Abstract
Background:
In continuous glucose monitoring (CGM) system performance studies, it is common to implement specific procedures for manipulating the participants’ blood glucose (BG) levels during the collection of comparator BG measurements. Recently, such a procedure was proposed by a group of experts, and this study assessed its ability to produce combinations of BG levels and rates of change (RoCs) with certain characteristics.
Methods:
During three separate in-clinic sessions conducted over 15 days, capillary BG measurements were carried out every 15 minutes for 7 hours. Simultaneously, the participants’ BG levels were manipulated by controlling food intake and insulin administration to induce transient hyperglycemia and hypoglycemia. Subsequently, the combinations of BG levels and RoCs were categorized into dynamic glucose regions distinguishing between rapidly increasing BG levels (Alert high), hyperglycemia (BG high), rapidly falling BG levels (Alert low), and hypoglycemia (BG low).
Results:
A total of 24 adult participants with type 1 diabetes were included. Capillary BG-RoC combinations showed 7.5% in the Alert high region, 13.3% in the BG high region, 9.8% in the Alert low region, and 11.0% in the BG low region. No adverse events related to the glucose manipulation procedure were documented.
Conclusions:
As recommended by the experts, the percentage of data points in regions was ≥7.5%, demonstrating the procedure’s feasibility. However, given that the recommendation for the alert high region was only barely achieved, we suggest optimizations to the procedure and definition of dynamic glucose regions to facilitate the procedures’ adoption in standardized CGM performance evaluations.
Introduction
The comparison of continuous glucose monitoring (CGM) performance results is challenging due to differing study procedures caused by a lack of comprehensive guidelines for the evaluation of CGM performance. This applies in particular to methods for the collection of comparator blood glucose (BG) measurements, which are used to determine CGM accuracy.1,2
To collect comparator data across a large range of concentrations, it is common to perform dedicated in-clinic sessions where the BG levels of participants are deliberately manipulated. However, the goal of these manipulation procedures and, therefore, the targeted distribution of comparator data in terms of BG levels and rates of change (RoC) is often not clearly defined or described in the study reports, despite its impact on the observed CGM performance. From the limited information available, there appear to be procedures that target either high or low BG levels per session.3-8 Another approach aims to produce high and low as well as rapidly increasing and decreasing BG levels within the span of approximately 8 hours.9-20
Recently, a group of experts, which included the investigators of the present study, proposed concise requirements for the distribution of comparator data to be collected during standardized CGM performance studies. These requirements were specifically developed for studies with adults that examine non-adjunctive CGM systems to be used by patients with insulin-dependent diabetes mellitus. Performance studies with CGM systems for other uses or with different participant populations (eg, minors, pregnant persons) may necessitate different study procedures. 21
To define requirements for the distribution of comparator data, the article introduced the concept of dynamic glucose regions (DGRs) which encompass certain BG-RoC combinations in which the accuracy of CGM systems is of particular importance and should, therefore, be sufficiently reproduced during performance testing (Table 1). The “BG low” and “BG high” regions account for the necessity to test a CGM system throughout its measuring range, whereas the “Alert low” and “Alert high” regions account for situations with rapid increase and decrease of BG levels toward hyperglycemia and hypoglycemia, respectively. Together with the DGRs, the publication also suggested a testing protocol for manipulating BG levels to fulfill the DGR requirements. This testing protocol aims to induce transient hyperglycemia and hypoglycemia followed by stable BG levels in the normoglycemic range within 6 to 8 hours, similar to the studies mentioned above. The alternative approach to target either high or low BG levels within the same period would likely not be feasible to generate sufficiently high RoCs to fill the Alert high and Alert low regions.
Definition of the Dynamic Glucose Regions and Recommended Percentages of Rate of Change (RoC)—Blood Glucose (BG) Data Points in Each Region. 21
To examine the feasibility of the published procedure 21 in terms of participant safety and its ability to produce the desired comparator data characteristics, a dedicated CGM performance study was designed and conducted. To our knowledge, no study with a similar goal of examining a glucose manipulation procedure for CGM performance studies has been reported in the scientific literature. The performance of the tested CGM systems is reported in a separate article. 22
Methods
The study was conducted between April and July 2024 as a prospective, interventional study in adult participants by the Institute for Diabetes Technology Ulm, Germany. Only participants with type 1 diabetes were included. A full list of inclusion and exclusion criteria is provided in the Supplemental Materials. The responsible ethics committee granted ethical approval, and the study was notified to the competent authority and registered with the German Clinical Trials Register (DRKS00033697).
