQR = 0.8) prior to Ramadan, decreased to 0 (IQR = 0.8) during Ramadan, and returned to 0.8 (IQR = 0.8) after Ramadan, demonstrating significant differences (P < .05 for all pairwise comparisons). Similarly, Chyper exhibited a median of 21.2 (IQR = 5) before Ramadan, which reduced to 18.3 (IQR = 4.55) during Ramadan, and subsequently increased to 21.8 (IQR = 5.05) post-Ramadan, with all pairwise comparisons indicating significant differences (P < .05).
Comparative Analysis of Metabolic and Glycemic Parameters Before, During, and After Ramadan.

Data are presented as median (interquartile range, IQR).
Statistical significance indicated; P1 compares Before vs During, P2 compares Before vs After, and P3 compares During vs After Ramadan; %TIR70-180 = time in range (70-180 mg/dL); %TAR181-250 = time above range (181-250 mg/dL); %TAR>250 = time above range (>250 mg/dL); %TBR54-69= time below range (54-69 mg/dL); %TBR<54 = time below range (<54 mg/dL).
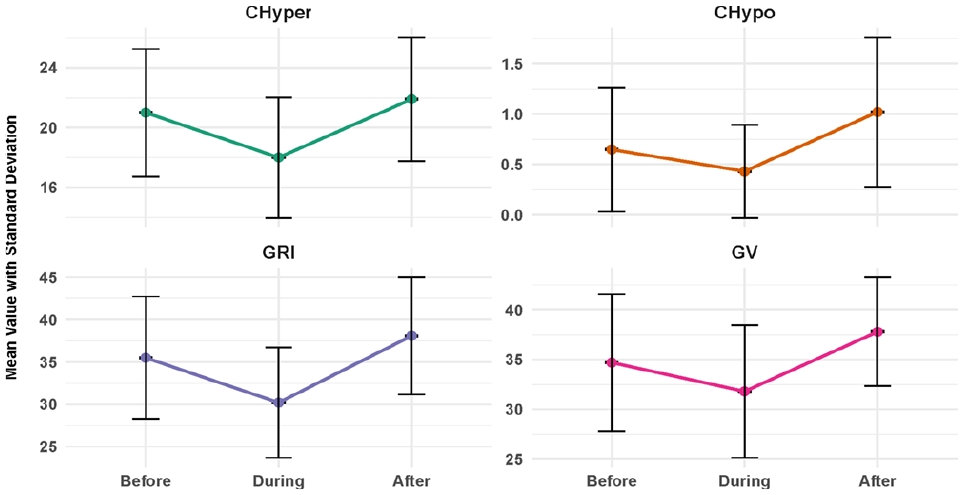
Variations in hypoglycemia component, hyperglycemia component, glycemia risk index, and glucose variability metrics before, during, and after Ramadan.
The GRI reflected a median of 35.2 (IQR = 10.3) before Ramadan, dropping to 30.5 (IQR = 8.08) during Ramadan, and rising again to 37.4 (IQR = 8.84) after Ramadan, with significant results for all comparisons (P < .001).
Trends in glycemic parameters and glycemic variability
The GV improved during Ramadan, showing a median of 36% (IQR = 8.6) before Ramadan, decreasing to 31.2% (IQR = 6.1) during Ramadan, and returning to 38.2% (IQR = 5.25) post-Ramadan (P < .05 for all pairwise comparisons). The number of low glucose events remained stable across all three periods, with a median of 2 (IQR = 1) before Ramadan, 2 (IQR = 2) during Ramadan, and 2 (IQR = 2) after Ramadan. No significant differences were noted between the pre-Ramadan and during-Ramadan periods (P1 = .189).
A comparable trend was seen in average glucose levels, which were recorded at a median of 187 mg/dL (IQR = 17.5) before Ramadan, decreased to 182 mg/dL (IQR = 14) during Ramadan, and returned to 187 mg/dL (IQR = 20) post-Ramadan.
The GMI% showed a median of 8% (IQR = 0.4) before Ramadan, decreased to 7.9% (IQR = 0.4) during Ramadan, and stabilized at 7.9% (IQR = 0.4) after Ramadan, with significant differences across all pairwise comparisons (P < .05). The active time of the sensor increased significantly from a median of 80% (IQR = 12) before Ramadan to 88% (IQR = 9) during Ramadan, then returned to 80% (IQR = 11.5) after Ramadan. Significant differences were noted between pre-Ramadan and during-Ramadan periods (P < .001) as well as between during-Ramadan and post-Ramadan periods (P < .001), with no significant difference between pre-Ramadan and post-Ramadan levels (P = .785).
