Abstract
Background:
Point-of-care (POC) strip-based blood glucose monitoring systems (BGMSs) used by health care professionals must be more robust than those used for patient self-testing, due to a higher likelihood of BG interferents present in the blood of hospitalized patients. The Cobas® Pulse system (Roche Diagnostics GmbH, Mannheim, Germany) is a novel strip-based POC BGMS designed for multiple patient use in professional health care environments. We report a series of experiments evaluating the effects of several potential interferents, and of extreme conditions, on the BGMS’s performance.
Methods:
Blood samples were spiked with glucose and various potential interferents or tested at extreme environmental conditions. Studies were performed using three lots of Cobas glucose test strips (Roche Diagnostics GmbH, Mannheim, Germany). Interference results over all test strip lots are expressed as the absolute or relative mean bias between test and control.
Results:
Compared with control, the mean absolute or relative bias did not exceed ±10 mg/dL or ±10% for any endogenous substance or drug tested, any combination of hematocrit level or glucose concentration over the ranges tested, or any test at altitudes up to 4300 m above sea level, with any of the three test strip lots. Varying oxygen concentrations (partial pressure) in a blood sample did not affect glucose measurements. All interference results were within the specified accuracy limits. Contamination of blood samples with skin disinfectants, extreme levels of ascorbic acid, or hematocrit levels outside the claimed range did not cause wrong test results.
Conclusions:
POC BGMS performance was not altered by interfering substances after a rigorous evaluation under a wide range of conditions.
Introduction
Point-of-care (POC) strip-based blood glucose (BG) meters are widely used by patients for self-monitoring of BG and by health care professionals for assessment of BG levels in health care settings. BG monitoring systems (BGMSs) used by health care professionals must be more robust than those used for patient self-testing. Hospitalized patients have inherently more complex medical issues than outpatients and are likely to be receiving multiple medications or have wide variations in laboratory parameters, both of which have the potential to interfere with BG measurements.1-4 This is particularly true in critically ill patients, where stress-induced hyperglycemia is common even without a prior diabetes mellitus diagnosis. 5 Hyperglycemia has been associated with longer hospital stays, increased complication rates, and higher mortality.6,7 Hence, control of hyperglycemia through frequent BG monitoring is vital in critically ill patients. Control of hyperglycemia is often achieved through treatment with insulin, which requires frequent BG monitoring for safe and effective use.
Biochemical, chemical, and electrochemical side reactions resulting from extreme variations in the properties of blood or the presence of electroactive substances, have the potential to interfere with the detection principle of BGMSs.8,9 Thus, potential interference from endogenous substances, drugs, and variations in hematocrit levels and environmental conditions must be thoroughly evaluated in the development of any novel strip-based BGMS. To facilitate this process, the U.S. Food and Drug Administration (FDA) has issued a guidance document for industry and FDA staff 10 that includes recommendations for evaluating the potential for interference with BGMSs intended for use in professional health care settings.
The Cobas® Pulse system (Roche Diagnostics GmbH, Mannheim, Germany) is a novel strip-based POC BGMS designed specifically for multiple patient use in professional health care environments. 11 A multicenter evaluation using samples from patients in diverse clinical settings by intended POC test operators showed that the BGMS is effective for measuring BG in venous, arterial, neonatal arterial, neonatal heel stick, and capillary whole blood samples, and that it is a valid alternative to other currently approved testing methods. 12
The BGMS has been designed with multiple failsafes to improve accuracy and significantly reduce the risk of incorrect test results caused by user error.
In principle, three basic interference mechanisms can potentially lead to problematic deviations in BG concentration measurements in a BGMS with enzymatic and electrochemical components. First, nonspecific reactions between the enzyme system and nonglucose sugars in a blood sample can cause false-positive bias. Second, the physical characteristics of the blood sample, including variation in hematocrit levels, lipid, protein, or electrolyte levels, and temperature can have a direct effect on the voltammetric measuring principle. Third, certain endogenous or exogenous substances (e.g., drugs) can electrochemically or chemically react with components of the detection chemistry and cause biased BG concentration measurements (mostly high and low bias). Moreover, the use of a dehydrogenase, rather than an oxidase enzyme, eliminates the influence of oxygen concentration in a blood sample. Variations in the physical components of blood are compensated for by incorporating a poly-frequent impedimetric measuring method in addition to the voltammetric measuring method, simultaneous application of both methods in the test sequence, and by the algorithm that calculates the glucose concentration. To compensate for endogenous or exogenous substances, which can produce background currents, both electrodes in the test strip are coated with the same reagent, but only one electrode contains the glucose-specific enzyme. Polarization is carried out in both directions during the test sequence and by appropriately calculating the measurement data, response currents that are not correlated to the glucose concentration can be canceled out.