Study Devices
As there is no consensus on the comparator measurement approach to be used in CGM performance studies, ie, which device and sample origin, comparator BG concentrations were measured using three different approaches. Capillary BG level measurements were obtained with the handheld Contour Next BG monitoring system (Ascensia Diabetes Care Holdings AG, Basel, Switzerland). All test strips originated from the same manufacturing lot, and each test strip vial was checked using the manufacturer-provided control solution. In addition, venous comparator measurements were performed with the YSI 2300 STAT PLUS laboratory analyzer (YSI Inc, Yellow Springs, Ohio) utilizing a glucose-oxidase-based method and the COBAS INTEGRA 400 plus Analyzer (Roche Diagnostics GmbH, Mannheim, Germany) using a hexokinase-based method. Participants additionally wore sensors of three CGM systems.
Study Procedures
The study duration was 15 calendar days, most of which were spent under free-living conditions. The exception was three in-clinic sessions with frequent sampling periods (FSP) carried out on study days 2, 5 and 15. The FSPs had a duration of 7 hours (08:00 to 15:00), during which capillary measurements and venous blood draws were scheduled every 15 minutes. Capillary measurements were obtained in duplicate (separate test strips with blood from the same finger-prick), and a third measurement was taken if the duplicate’s absolute and relative (to the first measurement) difference was greater than ±10 mg/dL and ±10%, respectively. Venous access was established by placing a catheter in the forearms, and venous BG measurements were also carried out in duplicate with both laboratory analyzers in separated plasma. Additional capillary measurements were also performed outside the FSPs throughout the study period.
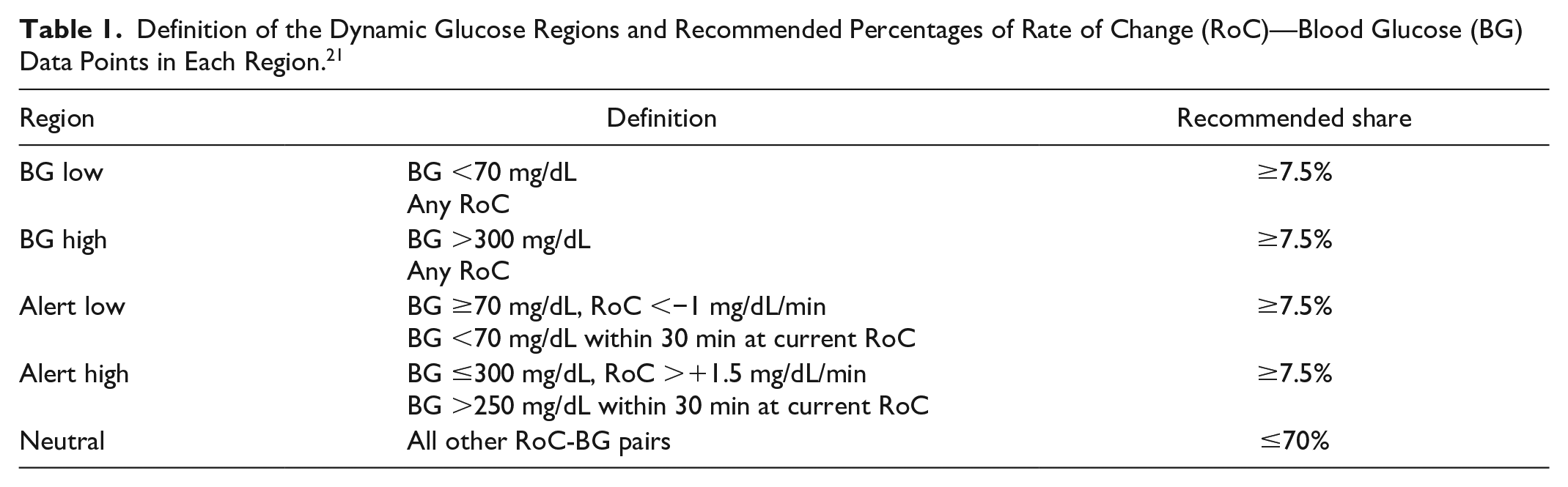
During the FSPs, the participants underwent the proposed glucose manipulation procedure. This procedure aimed to produce a comparator data distribution that complies with the suggested minimum percentage of 7.5% in each critical DGR. For that, the procedure aimed to approximate the capillary BG level profile shown in Figure 1 for every participant during every FSP.
Panel a: Profile of capillary blood glucose (BG) concentrations targeted during the frequent sampling periods (FSP) to adequately fill the dynamic glucose regions (DGRs). Panel b: DGR plot showing the associated profile of combined rate of change (RoC) and BG. The gray arrows indicate the direction of the curve with respect to its evolution over time. The colored background shows the different DGRs defined in Table 1 (Adapted from Eichenlaub et al 21 published under CC BY 4.0 license).
While undergoing the glucose manipulation procedure, participants were under constant supervision of study personnel. Furthermore, all interventions regarding food intake, insulin administration, and physical activity were supervised by the investigators (physicians experienced in diabetes management) to ensure participant safety while following the study protocol. Owing to the real-time availability of results, capillary measurements guided all interventions, and if deemed necessary by the investigators, additional measurements outside the 15-minute intervals could be carried out. Venous BG measurements were not available in real-time. Automated insulin delivery (AID) systems were switched to manual mode, ie, no automated adaptation of insulin delivery, during the FSPs, and the participants could consume calorie-free beverages, including coffee and tea ad libitum.
