Abstract
Background:
To evaluate the progression of diabetic retinopathy (DR) after the initiation of automated insulin delivery (AID) systems in adults with type 1 diabetes (T1D).
Methods:
In this longitudinal study with 152 adults, retinal exams and clinical variables were collected before and after AID initiation up to 2.7 years. The DR worsening was defined as an increase in Early Treatment of Diabetic Retinopathy Study (ETDRS) scores or qualitative retinal exam.
Results:
A total of 152 adults with mean age of 42 years (57% female), 26 years of T1D duration, and mean baseline HbA1c of 7.6% (60 mmol/mol) were included in this analysis. Of 152 adults with T1D, 42 (28%) adults had DR worsening after AID initiation. After adjusting for age, diabetes duration, and sex, baseline HbA1c (odds ratio [OR] = 2.1 [1.34-3.04]) and low-density lipoprotein cholesterol (LDL-C) >100 mg/dL with HbA1c >8% (OR = 3.33 [1.12-9.91]) were associated with two- and three-fold increased risk for DR worsening, respectively. The decline of HbA1c with AID initiation between DR worsening and no-DR worsening groups was not significant (−0.38 ± 1.2% vs −0.47 ± 0.9%;
Conclusions:
Higher baseline HbA1c with LDL-C >100 mg/dL may be associated with DR worsening after initiation of AID systems in T1D. Those with elevated HbA1c should get periodic ophthalmic examination after AID initiation to detect progression of DR. Prompt diagnosis may result in timely treatment.
Introduction
Rapid improvement in HbA1c in people with type 1 and 2 diabetes has been shown to worsen diabetic retinopathy (DR).1,2 In the Diabetes Control and Complications Trial (DCCT), the intensive treatment group had a two-fold increase in early worsening of DR compared with the conventional therapy group at 6- and/or 12-month visit.
3
In a recent study, rapid reduction in HbA1c with weekly glucagon-like peptide-1-receptor-agonist semaglutide in people with type 2 diabetes was also associated with a two-fold increased risk of DR worsening (hazard ratio [HR] = 1.76; 95% confidence interval [CI] = [1.11-2.78];
Automated insulin delivery (AID) systems have been shown to improve HbA1c without increasing hypoglycemia in people with type 1 diabetes.6-9 The improvement in HbA1c is fast with the use of AID systems mainly due to rapid improvement in nocturnal glucose. To our knowledge, DR worsening after initiation of AID system in relation to change in HbA1c has not been studied. Therefore, we aimed to investigate the DR progression after AID initiation in adults with type 1 diabetes.
Methods
Subjects and Study Design
In this longitudinal, retrospective cohort study, adults with type 1 diabetes at the Barbara Davis Diabetes Center who initiated Tandem Control-IQ between January 17, 2020, and January 20, 2022, and had at least one retinal examination before and after AID initiation were included in this study. Patients who were pregnant during the study were excluded.
For the AID systems, Tandem t:slim X2 Control-IQ system was selected for this study because it was the only FDA-approved advanced hybrid closed-loop system that provides auto corrections in addition to basal modulation during the study. It has been shown to improve glycemic outcomes in youth, adults, and older adults with type 1 diabetes.7,10,11 The Tandem t:slim X2 with Control-IQ technology (referred to as AID onwards) was commercially released in the United States in January 2020, and the release date was taken as the midpoint of the data collection period. The AID initiation date, HbA1c values with measurement dates, and eye examination results with exam dates were collected longitudinally before and after AID initiation between June 2017 and June 2022.
The following clinical variables were collected up to 2.7 years (till June 2022) from baseline, determined as the most recent record before AID initiation; demographics, age, sex, diabetes duration, body mass index (BMI) and race/ethnicity, insurance type, estimated glomerular filtration (eGFR), albumin/creatinine ratio, low-density lipoprotein cholesterol (LDL-C), hypertension status (yes/no; based on ICD-codes), smoking status (yes/no), and prescription of anti-hypertension drugs (yes/no; including angiotensin-converting enzyme [ACE] inhibitors). Two investigators (H.K.A. and K.E.K.) manually crosschecked electronic medical reports retrieved data for accuracy.
Qualitative (eg, no retinopathy, mild non-proliferative diabetic retinopathy [NPDR], mild-to-moderate NPDR, moderate NPDR, moderate-to-severe NPDR, severe NPDR, and proliferative DR) as well as quantitative reports (Early Treatment of Diabetic Retinopathy Study [ETDRS] scores) 12 of retinal exam for each eye were collected. This study was approved by the Colorado Multiple Institutional Review Board (COMIRB).
