Abstract
Introduction:
This article presents a cost-effective, modular infusion platform to help diabetes specialists customize and understand infusion pump mechanics and control principles. Traditional insulin pumps are costly and inflexible, limiting accessibility, and particularly in low-resource settings. Inspired by open-source initiatives like OpenAPS, this platform engages specialists in device operation and customization, offering practical insights into infusion technology.
Method:
An initial survey assessed technological literacy, customization interests, and feature preferences among Mexican diabetes specialists, followed by a hands-on engagement session with the platform’s hardware. Core components are described and chosen for reliability, affordability, and integration ease. A follow-up survey evaluated specialists’ confidence and interest in device customization, gathering feedback on usability and design.
Results:
Survey data showed strong specialist interest in understanding device mechanics and high confidence in customization after hands-on engagement. Most specialists found the hardware layout conducive to experimentation, with significant interest in closed-loop capabilities. Key valued features included safety, affordability, ease of use, customization, and integration of diverse continuous glucose monitors, with added suggestions for potential clinical certification, cost-effective supplies, and artificial intelligence integration.
Conclusion:
This platform offers a promising educational and developmental tool in diabetes management, bridging clinical application, and customization. Its low-cost, modular design provides a feasible solution for low-resource settings, equipping specialists to tailor devices for specific patient needs. While the platform’s educational potential is clear, further studies and validation are essential for a possible transition to a clinical-grade device. Continued development could democratize access to advanced diabetes technology, transforming specialist training, and patient care.
Keywords
Introduction
Long-term diseases requiring continuous medication pose significant challenges in ensuring accurate and consistent drug delivery. Advanced infusion technologies enable precise, adaptable dosing, minimizing complications, and improving treatment efficacy. 1 In diabetes management, continuous insulin infusion is critical for maintaining adequate glycemic control.2,3 Since their introduction in the 1970s, insulin pumps have become increasingly precise and portable. 4 However, their high cost and proprietary designs restrict access, particularly in low- and middle-income countries.
To address this, the Do-It-Yourself (DIY) and open-source communities have developed low-cost, customizable infusion systems using readily available hardware and software.5,6 Initiatives like OpenAPS or AndroidAPS have demonstrated that effective diabetes management is possible with accessible, open-source algorithms, empowering users with greater control.7-9 On the hardware front, infusion systems have been successfully developed and tested to tackle cost limitations across various regions and use cases.10-13 However, these systems often lack structured platforms designed to actively engage specialists in the design, optimization, and customization of infusion technologies.
The integration of smart devices and continuous glucose monitors (CGMs) further enhances DIY systems. 14 Smartwatches can track physical activity and adjust insulin delivery in real-time, while smartphones provide intuitive interfaces for monitoring, alerts, and remote control by caregivers.8,15 In addition, cloud platforms aggregate user data to refine control algorithms, enabling personalized treatment strategies. 16 The integration of these devices is illustrated in Figure 1.