Before each FSP, participants were instructed to arrive fasted if possible. After the first measurement at 08:00, a meal containing approximately 25% of the participants’ daily caloric demand consisting of 65% carbohydrates (CHO, both slow and fast absorbed), 20% fat and 15% protein was consumed at 08:15. The meal consisted of typical continental breakfast ingredients such as bread, cheese, jam, fruit yogurt, and apple. At the investigators’ discretion and under consideration of the starting BG level, individual components of the meal could be omitted, or orange juice could be added.
The bolus of short-acting insulin, which would usually be delivered around mealtime, was withheld until capillary BG levels had reached values of around 250 mg/dL. The size of this bolus was calculated based on the BG level before meal consumption, the CHO content of the breakfast, a target BG level of 100 mg/dL and the CHO-to-insulin ratio and correction factors provided by the participants. However, adaptations to the insulin bolus size and timing could be made at the investigators’ discretion. Similarly, additional CHOs or insulin boluses could be ordered if BG levels did not increase above 300 mg/dL or remained high for too long, respectively.
If BG levels decreased only slowly after reaching their peak, thereby decreasing the probability of achieving sufficient values in the Alert low and BG low DGRs, participants could be asked to administer small additional insulin boluses or perform mild exercise. Fast-decreasing BG levels and hypoglycemia were managed to approximate the target profile by consuming fast-acting CHOs (fruit gums, dextrose tablets), which could be administered in individual doses as small as 0.8 g. However, if at any time desired by the participant or deemed necessary by the investigators, the hypoglycemia could be resolved immediately by consuming at least 15 g of fast-acting CHOs. As an additional safety precaution, glucose solution was available for administration through the venous catheters, and glucagon emergency kits for intramuscular injection were kept nearby.
At approximately 14:00, a second meal with low carbohydrate content (<15% of calories) was served, and participants were asked not to administer any insulin to avoid insulin stacking. At the end of the FSP, AID systems, if available, were switched to auto-mode and another BG measurement was taken to ensure a safe departure. In addition, CHO-containing snacks were offered, and participants received post-intervention instructions prompting them, eg, to closely monitor BG levels before driving and keep fast-acting CHOs nearby.
Data Analysis
All interventions during the FSPs (food intake, insulin administration, and exercise) were documented. Duplicate capillary measurements were excluded if no measurement pair with a difference smaller than ±10 mg/dL and ±10% could be found or if the measurements were obtained outside the 15-minute schedule. Venous measurements were excluded if the duplicate coefficient of variation (CV) was >5%.
The BG-level RoCs were calculated from two consecutive measurements with a time difference of ≤20 minutes and ≥10 minutes and paired with the latter of the two measurements to form the BG-RoC pairs. These pairs were then assigned to the DGRs as defined in Table 1, and the overall percentage of pairs in each region was calculated. In addition, the lower (upper for the “Neutral” region) 95% confidence interval (CI) limits of these percentages were determined using the Clopper-Pearson approach. Furthermore, the mean absolute RoC (MARoC), an indicator of the overall speed of BG level changes, was calculated.
Summary statistics (median and interquartile range) of the BG level profiles were plotted. In addition, the shape of individual FSP BG level profiles was characterized by plotting the distribution of magnitude and timing of peak and nadir. Furthermore, the clinical decision-making during the glucose manipulation procedure was characterized by analyzing the BG levels at which the first insulin bolus was administered, the time delay between breakfast consumption and the first insulin bolus, the BG level at which the first intervention CHOs were consumed, and the total amount of intervention CHOs consumed between the BG level peak and lunch.
Safety Analysis
Participants were closely monitored during the time spent at the investigational site and were asked to report any adverse events (AEs) throughout the study period. The AEs were documented and classified as serious or non-serious. In addition, their intensity was rated by the investigators as mild, moderate, or severe, and they were categorized as related or unrelated to the study procedures and/or the CGM systems, respectively. Here, it should be emphasized that transient hypoglycemic and hyperglycemic episodes during the FSPs are an expected side effect of any glucose manipulation procedure targeting low and high BG levels and were therefore not considered as AE.
Results
A total of 24 adult participants with type 1 diabetes were included and completed the study. Their baseline characteristics are shown in Table 2.
Baseline Characteristics of the Participant Population.
Abbreviations: MDI, multiple daily injections; CSII, continuous subcutaneous insulin infusion; AID, automated insulin delivery; CHO, carbohydrates.
Results given as mean (minimum, maximum).
Included in CSII subset.