A key goal during the development of the BGMS was demonstrating the absence of clinically relevant interference with BG measurements. 11 Here we report the results of a series of experiments designed to evaluate the effects of endogenous substances, frequently used therapeutic drugs, and variation in hematocrit levels, oxygen partial pressure, and altitude on the performance of the BGMS. We also report a series of experiments (“flex studies”) designed to evaluate the effects of extremes of conditions (use of skin disinfectants, high ascorbic acid concentrations, hematocrit levels outside of claimed ranges) to assess risk mitigation in the context of BGMS risks or use by untrained operators.
Methods
This analytical study was performed at Roche in Mannheim, Germany. As such, Ethics Committee approval was only necessary for studies to which § 24 Nr. 1–3 German Medical Device Act (“MPG”) apply (i.e., invasive sampling is conducted either exclusively or to a considerably higher degree to obtain a specimen for the purpose of a performance evaluation of an in vitro diagnostic medical device; in the context of the performance evaluation study, additional invasive or other stressful examinations are conducted; or the results obtained in the context of the performance evaluation are to be used for diagnostic purposes without it being possible to confirm them by means of established procedures). In the remaining cases, the consent of the person from whom the specimen was to be taken was necessary, insofar as this person’s personal rights or commercial interests were affected. The Ethics Committees in Germany decline to review clinical studies using samples that do not fall under the conditions described above, unless they are performed by a medical doctor. In case of involvement of a medical doctor, the Ethics Committee review and counsel the doctor.
The blood samples used in this study were collected from healthy Roche employees in Germany under informed consent via the Roche Medical Services. Samples were collected in anticoagulant tubes and stored overnight at room temperature for glycolyzing. They were then spiked with glucose (to adjust for different glucose levels) and the various interference substances according to internal test protocols. Depending on the experiment, the hematocrit level of the samples was also altered.
Studies of endogenous substances and drugs were performed according to the stipulations of the Clinical and Laboratory Standards Institute (CLSI) EP07-A3 13 and CLSI EP37-A1. 14
Studies of endogenous substances, hematocrit level, oxygen partial pressure, and altitude, and the flex studies, were conducted with spiked venous lithium heparin whole blood. Lithium heparin whole blood was also used to test potentially interfering drugs if the drug was dissolved in an inorganic solvent. Studies with drugs were conducted with spiked lithium heparin plasma if the drug was not sufficiently soluble in aqueous media and had to be dissolved in an organic solvent. In the case of mannitol, the drug was dissolved directly in lithium heparin whole blood.
Both endogenous substances and drugs were tested across three glucose concentration ranges (50-70, 110-130, and 225-270 mg/dL).
Performance was evaluated at four partial pressures of oxygen (30, 60, 90, and 200 mm Hg) that were confirmed on a Cobas b 221 system (blood gas and electrolyte analyzer, Roche Diagnostics GmbH, Mannheim, Germany) and across three glucose concentration ranges (50-70, 110-130, and 225-270 mg/dL).
Studies of hematocrit levels were conducted with 13 hematocrit values (5%, 10%, 15%, 20%, 25%, 30%, 35%, 50%, 55%, 60%, 65%, 70%, and at a nominal value of 42%) across five glucose concentration ranges (30-50, 51-110, 111-150, 151-250, and 251-400 mg/dL).
Studies of altitude were conducted in Mannheim, Germany at an altitude of approximately 90 m above sea level (control) and in a helicopter at altitudes up to approximately 4300 m above sea level across three glucose concentration ranges (50-65, 100-120, and 200-250 mg/dL). The crew was provided with oxygen as a safety measure. After the height was achieved, the sample container was opened, resealed, and inverted to allow the blood sample to adjust to atmospheric oxygen pressure before testing.
All studies were performed using three lots of Cobas glucose test strips (Roche Diagnostics GmbH, Mannheim, Germany). In evaluations of endogenous substances, drugs, and hematocrit levels, each test strip lot was tested with 10 replicates per glucose concentration and condition. In evaluations of oxygen partial pressure, each strip lot was tested with 16 replicates per glucose concentration and condition. In evaluations of altitude, each strip lot was tested with eight replicates per glucose concentration and condition.