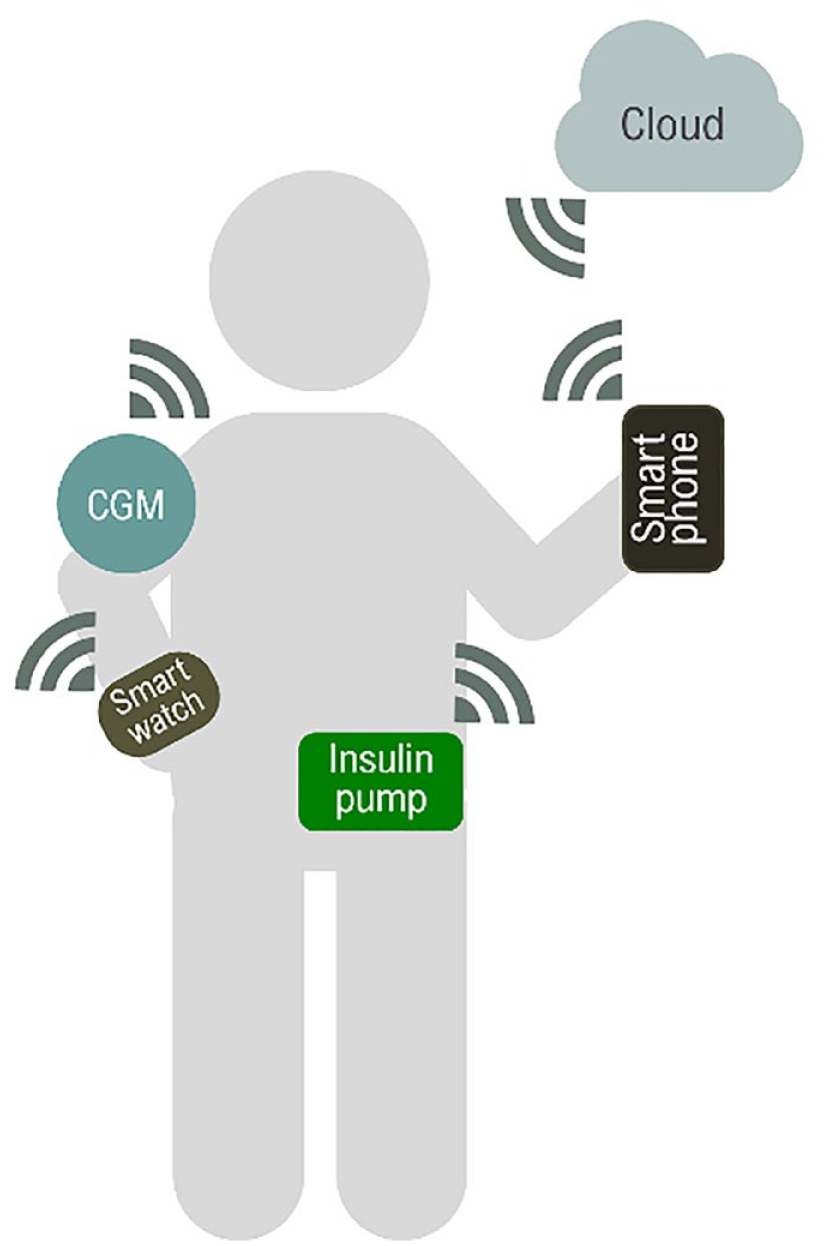
Integration of devices and platforms.
Control algorithms are central to infusion systems, determining both the timing and dosage of medication. These systems operate in open-loop (manual input), closed-loop (automatic adjustments using real-time data), or hybrid modes.17,18 Advanced techniques like proportional-integral-derivative (PID) control, fuzzy logic, and model predictive control (MPC) enable adaptive responses to physiological changes, enhancing safety and performance.19-22 Moreover, the emergence of cloud-based artificial intelligence (AI) and data analytics highlights a potential area of development.23-25
This study introduces a novel, modular platform that emphasizes affordability, modularity, and user-centered design, serving as a model for advancing diabetes management, particularly in low-resource settings. By offering a transparent, hands-on educational framework, the platform fosters a deeper understanding of infusion system mechanics and empowers specialists to create personalized, clinically relevant solutions.
The article details the methodology, starting with an initial survey to assess specialists’ technological literacy, interest in customization, and feature preferences. It then describes the design and integration of key hardware and firmware components that enable hands-on experimentation. A follow-up survey, conducted after specialists interacted with the platform, evaluates its potential for addressing device limitations and inspiring innovation. The results and discussion explore the findings, implications, and opportunities for future advancements, while the conclusion highlights the platform’s contributions to diabetes management and its broader impact on specialist training and patient care.
Method
Initial Survey
We conducted an initial survey to assess self-perceived technological literacy and interest in infusion pump principles among Mexican diabetes specialists. Administered in person at the Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán in Mexico City, the survey targeted 16 endocrinologists with hands-on experience using infusion pumps in their clinical practices. Participation was voluntary and anonymous. The survey questions, along with their rationale, are listed in Table 1, with Q1 to Q4 evaluated on a Likert scale and Q5 as an open-ended question.