As capillary measurements guided the glucose manipulation procedure, their results are predominately shown and discussed. The corresponding results for venous measurements are provided in the Supplemental Materials. Of the 2088 measurements planned during the 72 FSPs, 2053 capillary measurements from 71 FSPs were included in the data analysis. Data from one participant’s FSP were excluded because a kink in the subcutaneous insulin infusion catheter was noticed after the FSP began. After the catheter was replaced, no further measures to manipulate BG levels were carried out, and the participants’ BG levels were only monitored to ensure a safe release. After calculating the RoCs, 1976 BG-RoC pairs were available.
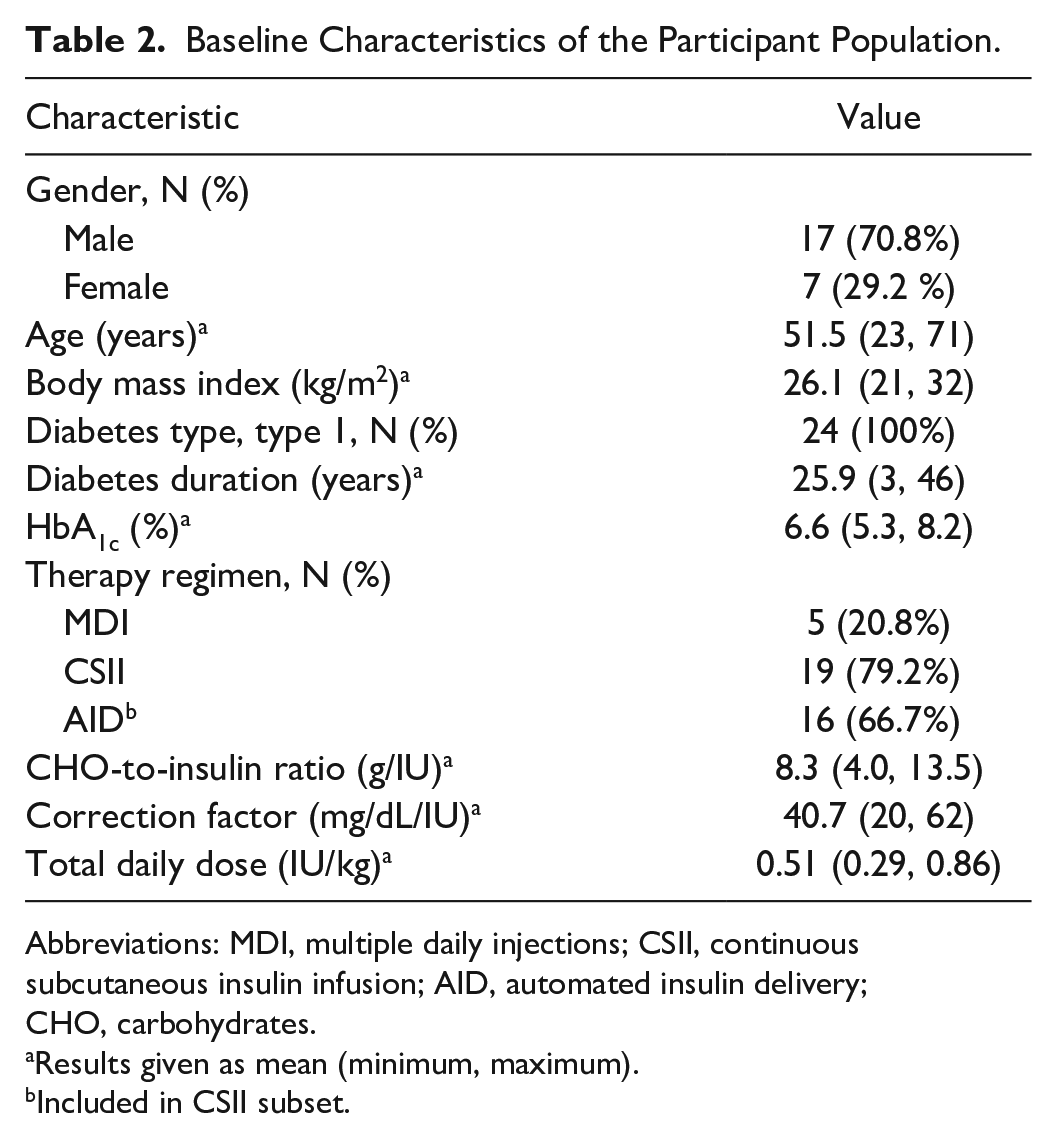
The median profile of capillary measurements and scattering of BG-RoC pairs across the DGRs are displayed in Figure 2, indicating that the target glucose profile could be approximated on average. The distribution according to the DGRs showed percentages ≥7.5% in the critical regions (Table 3). The corresponding information from the venous BG measurements is shown in the Supplemental Materials.
Panel a: Median profile of capillary blood glucose (BG) level measurements during the frequent sampling periods. The error bars indicate the interquartile range. Panel b: Dynamic glucose region plot.
Distribution of Capillary BG-RoC Pairs With Respect to the Dynamic Glucose Regions.