Endogenous Substances and Drugs
Each potential interfering endogenous substance (or drug) was measured against a control condition in blood or plasma containing the same solvent as the test condition. Where possible, the concentrations tested were derived from CLSI EP7/EP37 or FDA guidance,10,13,14 or published literature; for details please refer to the lists of potential interferents (Supplementary Tables S1 and S2).
Hematocrit Levels
The results for hematocrit levels were compared to BG results obtained with a comparator method (plasma-based hexokinase [HK] method on Cobas 6000 Analyzer Series, Roche Diagnostics GmbH, Mannheim, Germany) and to the mean value for the nominal hematocrit value of 42%.
Oxygen
The results for oxygen partial pressure were compared to BG results obtained with the comparator method used for hematocrit, above.
Statistical Methods
Results over all test strip lots are expressed as the absolute or relative mean bias between test and control. Upper and lower 95% confidence intervals were calculated for informational purposes only.
Acceptance Criteria
Acceptance criteria for endogenous substances and drugs were x ±10.0 mg/dL for glucose concentrations <75 mg/dL, where x is the mean absolute bias between test and control value, and x ±10.0% for glucose concentrations ≥75 mg/dL, where x is the mean relative bias between test and control value.
Acceptance criteria for hematocrit levels were x ±10.0 mg/dL for glucose concentrations <75 mg/dL, where x is the mean absolute bias between test and reference value, and x ±10.0% for glucose concentrations ≥75 mg/dL, where x is the mean relative bias between test and reference value. Note that the bias is not only between the test and reference value, but also between the test and mean value for the nominal hematocrit value of 42%.
Acceptance criteria for altitude were x ±10.0 mg/dL for glucose concentrations <100 mg/dL, where x is the mean absolute bias between test and control condition, and x ±10.0% for glucose concentrations ≥100 mg/dL, where x is the mean relative bias between test and control condition.