Initial Survey Questions.

Hardware Overview
Designing an infusion pump requires careful selection of components to ensure precise, reliable, and safe medication delivery. Key components include:
Microcontroller
Manages timing, dosage calculations, infusion, and intercomponent communication. It must be powerful enough to autonomously execute control algorithms, requiring a high clock speed for fast processing and sufficient memory for storing instructions and settings. Communication with peripherals is facilitated through direct general-purpose input/output (GPIO) connections or standardized protocols.
Infusion mechanism
Typically utilizes a stepper motor to control the movement of fluid from the reservoir to the patient. Precision in controlling flow rate and volume is critical for accurate dosing. A motor driver is required to manage the electrical signals that control the motor’s speed and direction.
Memory storage (EEPROM)
Retains critical data like settings and history even when powered off, preserving treatment consistency.
Real-time clock (RTC)
Tracks time for accurate medication scheduling, ensuring timely administration and accurate event timestamps for monitoring.
Current and position sensors
Detect potential blockages during delivery by monitoring increased current or verifying plunger movement.
User interface components
The printed circuit board (PCB) must include both visual and audible alerts to provide clear user feedback. Two basic LEDs indicate power status and infusion delivery, with the option for additional system-status LEDs if microcontroller pins are available. A buzzer offers audible alerts for key events, ensuring important notifications are received even if visual cues are missed. In addition, buttons and displays allow users to input commands and view real-time information.
Communication interfaces
The PCB features communication interfaces for external components and data exchange.
I2C and UART: Wired interfaces that connect the pump to displays and sensors.
BLE (Bluetooth Low Energy): Enables wireless communication with CGMs and smartphones, allowing real-time monitoring and data transfer. Smartphones can act as a bridge to send data to the cloud for analysis.
USB: Provides direct connection to computers for data transfer and programming updates.
Power source and voltage regulators
A battery provides continuous power, with voltage regulators ensuring stable voltage to protect components from fluctuations. The battery’s status is monitored by the microcontroller, enabling timely alerts for low power and ensuring uninterrupted operation.
Using a morphological chart, we systematically evaluated potential components based on criteria like functionality, availability, and cost-effectiveness. The final choices are summarized in Table 2.
Components Chosen for the Prototype.
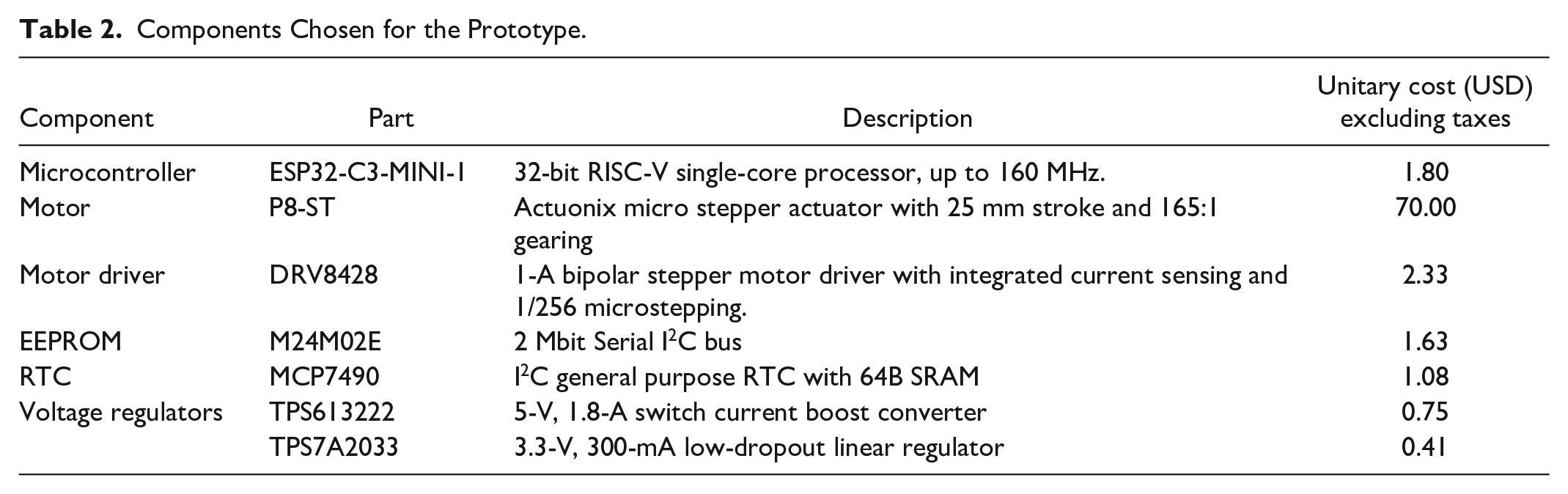
PCB Design and Assembly
The PCB design must adhere to IPC-2221 standard to ensure proper layout, electrical clearances, and thermal management. Moreover, it must comply with key medical device standards, including IEC 60601-1, IEC 60601-1-2, and ISO 14971. 26 Figure 2 shows the block diagram with the proposed integration.