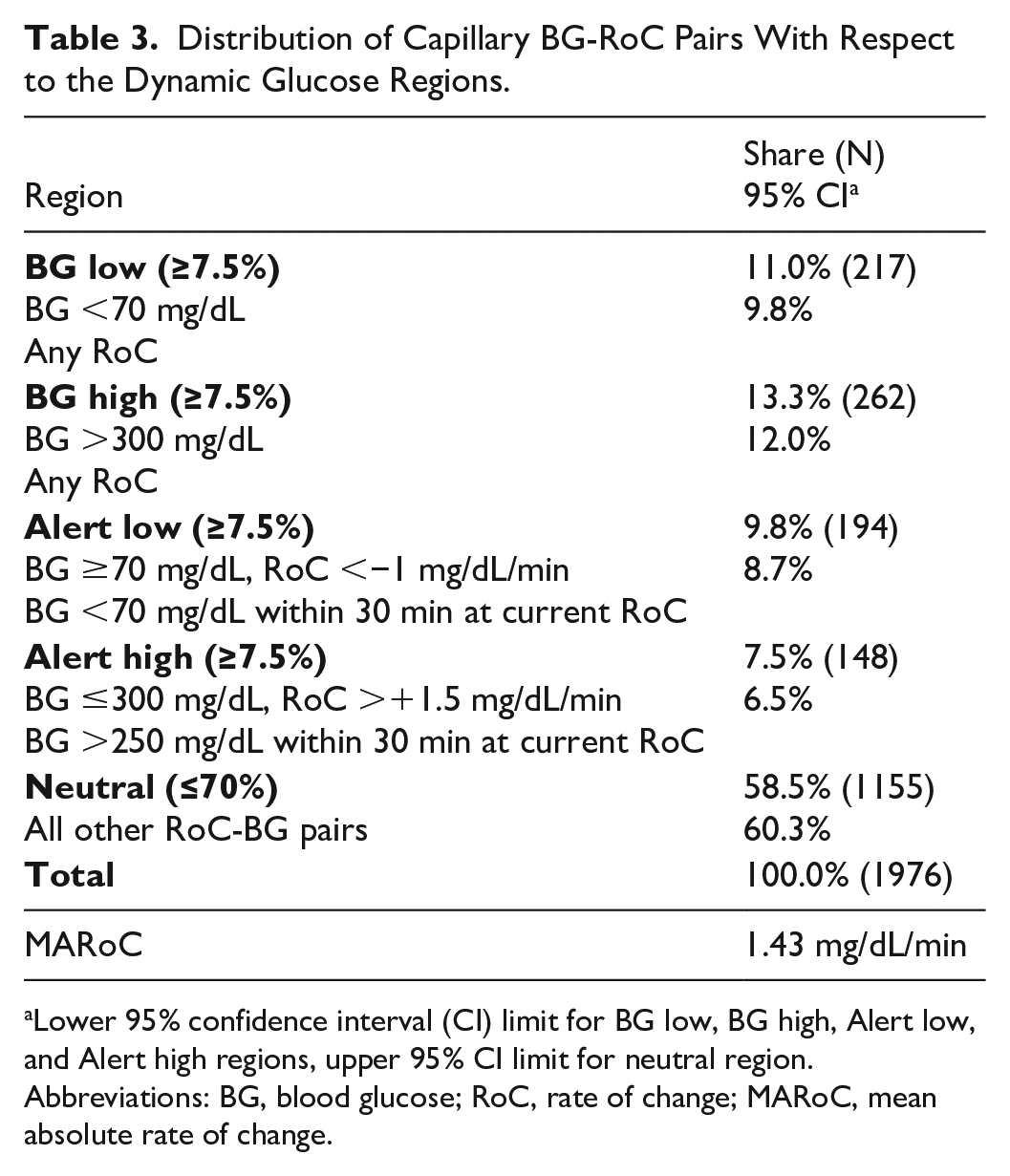
Lower 95% confidence interval (CI) limit for BG low, BG high, Alert low, and Alert high regions, upper 95% CI limit for neutral region.
Abbreviations: BG, blood glucose; RoC, rate of change; MARoC, mean absolute rate of change.
The characteristics of the individual capillary BG level profiles are given in Figure 3, showing the variability in individual profiles. Figure 4 characterizes different aspects of the clinical decision-making process during the glucose manipulation procedure. In 18 of 71 (25.4%) FSPs, the planned CHO content of the breakfast was altered (16 reductions, 2 increases in CHO content). Furthermore, in 27 of 71 (38.0%) FSPs, the insulin bolus calculated based on the participant’s therapy parameters was altered by more than 20% (18 reductions, 9 increases in bolus size).