Acceptance criteria for oxygen partial pressure required that 95% of results fall within ±12 mg/dL (for glucose concentrations <75 mg/dL) and ±12% (for glucose concentrations ≥75 mg/dL) of the reference value, and that 98% of glucose results for within ±15 mg/dL (for glucose concentrations <75 mg/dL) and ±15% (for glucose concentrations ≥75 mg/dL) of the reference value.
Flex Studies
Methodologies for flex studies are shown in the Supplementary Information.
Data and Resource Availability
This study was conducted in accordance with applicable regulations. There may be ethical, legal, or other restrictions on sharing the de-identified dataset used for our analysis. Please send any data requests to
Results
Endogenous Substances
When compared with control, the mean absolute or relative bias did not exceed ±10 mg/dL or ±10% for any endogenous substance tested with any of the three strip lots. Pooled results for 30 replicates (10 for each of three strip lots tested) are presented in Table 1.
Potential Interference by Endogenous Substances Pooled Over Three Test Strip Lots.
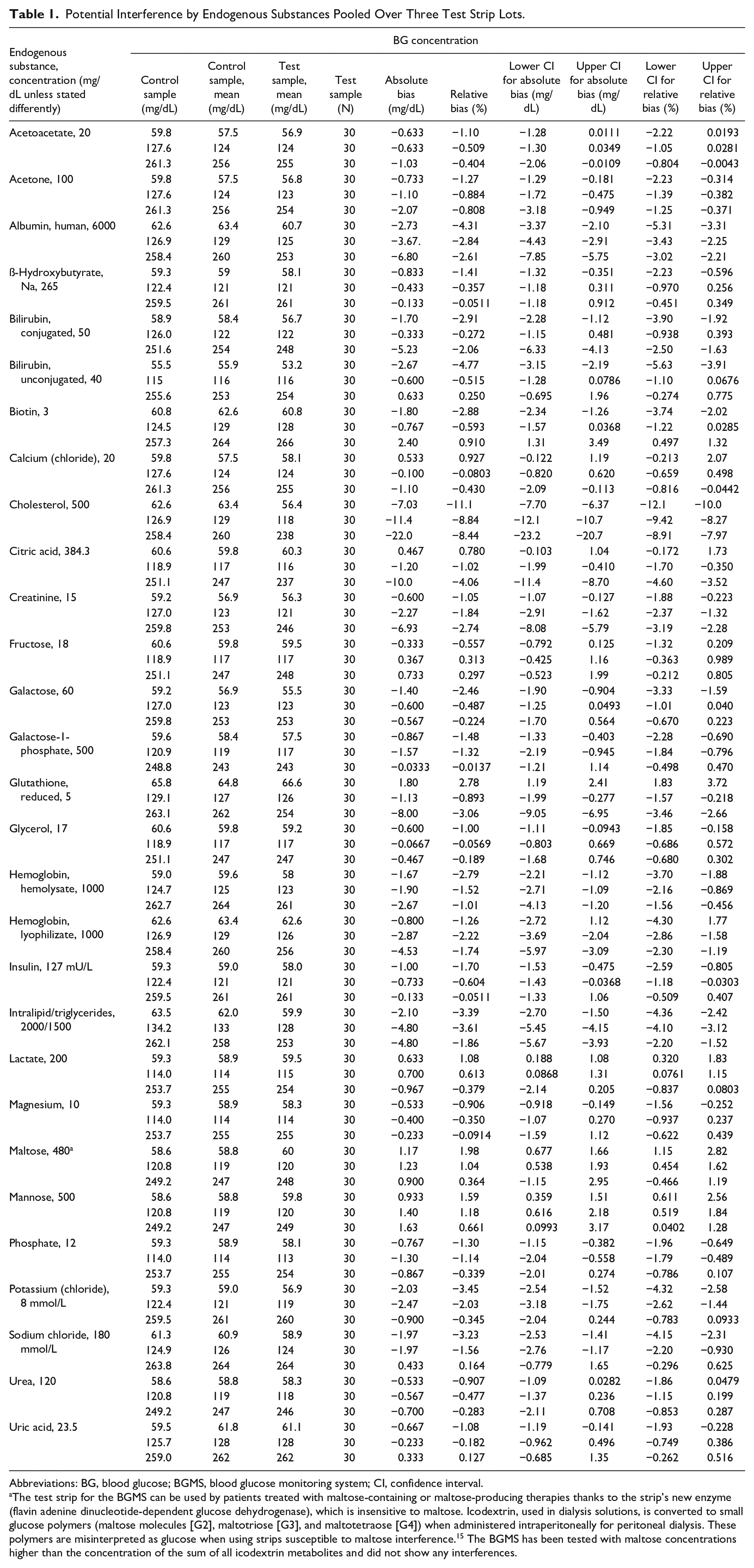
Abbreviations: BG, blood glucose; BGMS, blood glucose monitoring system; CI, confidence interval.
The test strip for the BGMS can be used by patients treated with maltose-containing or maltose-producing therapies thanks to the strip’s new enzyme (flavin adenine dinucleotide-dependent glucose dehydrogenase), which is insensitive to maltose. Icodextrin, used in dialysis solutions, is converted to small glucose polymers (maltose molecules [G2], maltotriose [G3], and maltotetraose [G4]) when administered intraperitoneally for peritoneal dialysis. These polymers are misinterpreted as glucose when using strips susceptible to maltose interference. 15 The BGMS has been tested with maltose concentrations higher than the concentration of the sum of all icodextrin metabolites and did not show any interferences.
Drugs
When compared with control, the mean absolute or relative bias did not exceed ±10 mg/dL or ±10% for any drugs tested with any of the three strip lots. Pooled results for 30 replicates (10 for each of three strip lots tested) are presented in Table 2.
Potential Interference by Drugs Pooled Over Three Test Strip Lots.
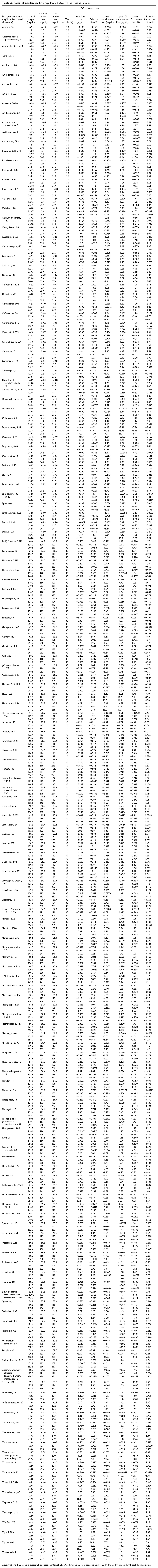
Abbreviations: BG, blood glucose; CI, confidence interval; EDTA, ethylenediaminetetraacetic acid; HES, hydroxyethyl starch; PAM, pralidoxime iodide.