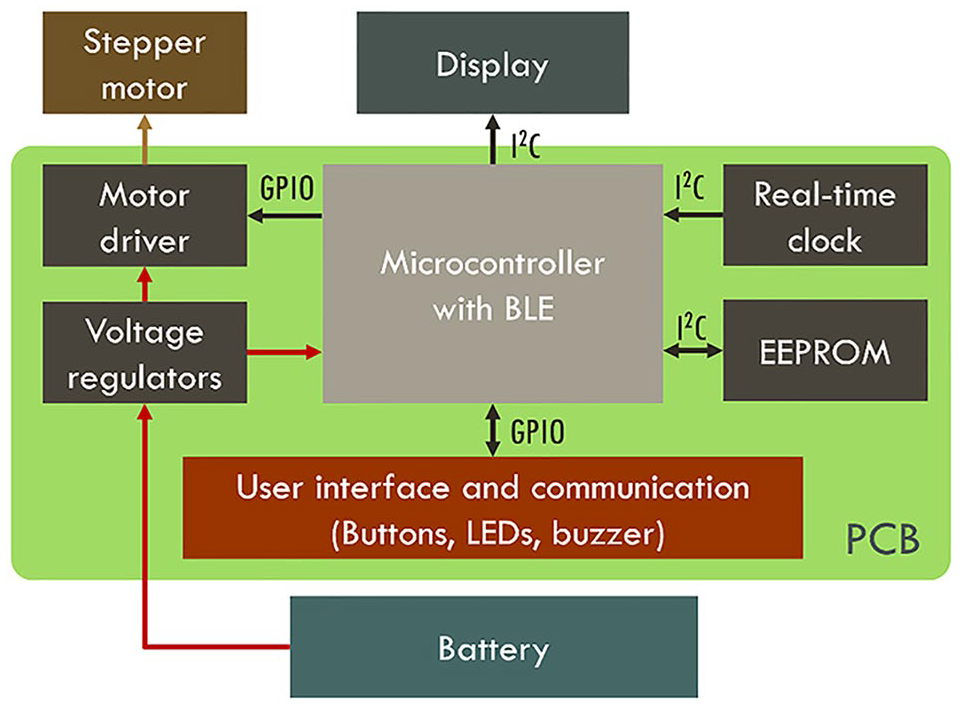
Prototype component block diagram.
Firmware Architecture
The firmware uses object-oriented programming (OOP) to modularize system functions, allowing users to easily modify components and algorithms without requiring extensive coding expertise. A main template handles core system functions, with predefined classes managing motor control, RTC and EEPROM operations, user interface elements, and BLE-based data communication. Specialists need to develop only two key libraries without altering the core system architecture:
CGM: For integrating their desired sensor if the library is not available yet.
Control: For implementing and testing different control algorithms.