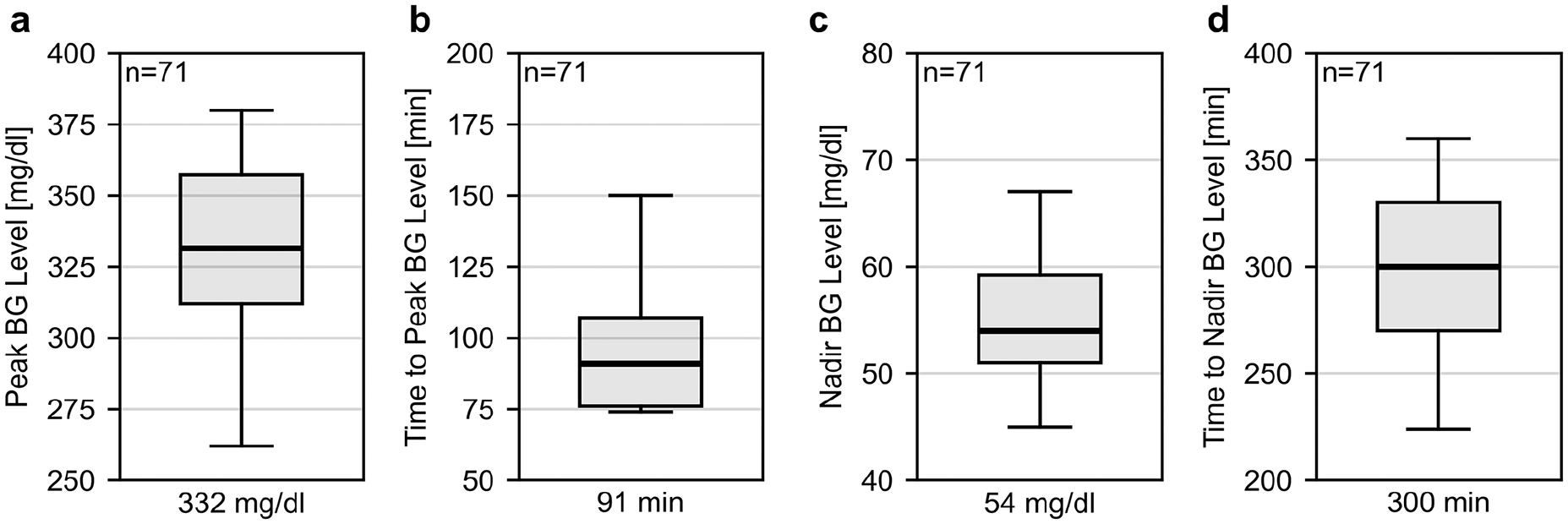
Distribution of capillary BG level peak (panel a) time to BG level peak from the start of the frequent sampling period (FSP, 15 minutes before breakfast consumption) (panel b), BG level nadir (panel c), and time to nadir from the start of the FSP (panel d) of individual glucose profiles, respectively. The values below the figures give the median. The boxplots indicate median (line), interquartile range (box) and 90% range (antennae).
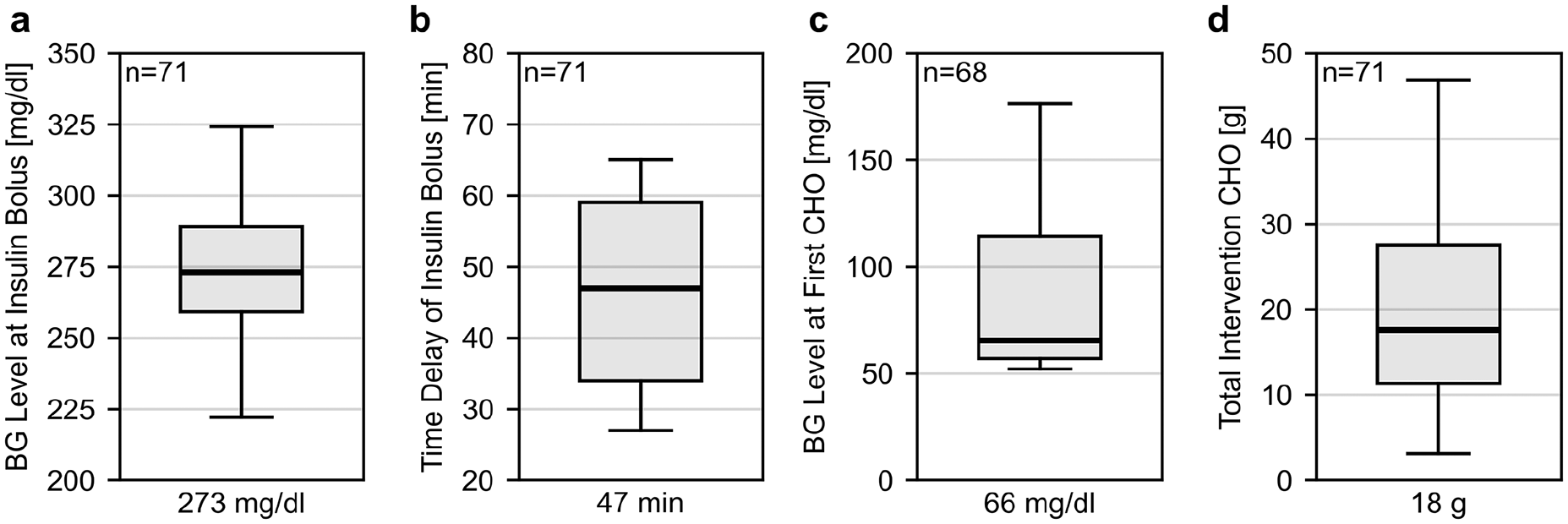
Clinical decision-making during the glucose manipulation procedure in terms of the capillary BG level at which the first insulin bolus was administered (panel a), the time delay between breakfast consumption and the first insulin bolus (panel b), the BG level at which the first intervention CHOs were consumed (panel c), and the total amount of intervention CHOs consumed between the BG level peak and the end of the FSP (panel d). The values below the figures give the median. The boxplots indicate median (line), interquartile range (box), and 90% range (antennae).