The %TIR70-180 was 42% (IQR 15) before Ramadan, increased to 66% (IQR = 10.5) during Ramadan, and decreased to 41% (IQR = 15) after Ramadan. Significant differences were observed between pre-Ramadan and during-Ramadan periods (P < .001), as well as between during-Ramadan and post-Ramadan periods (P < .001); however, no significant difference was noted between pre-Ramadan and post-Ramadan levels (P = .169).
The %TBR54-69 was minimal, with medians of 1% (IQR = 1) before Ramadan, 0% (IQR = 1) during Ramadan, and returning to 1% (IQR = 1) after Ramadan. Significant differences were evident between pre-Ramadan and during-Ramadan levels (P = .005), as well as values between during-Ramadan and post-Ramadan values (P < .001); however, no significant difference was observed between pre-Ramadan and post-Ramadan levels (P = .165). The %TBR<54 was recorded with medians of 0% (IQR = 0) both before and during Ramadan and 0% (IQR = 1) after Ramadan.
Participants spent a median of 30% (IQR = 10) of their %TAR181-250 before Ramadan, decreasing to 24% (IQR = 9.5) during Ramadan, and returning to 30% (IQR = 8) after Ramadan. Significant differences were noted between pre-Ramadan and during-Ramadan periods (P < .001), as well as between during-Ramadan and post-Ramadan levels (P < .001), but no significant difference was found between pre-Ramadan and post-Ramadan levels (P = .630). Moreover, %TAR>250 had a median of 5.5% (IQR = 2) before Ramadan, increased to 6.3% (IQR = 3.15) during Ramadan, and decreased to 6% (IQR = 2.9) after Ramadan, with no significant differences noted across the periods.
Fasting Breaks and Hospital Visits Due to Glycemic Issues
Notably, 104 of 111 participants (93.7%) reported no fasting breaks due to glycemic issues, and all participants (100%) did not require hospital visits due to glycemic acute complications.
Diabetes Therapy-Related Quality of Life Questionnaire
The DTR-QOL questionnaire revealed high scores across all domains, indicating a generally positive therapy-related QoL among participants following Ramadan. The median overall DTR-QOL score was 5.48 (IQR = 5.28-5.69), with a scale score of 78.3 (IQR = 75.4-81.3), reflecting strong overall satisfaction with diabetes therapy.
The results presented in Table 3 and Supplemental Figure S2 provide an overview of the relationship between GRI zones and scores on the DTR-QOL after Ramadan. The included participants were categorized into two GRI zones: zone B (scores 21-40) and zone C (scores >40). The median score for the DTR-QOL scale’s median score was 78.3 (IQR = 75.4-81.3), with zone B participants achieving a median score of 78.3 and zone C participants scoring a median of 77.3. There were no significant differences in overall DTR-QOL scores between the zones (P = .85).
Relationship Between GRI Zones and the Scores on Diabetes Therapy-Related Quality of Life Questionnaire (DTR-QOL) After Ramadan.

Abbreviations: BSDA, burden on social and daily activities; ADT, anxiety and dissatisfaction with treatment; HG, hypoglycemia; ST, satisfaction with treatment.
GRI zone B.
GRI zone C.
Correlation Analysis of Glycemic Indices and Their Interrelationships
A significant positive correlation between average glucose levels and Chyper scores (Spearman’s rho = 0.277, P = .003) indicates that higher average glucose levels are associated with an increased risk of hyperglycemia. In addition, average glucose levels also show a significant positive correlation with GRI scores (Spearman’s rho = 0.239, P = .012), further suggesting that elevated average glucose correlates with higher GRI during Ramadan. Furthermore, the percentage of time the sensor is active positively correlates with GRI scores (Spearman’s rho = 0.194, P = .042). In contrast, other variables, including HbA1c, glucose variability, GMI, and low glucose events, did not demonstrate significant correlations with Chypo, Chyper, or GRI scores during Ramadan. The %TIR70-180 exhibited a negative correlation with GRI scores (Spearman’s rho = −0.177), indicating a trend suggesting that a higher %TIR70-180 is associated with lower GRI scores; however, this correlation was borderline insignificant (Figure 2). After Ramadan, no significant correlations were observed between the DTR-QOL scores and Chypo, Chyper, or GRI scores (Supplemental Figure S3).