Figure 3. Flow diagram of the main source code presents a flow diagram of the main source file, where customizable methods for infusion control are highlighted in green.
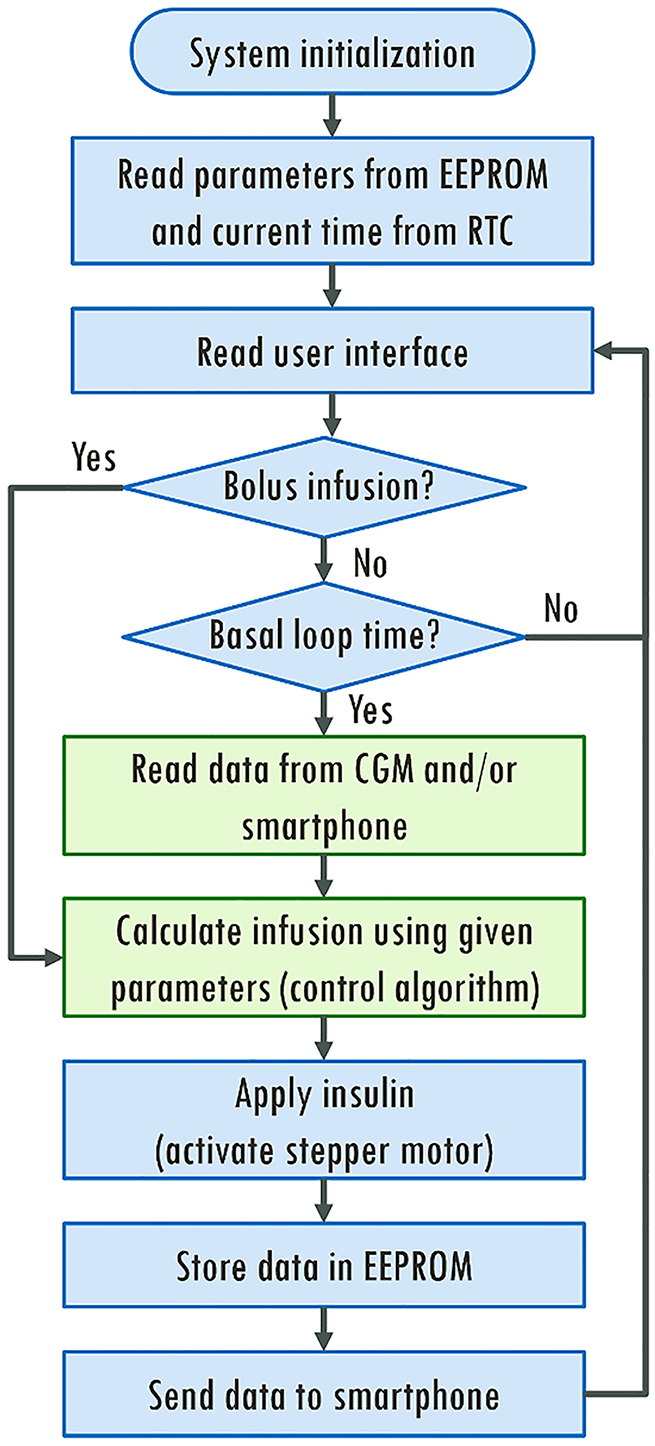
Flow diagram of the main source code.
Platform Overview and User Interaction
We presented an overview of the platform’s developmental framework, highlighting core hardware components, their layout, and how control algorithms regulate insulin delivery based on glucose data within the proposed firmware structure. Users interacted with the assembled PCB, evaluating its usability firsthand.
Postsurvey
Following the presentation, specialists participated in a postsurvey to capture their impressions of the platform’s educational and developmental potential. The survey questions, along with their rationale, are listed in Table 3, where Q1 to Q3 are evaluated on a Likert scale and Q4 is open ended.
Postsurvey Questions.