A total of 16 AEs from 12 participants were documented, all classified as non-serious and resolved within the study’s follow-up period. Of these 16 AEs, 4 were neither related to the study procedures nor the CGM systems, and 11 were related to the CGM systems but not the procedures. Only one AE related to the study procedures was observed and involved a hematoma caused by the catheter for venous sampling. No AEs associated with the manipulation of glucose levels were documented. In particular, no invasive interventions to treat hypoglycemia or hyperglycemia, such as intravenous administration of glucose or insulin or the intramuscular injection of glucagon, were required during the study.
Discussion
This study evaluated the ability of a recently published glucose manipulation procedure 21 to safely produce a distribution of BG-RoC pairs with specific characteristics to be used as comparator data in CGM performance studies. As recommended, the percentage of capillary data points in each critical region was ≥7.5% (Table 3). However, although the percentages in the BG low, BG high, and Alert low regions, particularly their lower CI limits, were clearly above 7.5%, this was not the case for the Alert high region. As shown in Figures 2a and b, this was caused by BG levels rising too fast above 300 mg/dL compared to the 15-minute sampling interval, so the Alert high region was traversed without capturing a sufficient number of measurements. These fast-rising glucose levels also made it challenging to find the optimal timepoint for the meal bolus (Figure 4b), which led to a large variability in BG levels at which this bolus was administered (Figure 4a).
Another challenge was the management of hypoglycemia due to the goal of producing several Alert low data points, indicating moderately fast decreasing BG levels, directly followed by approximately 30 minutes of hypoglycemia to generate a sufficient number of data points in the BG low region. This required close supervision and experience from the investigators to select the timing and amount of intervention CHOs (Figure 4c and d) to ensure participant safety while avoiding to resolve the hypoglycemia too quickly. Here, it should be pointed out that, in more than 25% of FSPs, the first intervention of fast-acting CHOs was made at BG levels above 100 mg/dL to reduce the rate at which BG levels were decreasing (Figure 4c). In addition, the size of the initial meal bolus was decreased in several FSPs in comparison to the calculated dose to reduce the risk of BG levels decreasing too rapidly. This was also important as the therapy parameters reported by the participants were not always reliable. Overall, the results shown in Figure 4 and the fact that the planned breakfast CHO content and insulin dose were adapted in several cases highlight the need for individualized and participant-specific clinical decision-making when implementing this procedure.
Despite the challenges in the management of decreasing BG levels and hypoglycemia, the requirements for the alert low and BG low regions were fulfilled, while no AEs related to the glucose manipulation procedure were documented. However, we should note that, as expected, some participants reported symptoms of hypoglycemia, which could be resolved through oral intake of CHOs. In particular, we recorded 42 (2.0%) capillary comparator measurements <54 mg/dL, occurring in 42.3% of FSPs. This is, however, not uncommon in CGM performance studies, as shown by results published by the Food and Drug Administration (FDA) for the approval of well-established CGM systems.23-26 Here, the percentage of comparator measurements <54 mg/dL ranged from 1.9% to 2.5%. Furthermore, the percentage of comparator data >400 mg/dL was similar in this study compared to the data published by the FDA (Table 2 in the Supplemental Materials). In contrast, the MARoCs in these studies ranged between 0.8 and 0.9 mg/dL/min (Table 2 in the Supplemental Materials), which is considerably lower than the 1.4 mg/dL/min found in this study. This indicates that a different glucose manipulation procedure with slower BG level changes was used. In addition, there were 20 capillary measurements <54 mg/dL outside the FSPs. We therefore conclude that the examined procedure, particularly its goal to produce high RoCs, is not associated with an increased risk for severe hypoglycemia or hyperglycemia compared with other CGM performance studies and does not pose an undue risk compared to the participants’ daily lives.