Hematocrit Level
When compared with results obtained by the plasma-based HK comparator method, the mean absolute or relative bias did not exceed ±10 mg/dL or ±10% for any combination of hematocrit level or glucose concentration over the ranges tested (Table 3). This was also true for the comparison to the nominal hematocrit value of 42% as a control.
Mean Results for the Combination of Hematocrit 5-70% and BG Concentration Range 30-400 mg/dL Pooled Over Three Lots.
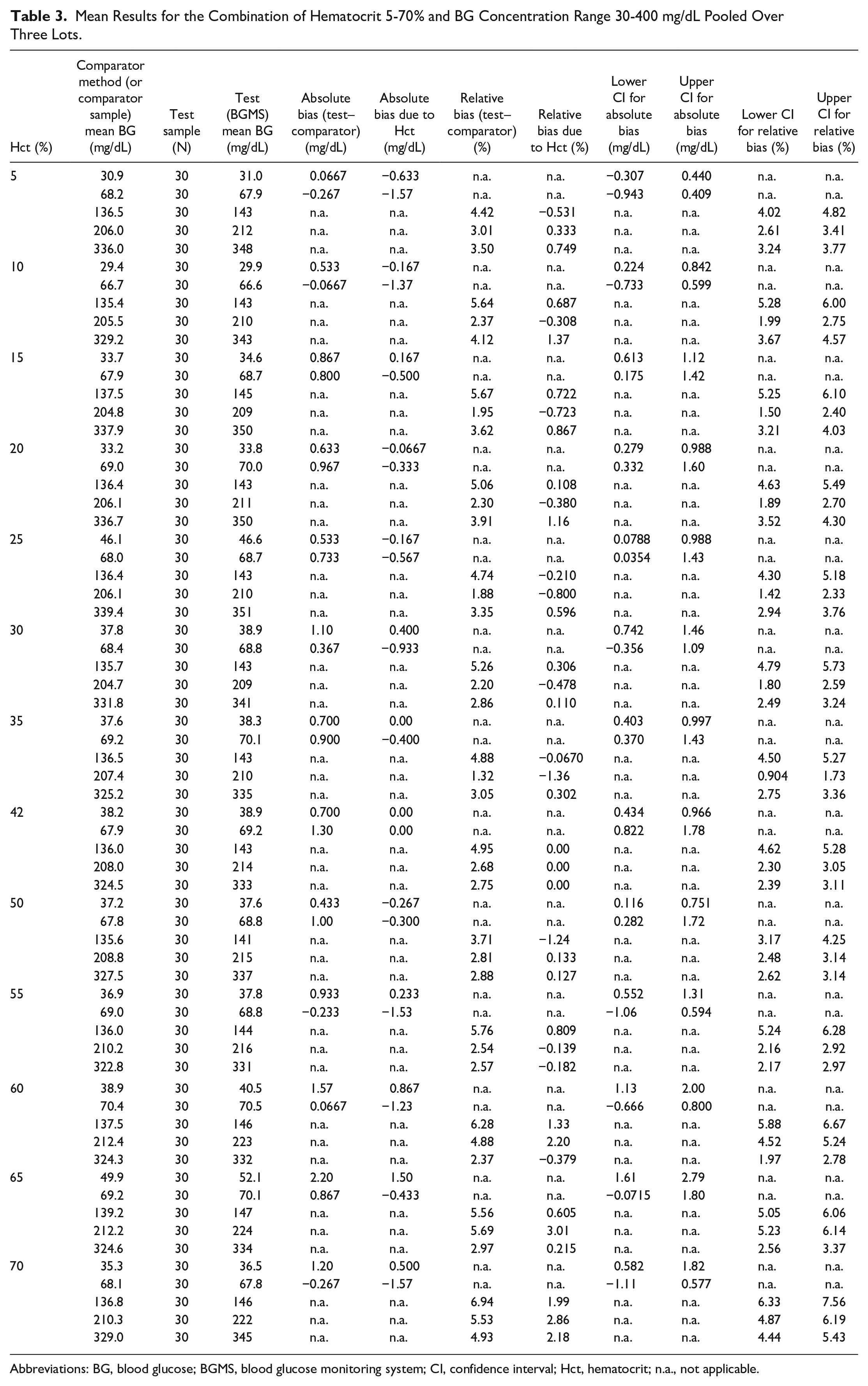
Abbreviations: BG, blood glucose; BGMS, blood glucose monitoring system; CI, confidence interval; Hct, hematocrit; n.a., not applicable.