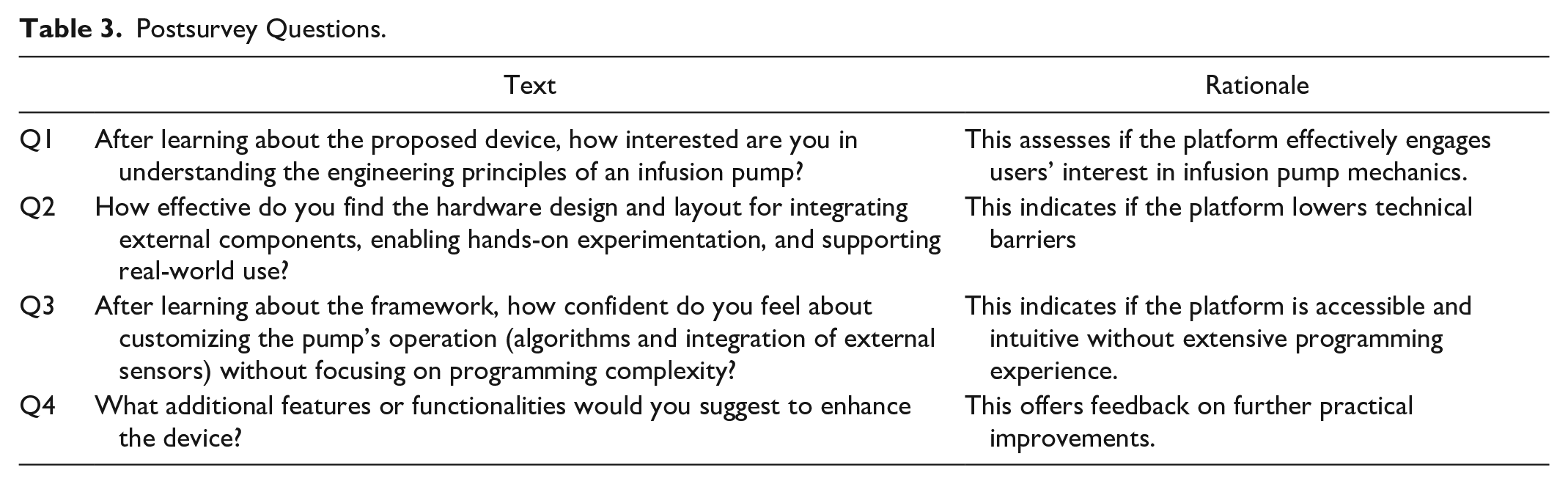
Results
Initial Survey
As shown in Figure 4, most specialists rated their technological literacy as moderate (38%) or high (38%), with only 6% indicating low literacy. This suggests that while they possess foundational knowledge, there is room for deeper technical engagement.
Initial survey results.
Interest in understanding core hardware components and control principles was substantial, with 63% rating this as high or very high. Programming and customizing control algorithms received a more varied response, with 44% showing moderate interest, 31% high, and 13% very high, indicating some enthusiasm for customization but also a need for accessible resources.
Integration with external devices, such as CGMs, was rated as highly important by most specialists, with 56% indicating very high interest and 44% high. This reflects a strong preference for a platform that supports interoperability and flexible adaptation to clinical needs.
In terms of desired basic features for a new insulin pump (Table 4), safety was the top priority, noted by all 16 specialists, followed by cost-effectiveness (87.5%), ease of use (68.75%), and customization (50%). Closed-loop functionality also gained significant interest, with 56.25% of specialists highlighting it as valuable. These findings underscore the need for a user-friendly, safe, affordable, and adaptable system that supports both basic functionality and advanced customization.
Most Valued Basic Features for a New Pump.

Hardware Integration
The modular PCB, shown in Figure 5, measures 64 × 25 mm, adheres to medical-grade compliance standards, and targets a final prototype size of around 80 × 35 × 25 mm. Compactness was achieved by using surface-mount components selected for their size efficiency and the availability of pin-to-pin compatible replacements, ensuring both space optimization and ease of substitution. Power-critical components like regulators, the microcontroller, and the motor driver were positioned close together for optimal performance. The motor driver, critical for infusion control, is activated via direct GPIO connections for precision, while components requiring I2C communication are clustered on the right side to streamline organization.