Finally, the distribution of venous comparator data, unavailable during glucose manipulation, should be discussed. For the YSI laboratory analyzer, there were an insufficient number of data points in the BG high region (6.7%, Table 1 in the Supplemental Material). This can be explained by the fact that the capillary data, lying on average approximately 10% above venous YSI data, were used to decide the time point of insulin dosing after the meal. Therefore, insulin doses were likely administered too early in order to achieve a sufficient number of venous YSI measurements >300 mg/dL. This reveals the difficulties in achieving any targets for the distribution of comparator data when BG levels are managed with a measurement approach that is substantially biased against another approach used to obtain comparator measurements. Ideally, the two measurement approaches should be identical or aligned.
Conclusion and Recommendations for Future Studies
For the first time, this article reported and evaluated the outcomes of a glucose manipulation procedure to be utilized in CGM performance studies in detail. Although the procedure as implemented in the study was feasible, it has some limitations that might hinder its adoption when recommended in a future standard for CGM performance evaluations. First, the percentage of data points in the Alert high region was 7.5%, therefore just reaching the recommended minimum percentage of 7.5%. Consequently, we suggest adapting the definition of the Alert high region and/or the minimum recommended percentage to facilitate compliance. However, we do not recommend adapting the sampling frequency. 21
Second, the management of decreasing BG levels and hypoglycemia was carried out without formulating specific rules for administering intervention CHOs. This was made possible by the experience of the investigators at the Institute for Diabetes Technology, who have been conducting studies with similar procedures for more than 15 years. However, to facilitate the implementation of this procedure at less experienced study centers and reduce the complexity of decision-making, we recommend the safety precautions listed in Table 4, which should decrease the risk of an AE, particularly severe hypoglycemia. Note that this is not an exhaustive list, and standard safety measures must be implemented when conducting research with human participants. In addition, we suggest an adaptation of the definition of the Alert low region to include higher BG levels. This would allow for earlier CHO intervention, thus further reducing the risk of severe hypoglycemia.
Recommended Procedure-Specific Safety Precautions for Future Studies Implementing the Tested Glucose Manipulation Procedure.

Abbreviations: BG, blood glucose; RoC, rate of change; CHO: carbohydrate.
Although not required in this study, we should emphasize that there is the opportunity to carry out alternative procedures in a subset of participants or FSPs that target specific critical DGRs in case the presented procedure with the recommended safety precautions fails to yield the desired distribution. For example, if BG levels are raised more slowly, it is possible to produce more Alert high and BG high data points. Another option for specifically producing BG low data points is to reduce BG levels from fasting conditions slowly. Finally, if the distribution of comparator data fails to comply with recommendations, the option to systematically exclude data from the Neutral region is still available. 21
In summary, we can conclude that the examined procedure for manipulating glucose levels is feasible and safe. However, we suggest optimizations to the procedure and definition of DGRs to facilitate the procedures’ adoption in standardized CGM performance evaluations.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251317526 – Supplemental material for Feasibility of a Glucose Manipulation Procedure for the Standardized Performance Evaluation of Continuous Glucose Monitoring Systems
Supplemental material, sj-docx-1-dst-10.1177_19322968251317526 for Feasibility of a Glucose Manipulation Procedure for the Standardized Performance Evaluation of Continuous Glucose Monitoring Systems by Manuela Link, Manuel Eichenlaub, Delia Waldenmaier, Stephanie Wehrstedt, Stefan Pleus, Nina Jendrike, Sükrü Öter, Cornelia Haug, Stefanie Hossmann, Martina Rothenbühler, Derek Brandt and Guido Freckmann in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors thank all volunteers for their participation in the study, the staff at the Institute for Diabetes Technology and the funders of this study for their support.
Abbreviations
AE, adverse event; AID, automated insulin delivery; BG, blood glucose; CGM, continuous glucose monitoring; CHO, carbohydrates; DGR, dynamic glucose region; FSP, frequent sampling period; MARoC, mean absolute rate of change; RoC, (glucose) rate of change.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.F. is the general manager and medical director of the Institute for Diabetes Technology (Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany), which carries out clinical studies, eg, with medical devices for diabetes therapy on its own initiative and on behalf of various companies. G.F./IfDT have received research support, speakers’ honoraria, or consulting fees in the last three years from Abbott, Ascensia, Berlin Chemie, Boydsense, Dexcom, Lilly Deutschland, Novo Nordisk, Perfood, Pharmasens, Roche, Sinocare, Terumo, and Ypsomed. ML, ME, DW, SW, SP, NJ, SÖ, and CH are the employees of IfDT. S.H., M.R. and D.B. have no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support to partially cover the costs of this study was provided by BIONIME Corporation, Diabetes Center Berne, i-SENS, Inc, and Roche Diabetes Care GmbH. In addition, Ascensia Diabetes Care Holdings AG provided blood glucose monitoring systems and associated consumables free of charge. None of the commercial entities had any influence on the study design, data analysis, presentation, or publication of results. The remaining costs were carried by the Institute for Diabetes Technology. No funding was provided by any of the manufacturers of the examined CGM systems.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