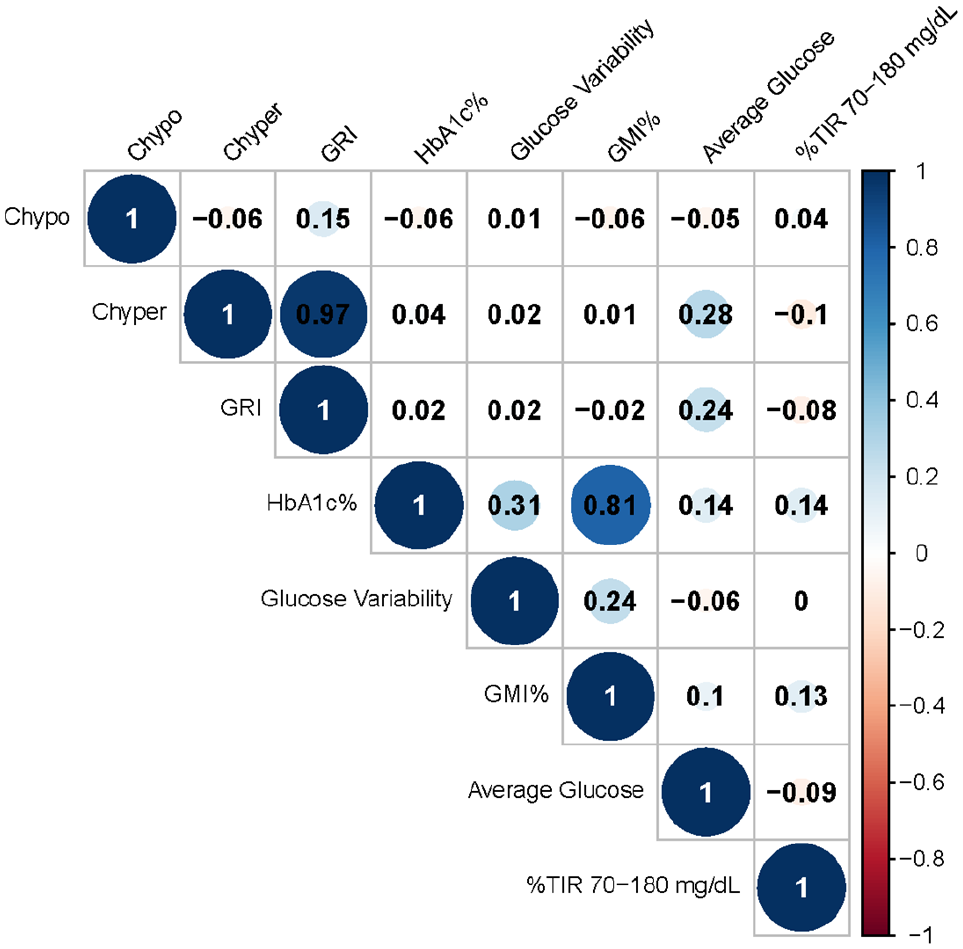
Correlation plot visually represents the relationships between various glycemic-related metrics during Ramadan.
Discussion
This study explored the impact of Ramadan fasting on the GRI and its components—CHypo and CHyper—alongside other CGM metrics in adults with T2D. By analyzing real-world data, it aims to enhance fasting safety and guide health care providers in optimizing care for patients observing Ramadan. Results showed a reduction in the GRI during Ramadan. Although statistical differences in GRI were observed between the three months, the numerical differences were small, and both values fell within the same zone B. This finding underscores the need for further investigation into the factors influencing these minor but significant changes, including physical activity, medication adjustments, and individual variability during fasting.