Altitude
When compared with control, the mean absolute or relative bias did not exceed ±10 mg/dL or ±10% for any test at altitudes up to 4300 m above sea level with any of the three strip lots (Table 4).
Effects of Altitude.
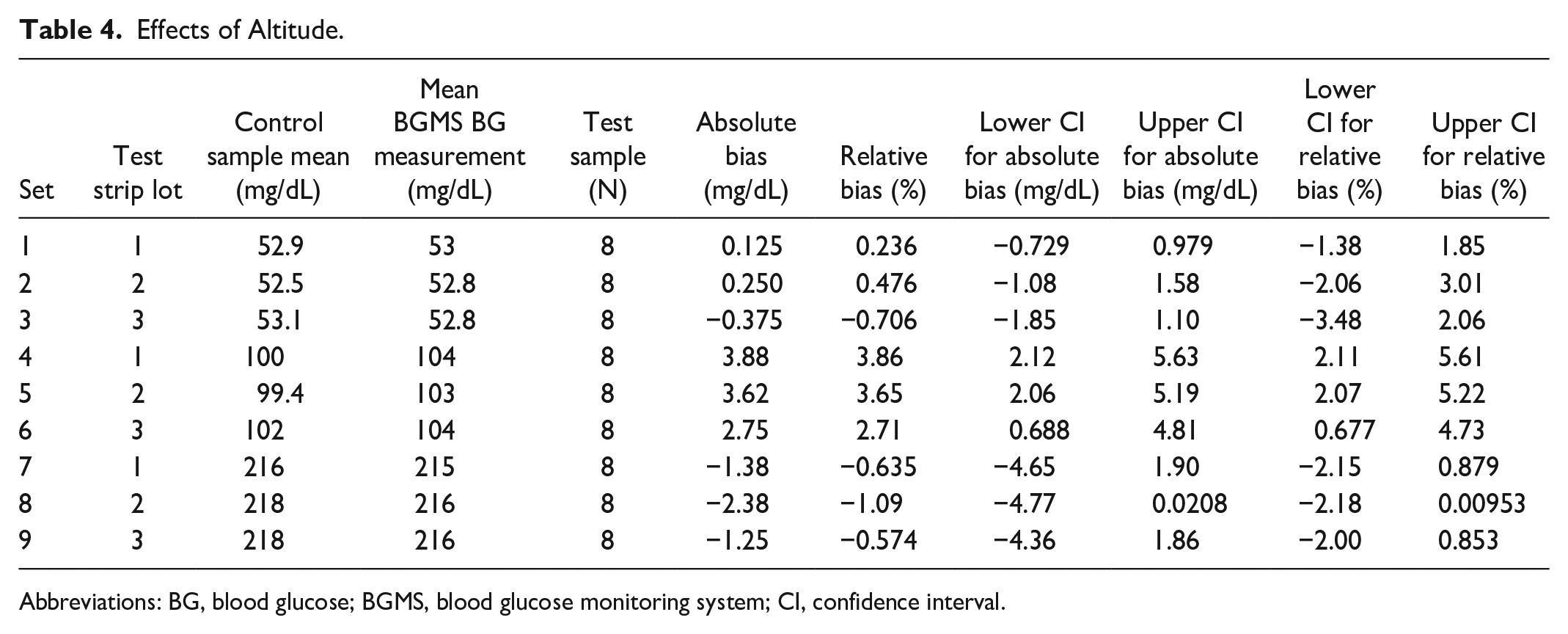
Abbreviations: BG, blood glucose; BGMS, blood glucose monitoring system; CI, confidence interval.
Oxygen Partial Pressure
When compared with results obtained by the plasma-based HK comparator method, all test results with all strip lots were within ±12 mg/dL or ±12% (absolute or relative) for each of the three concentration ranges tested (50-70, 110-130, and 225-270 mg/dL; Table 5).
Analysis of pO2 Interference Testing Accuracy per BG Level and Test Strip Lot for All pO2 Levels.