Statistical Analysis
In this retrospective feasibility study, no formal sample size calculation was performed. Continuous data were presented as mean and standard deviation, and categorical data were presented as absolute numbers and percentages. Retinal exams and HbA1c measurements were separated into time periods before and after the initiation of AID as baseline to 3 months, 3 to 6 months, 6 to 12 months, 12 to 24 months, and >24 months. For the multiple HbA1c and ETDRS values in the same period, the mean value was used. An increase in ETDRS score or progression in qualitative retinal exams in either eye after AID initiation compared with baseline was categorized as DR worsening. Patients were separated into two groups (DR worsening vs no-DR worsening) based on the progression of DR in either eye. Student’s t-test was used to compare the continuous baseline characteristics of two groups, whereas a Chi-square test was used to compare categorical baseline characteristics. Linear mixed models were used to examine DR levels by time points and by HbA1c values, adjusted for diabetes duration, age, and gender. Logistic regression models were used to examine the odds of worsening retinopathy following AID initiation. Qualitative DR stepwise changing was numbered in a stepwise fashion according to severity, Pearson’s correlation test was performed between the change in HbA1c and the number of steps in the worsening of DR grading. SAS software was used for statistical analysis, and
Results
Of 683 adults with type 1 diabetes who initiated the AID system, 152 patients meeting inclusion criteria were included in the analysis (Supplementary Figure 1). The mean age of participants was 42 ± 14 years with 26 ± 13 years of diabetes duration; 57% were female and 85% were non-Hispanic White (Table 1).
Characteristics of Study Population and Separated Groups by Worsening of Diabetic Retinopathy.

Abbreviations: DR, diabetic retinopathy; NHW, non-Hispanic Whites; ACR, albumin/creatinine ratio; ETDRS, early treatment of diabetic retinopathy study score; LDL-C, low-density lipoprotein–cholesterol; BMI, body mass index (kg/m2); eGFR, estimated glomerular filtration rate.
Three subjects could not be classified because of missing retinopathy data in one eye at one of the time points.
Of 152 participants, 42 (28%) had DR worsening over a mean of 1.6 years after an AID initiation. Most of these progressions were from no DR to mild non-proliferative DR. The baseline characteristics of participants by two groups (DR worsening and no-DR worsening) are shown in Table 1. Baseline HbA1c was significantly higher in the DR worsening group compared with no-DR worsening group (8.0 ± 1.5 vs 7.4 ± 1.1;
The time course of mean HbA1c and ETDRS scores before and after AID initiations were shown in Figure 1. After the AID initiation, HbA1c decreased significantly over time (
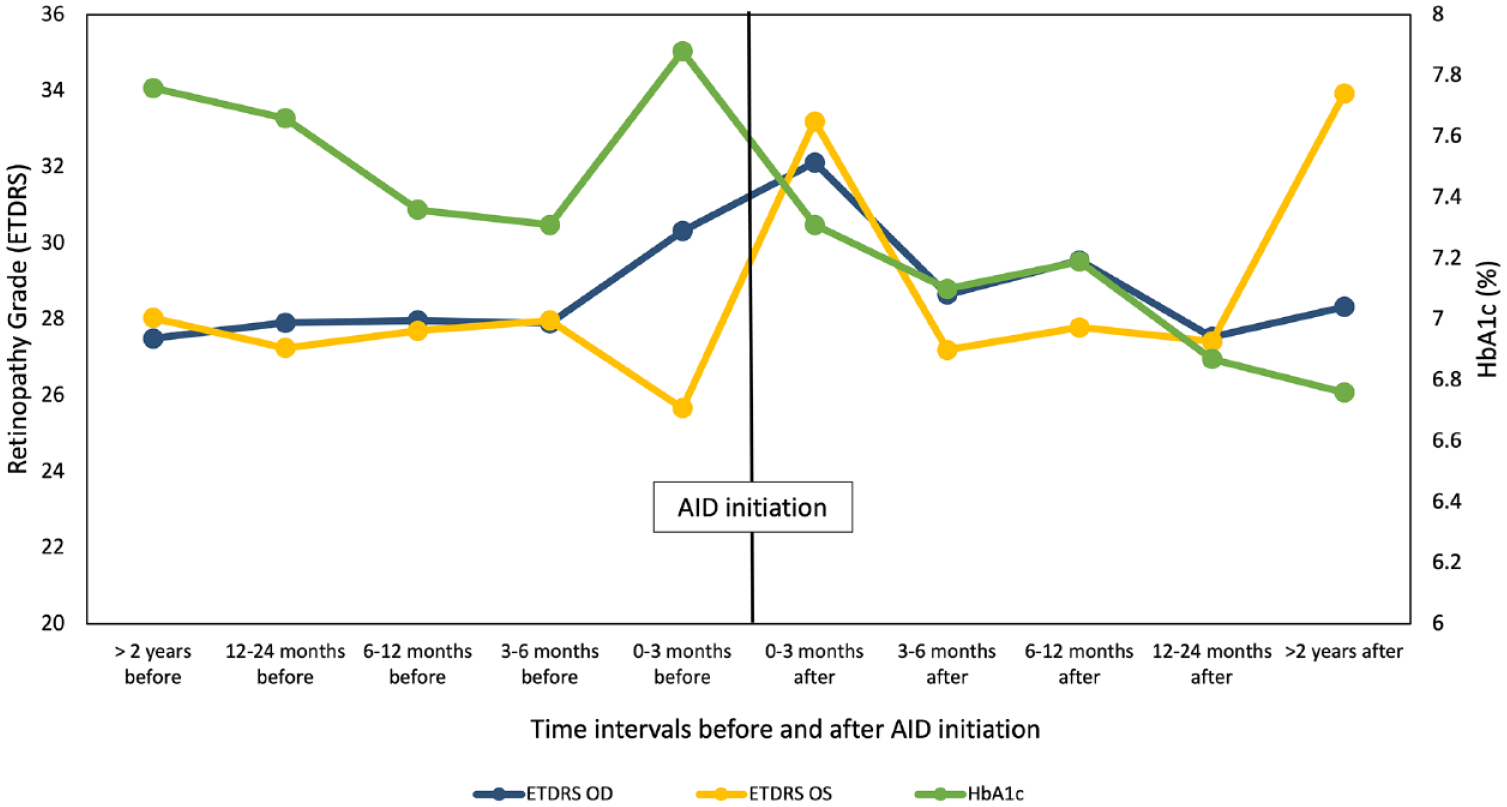
Mean HbA1c and retinopathy grade (ETDRS scores) for both eyes before and after AID initiation.
There was no correlation between the change in HbA1c and the worsening of DR stepwise changing (r = −0.006,
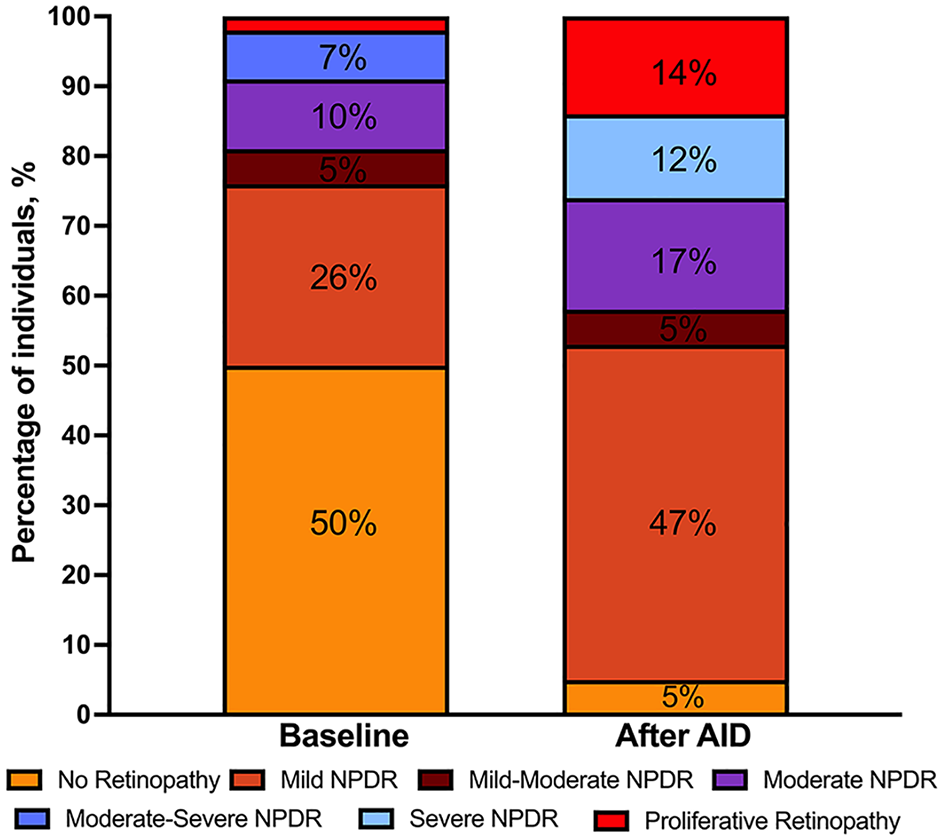
Qualitative eye examinations of DR worsening (n = 42) group at baseline and after AID initiation.