The GRI complements established glycometric parameters, such as %TIR70-180, by providing a more comprehensive assessment of glycemic control through the integration of both hypoglycemic and hyperglycemic episodes. This is especially beneficial for individuals prone to severe GV.16,17,20,31
Moreover, studies have linked GRI to various diabetes-related complications, including arterial stiffness, diabetic retinopathy (DR), and diabetic peripheral neuropathy (DPN).32 -34 A primary focus of GRI research is its relationship with arterial stiffness, 32 a known predictor of cardiovascular events and mortality that can be influenced by endothelial dysfunction and vascular inflammation. 35 Cai et al 32 demonstrated a strong correlation between an elevated GRI and higher levels of arterial stiffness. Likewise, GRI has been connected to DPN, 33 with chronic glucose fluctuations possibly exacerbating endothelial damage and nerve impairment through oxidative stress, inflammation, neuroinflammation, and microvascular injury.36 -38 The GRI’s association with DR, meanwhile, suggests that high GRI values could indicate an increased risk of DR,34,39 a leading cause of visual impairment in diabetes. 40
In practical terms, patients with high GRI values may benefit from more frequent monitoring and tailored therapeutic adjustments to mitigate the risk of cardiovascular complications, neuropathy, and retinopathy. Incorporating GRI into routine diabetes care could thus facilitate early intervention and improve long-term outcomes for individuals with T2D. By offering insights into both hypoglycemic and hyperglycemic risk, GRI allows clinicians to address glycemic fluctuations proactively—an especially valuable approach in the context of Ramadan fasting.
Beyond the GRI and its components, our study observed improvements in glycemic control among T2D patients during Ramadan fasting. Specifically, %TIR70-180 increased and HbA1c showed the modest reductions, while average glucose levels decreased slightly but returned to baseline after Ramadan. These findings suggest that well-managed fasting protocols can enhance glycemic metrics. This aligns with prior evidence demonstrating safe and effective glycemic improvements during Ramadan—Tahapary et al 7 reported significant reductions in fasting plasma glucose, HbA1c, and lipid profiles without a surge in hypoglycemic events, while Aydin et al 41 also confirmed reductions in fasting blood glucose, HbA1c, and BMI. Gad et al 42 showed that sodium-glucose cotransporter 2 inhibitor use improved glycemic control with minimal adverse effects, and another meta-analysis 43 involving 5554 participants found significantly lower HbA1c and fasting blood glucose levels post-Ramadan, although body weight remained largely unchanged.
Despite the overall benefits, certain challenges persist—particularly regarding hypoglycemia. A review by Abdelrahim et al 44 found sulfonylurea users face higher hypoglycemia rates than those on sodium-glucose cotransporter-2 (SGLT-2) or dipeptidyl peptidase-4 (DPP-4) inhibitors, emphasizing the need for medication adjustments and vigilant monitoring. In a case-control study by Al-Taher and Zabut, 45 some patients experienced reductions in weight and waist circumference, but fasting blood glucose remained unchanged while triglycerides rose, reinforcing the notion that Ramadan fasting outcomes can vary widely and should be managed on an individual basis.
Risk parameters, including GRI, CHypo, and CHyper, often rebound once Ramadan ends, reflecting a return to less-structured eating patterns and medication regimens. This underscores the importance of gradual medication adjustments and consistent follow-up after fasting. As studies indicate, personalized post-Ramadan transition plans can help sustain metabolic benefits and maintain long-term glycemic stability in T2D patients. 46
In our study, the DTR-QOL scores were uniformly high across all domains, pointing to a generally positive therapy-related QoL post-Ramadan. Median DTR-QOL scores did not differ significantly between GRI zones B (21-40) and C (>40), suggesting that even patients with broader glycemic fluctuations perceived minimal negative impact. Previous research similarly highlights the beneficial influence of Ramadan-focused programs on clinical outcomes and QoL.47 -49 However, some studies report different findings, such as reduced QoL among pediatric populations, 50 indicating that age, autonomy, and experience in diabetes self-management may shape QoL during fasting.
A positive correlation between average glucose levels and both CHyper and GRI reinforces the clinical importance of controlling mean glucose to lower hyperglycemic episodes in T2D. Existing literature links GV to complications such as arterial stiffness, echoing the need for metrics like CHyper and GRI in capturing hyperglycemic risk.51,52 Real-time CGM data remain pivotal during Ramadan when structured eating patterns can help stabilize glucose.53 -55 Su et al 56 further demonstrated how mean amplitude of glycemic excursions (MAGE) parallels GRI in identifying risk patterns in T2D. Notably, traditional markers such as HbA1c and low glucose events did not correlate strongly with CHypo, CHyper, or GRI during Ramadan, underscoring the unique value of continuous monitoring in these circumstances.