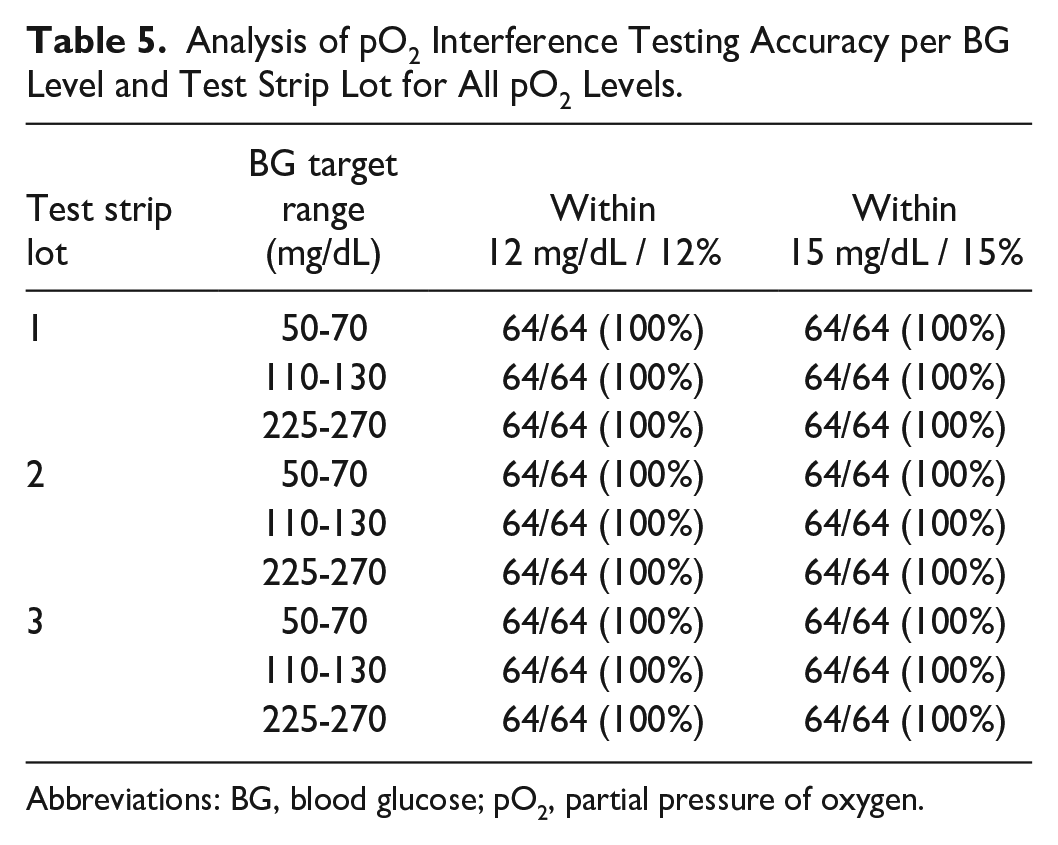
Abbreviations: BG, blood glucose; pO2, partial pressure of oxygen.
Flex Studies
Results for skin disinfectants met the acceptance criteria. Accuracy standards were met with proper usage of skin disinfectants. No test result was within the limits for the erroneous results zone. In cases of improper usage of the disinfectants, the failsafe was effective.
All measurements of ascorbic acid concentrations higher than 30 mg/dL were failsafe effective, and for samples with ascorbic acid concentrations of 15 and 30 mg/dL, accurate glucose readings were shown.
Correct glucose results were shown, and no failsafe was needed for hematocrit levels outside of claimed ranges.
Discussion
The BGMS assessed in the current analysis has been designed to include multiple mechanisms to reduce the potential for interference. Studies included in the comprehensive interference analysis were designed to rigorously evaluate the potential for endogenous substances and drugs, and variations in hematocrit level, oxygen partial pressure, and altitude to interfere with the performance of the BGMS. No interfering substance or condition was identified that altered the performance of the BGMS, and variations were all well inside the acceptance criteria.
In total, 29 endogenous substances and 160 drugs were evaluated in the present interference study. The results demonstrate that the BGMS is free from interference by substances that interfere with other strip-based BGMSs, such as bilirubin, galactose, and other nonglucose sugars, triglycerides, and uric acid, which are endogenous substances that have been shown to interfere with the performance of other strip-based BGMSs,8,16,17 but not with the BGMS in this analysis. This is also true for the drugs acetaminophen, ascorbic acid, dopamine, mannitol, and icodextrin metabolites.17-20
The potential for hematocrit level to interfere with the performance of the BGMS was thoroughly evaluated. Wide variation in hematocrit level has been shown to interfere with the performance of some strip-based BGMSs, with higher hematocrit levels being associated with lower BG values.17,21-23 A large proportion of patients in adult intensive care units have abnormally low hematocrit levels and these values can also fluctuate daily. 24 Conversely, patients in neonatal intensive care units can have abnormally high hematocrit levels. 25 The performance of the BGMS was evaluated over a wide range of hematocrit values (5-70%) and glucose concentrations (30-400 mg/dL) without any evidence of interference.