Discussion
To our knowledge, this is the first study that investigated DR progression after AID initiation in adults with type 1 diabetes. Prior studies have suggested a rapid decline in HbA1c as one of the major reasons for DR worsening.1-5,13-15 In addition, we found that LDL >100 mg/dL as one of the risk factors for DR worsening. Hyperlipidemia is a known risk factor for diabetic microvascular complications.16-19 Therefore, adequate treatment of lipids with the goal of LDL <70 mg/dL as recommended by the American Diabetes Association should be achieved in adults with type 1 diabetes 16 especially in the presence of microvascular complications. High HbA1c and high cholesterol are also acknowledged as risk factors for DR by the National Health Service (NHS). 20 The NHS established a 5-year AID implementation strategy for almost all people with type 1 diabetes to improve their glycemic control, quality of life, and reduce mental burden. 21 However, there is no guidance on monitoring retinopathy after initiating AID. Based on our research, more frequent DR screening and monitoring should be considered for those who may be at high risk for proliferative retinopathy.
The HbA1c reduction in our cohort was similar to what was reported in randomized controlled trials suggesting AID system have a great potential to improve glycemic control in the real world in type 1 diabetes.6-9 Although people with higher baseline HbA1c may have worsening of DR after initiation of AID, we expect to have overall better glycemic control and reduction in long-term microvascular complications with AID use as reported in prior studies.1,3,5,13 Therefore, high-risk individuals for DR worsening should not be discouraged for AID system. As there is no guidance on the frequency of eye exam after AID initiation, our study calls for adding frequent eye examination after AID initiation as a part of the American Diabetes Association (ADA) standards of care.
Large sample size and 2.7-year follow-up of AID use are major strengths of this study. Retrospective design and relatively infrequent eye examinations are major limitations. Majority of the patients who started AID therapy had to be excluded from the analysis for not having retinal exam before and/or after AID initiation (Supplementary Figure 1). Pandemic may play a role in missing retinal exams. Most of our patients were non-Hispanic White; therefore, these findings may have limited generalizability for other race and ethnicities. Moreover, majority of retinopathy worsening were mild (from no retinopathy to mild non-proliferative changes) and may not require significant changes in treatment. However, we also saw a modest increase in progression from mild non-proliferative retinopathy to proliferative retinopathy, which would require treatment to prevent progression and vision loss. Owing to the retrospective nature of the study and variable HbA1c measurement intervals, the rate of decline in HbA1c could not be estimated.
Conclusions
In conclusion, higher baseline HbA1c and LDL-C >100 mg/dL are associated with a three-fold increased risk of worsening DR after initiation of AID systems in type 1 diabetes. Patients with a higher risk for DR worsening should be screened for DR before and frequently after AID initiation. Future prospective studies with longer follow-up duration are needed to evaluate factors affecting DR progression after initiation of the AID system in people with type 1 diabetes.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251318740 – Supplemental material for Progression of Diabetic Retinopathy After Initiation of Automated Insulin Delivery System in Adults With Type 1 Diabetes
Supplemental material, sj-docx-1-dst-10.1177_19322968251318740 for Progression of Diabetic Retinopathy After Initiation of Automated Insulin Delivery System in Adults With Type 1 Diabetes by Kagan E. Karakus, Halis K. Akturk, Janet K. Snell-Bergeon and Viral N. Shah in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
AID, automated insulin delivery; BMI, body mass index; DCCT, Diabetes Control and Complications Trial; DR, diabetic retinopathy; ETDRS, Early Treatment of Diabetic Retinopathy Study; eGFR, estimated glomerular filtration rate; LDL-C, low-density lipoprotein–cholesterol; NPDR, non-proliferative diabetic retinopathy; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.E.K and J.K.S.-B. have no conflict of interest. H.K.A. received through University of Colorado research support from Dexcom, Medtronic, Tandem Diabetes, Eli Lilly, Senseonics, IM Therapeutics, REMD Biotherapeutics, and IAFMS and honoraria from Mannkind and Senseonics for advisory board attendance. V.N.S.’ employer, Indiana University, has received research support from NovoNordisk, Dexcom, Enable Bioscience, and Zucara Therapeutics. V.N.S. has also received honoraria from Dexcom, Insulet Tandem Diabetes Care, Sequel Medtech, Sanofi, NovoNordisk, Lilly, Embecta, Genomelink, and Lumosfit for consulting, speaking, or advising.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