Finally, our study noted no hospital visits due to glycemic acute complications during Ramadan, aligning with other data suggesting that diabetes-related emergencies are not necessarily more prevalent with proper supervision.57 -59 Moreover, Chowdhury et al 60 reported no significant differences in clinical outcomes between fasting and non-fasting T2D patients with stage 3 CKD, while Elbarsha et al 61 found no notable change in emergency admissions or mortality rates across fasting and non-fasting periods. Collectively, these findings emphasize that Ramadan fasting can be safe for T2D patients when supported by individualized management, pre-Ramadan education, and routine glucose monitoring. Further research should aim to refine fasting protocols, ensuring maximum benefit and minimal risk for diverse T2D populations.
Strengths and Limitations
A key strength of this study is that it is among the first to investigate Ramadan fasting’s impact on the GRI and other CGM metrics in individuals with T2D. By focusing on GRI—a novel metric—this work offers a holistic view of glycemic stability during fasting, enabling clinicians to gain nuanced insights into both hypoglycemic and hyperglycemic tendencies. Such comprehensive data can help tailor fasting protocols and optimize medication regimens in real-world T2D management.
Despite these strengths, the study has several limitations. The relatively small sample size may limit the generalizability of the findings, and the short follow-up period—just one month post-Ramadan—restricts the evaluation of longer-term glycemic control. Accordingly, the sustainability of Ramadan-related improvements remains uncertain. The specific geographic setting further narrows the applicability of the results, as cultural and health care differences may influence outcomes in other regions. In addition, reliance on self-reported data for fasting breaks, medication adjustments, and other behaviors introduces potential recall bias and inaccuracies. Future research should include larger, more diverse populations and extended follow-up periods to verify whether observed benefits persist over time and across different demographic groups. Such efforts would provide clearer insights into the long-term sustainability of Ramadan-related glycemic improvements in T2D.
Conclusion
Our study found that Ramadan fasting positively impacted the GRI and its components—CHypo (hypoglycemia risk) and CHyper (hyperglycemia risk)—by improving glycemic stability and reducing glucose fluctuations in individuals with T2D. This suggests that structured fasting can promote a balanced glycemic profile. When combined with pre-Ramadan education and CGM use, Ramadan fasting can be safely implemented, offering significant glycemic benefits. The integration of CGM with GRI provides an effective monitoring framework, enabling real-time detection and management of glycemic events during fasting.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251321860 – Supplemental material for Implications of the Glycemia Risk Index in Assessing Metabolic Control and its Correlation With Therapy-Related Quality of Life During-Ramadan Fasting in Adults With Type 2 Diabetes
Supplemental material, sj-docx-1-dst-10.1177_19322968251321860 for Implications of the Glycemia Risk Index in Assessing Metabolic Control and its Correlation With Therapy-Related Quality of Life During-Ramadan Fasting in Adults With Type 2 Diabetes by Ayman Al Hayek, Wael M. Al Zahrani and Mohammed A. Al Dawish in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
Editorial assistance for this manuscript was provided by VivoSolve Ltd, London, The United Kingdom. The article processing charges were funded by Abbott Diabetes Care.
Abbreviations
ADT, anxiety and dissatisfaction with treatment; BMI, body mass index; CHypo, hypoglycemia component; CHyper, hyperglycemia component; CGM, continuous glucose monitoring; CV%, coefficient of variation; DPP-4, dipeptidyl peptidase-4; DR, diabetic retinopathy; DTR-QOL, diabetes therapy-related quality of life; GMI%, glucose management indicator; GV, glycemic variability; GRI, glycemic risk index; HbA1c, hemoglobin A1c; HG, hypoglycemia; IDF-DAR, International Diabetes Federation Diabetes and Ramadan; IQR, interquartile range; MAGE, mean amplitude of glycemic excursions; SD, standard deviation; ST, satisfaction with treatment; %TIR70-180, percentage of time in range (70-180 mg/dL); %TAR181-250, percentage of time above range (181-250 mg/dL); %TAR>250, percentage of time above range (>250 mg/dL); %TBR54-69, percentage of time below range (54-69 mg/dL); %TBR<54, percentage of time below range (<54 mg/dL).
Author Contributions
AAH and MAAD contributed to the study’s conception and design. Material preparation, data collection, and analysis were performed by AAH, MAAD, and WMAZ. All authors contributed to the writing of the first draft of the manuscript. AAH revised the manuscript critically for important intellectual content. All authors approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by our Ethics Committee (no. 1394).
Consent to Participate
All participants provided informed consent to partake in the study. Before enrollment, they were thoroughly briefed on the study’s aims, potential risks, and benefits. Participants were informed of their right to withdraw from the study at any point without penalty or loss of benefits to which they are otherwise entitled.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