Oxygen has the potential to interfere with the performance of BGMSs. 8 The BGMS in this analysis is designed with oxygen-independent chemistry, eliminating per design a bias associated with changes in sample oxygen partial pressure or ambient oxygen concentration. The results of the present analysis show that the BGMS is not affected by variations in oxygen partial pressure ranging from approximately 30 to 200 mm Hg, or by elevation ranging up to approximately 4300 m above sea level.
In the flex studies, results demonstrated the BGMS’s ability to measure accurately in cases of contamination of a blood sample with skin disinfectants, high levels of ascorbic acid, or hematocrit levels outside of the claimed range.
The present study has certain limitations. All analyses involved contrived blood samples and each interfering substance or condition was evaluated in isolation. Blood samples taken from patients in health care settings may be more complex. For example, the concentration of endogenous substances may be higher or lower than that contained in our contrived samples, and patients often receive several drugs concurrently. Samples were prepared and analyzed by trained technicians in our study, whereas the instrument has been designed for use by health care professionals who may not necessarily have been trained on the proper use of the BGMS. Finally, not all potential interfering substances or environmental conditions were assessed.
Conclusions
POC performance of the BGMS in this analysis was not altered by interfering endogenous substances or drugs commonly found in the blood of hospitalized patients after a rigorous evaluation under a wide range of conditions (extreme variations in hematocrit level, partial pressure of oxygen, or altitude). Contamination of blood samples with skin disinfectants, extreme levels of ascorbic acid, or hematocrit levels outside the claimed range did not cause wrong test results.
Such a BGMS seems to be reliable and therefore of high value to patients in professional health care settings.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251319344 – Supplemental material for Evaluation of Potential Interference Caused by Endogenous Substances, Drugs, and Variations of Blood Sample Properties and Environmental Conditions, With Blood Glucose Concentrations Measured With a New Strip-Based Blood Glucose Monitoring System
Supplemental material, sj-docx-1-dst-10.1177_19322968251319344 for Evaluation of Potential Interference Caused by Endogenous Substances, Drugs, and Variations of Blood Sample Properties and Environmental Conditions, With Blood Glucose Concentrations Measured With a New Strip-Based Blood Glucose Monitoring System by Michael Marquant, Simon Gessler, Michael Diechtierow, Julia Gerber and Zekiye Adar née Oeztuerk in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
Research support in the form of third-party medical writing assistance for this manuscript, furnished by Blair Jarvis, MSc, on behalf of Nucleus Global, an Inizio Company, was provided by Roche Diagnostics.
Abbreviations
BG, blood glucose; BGMS, blood glucose monitoring system; CI, confidence interval; CLSI, Clinical and Laboratory Standards Institute; EDTA, ethylenediaminetetraacetic acid; FDA, Food and Drug Administration; Hct, hematocrit; HES, hydroxyethyl starch; HK, hexokinase; LER, limits for erroneous results; n.a., not applicable; PAM, pralidoxime iodide; pO2, partial pressure of oxygen; POC, point of care.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Michael Marquant, Simon Gessler, Michael Diechtierow, and Zekiye Adar née Oeztuerk are employees of Roche Diagnostics GmbH and hold stocks/shares in F. Hoffmann-La Roche Ltd. Julia Gerber is an employee of Roche Diagnostics International Ltd and holds stocks/shares in F. Hoffmann-La Roche Ltd. All authors received third-party writing assistance from Roche.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The development of the blood glucose monitoring system and the work described in this article was funded by Roche Diagnostics GmbH, Mannheim, Germany (no grant number applicable). The blood glucose monitoring system is approved for clinical use in Europe (CE mark-accepting countries). The blood glucose monitoring system is not yet available in the United States, but it is FDA-cleared for 510K and CLIA waived for use with arterial and heel stick whole blood samples. All product names and trademarks are the property of their respective owners.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