Prototype PCB.
The user interface looked for simplicity and functionality. The left button navigates menus, while the right confirms commands. Five LEDs, visible even during button operation, indicate power, motor activity, CGM pairing, data transfer, and operating mode. The layout can accommodate an optional LCD above the microcontroller without obstructing the LEDs.
Postsurvey
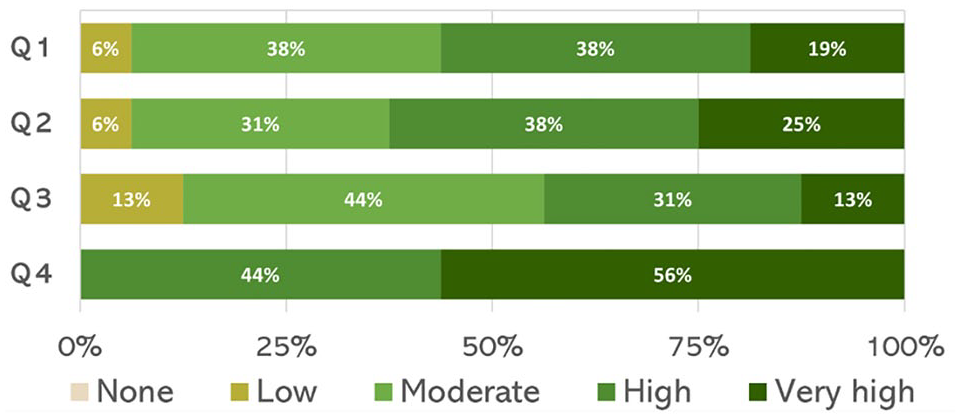
Figure 6 illustrates that 88% of specialists expressed high or very high interest in understanding the platform’s technical workings, with 50% indicating high interest and 38% very high.
Postsurvey results.
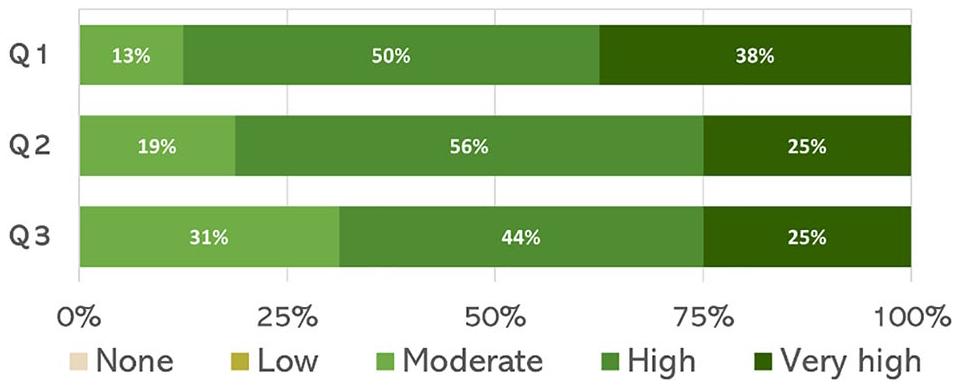
Building on this, Figure 7 highlights the change in specialists’ interest in understanding engineering principles before and after the engagement. Interest increased from 63% in the initial survey to 88% in the follow-up, with a statistical test confirming this improvement as significant
Evolution of specialists’ interest in engineering principles.
Confidence intervals were calculated to indicate the range within which the true percentage is likely to fall. For the initial survey, it spanned 39% to 87%, increasing to 72% to 100% in the follow-up.
As expected from the comments, specialists also rated the hardware design positively, with 81% viewing its effectiveness as high or very high. The modular layout and ease of external component integration were seen as conducive to hands-on experimentation and real-world application, highlighting its adaptability.
Confidence in customization grew as well, with 69% of specialists feeling high or very high confidence in modifying algorithms and integrating sensors without extensive programming skills. Although 31% rated their confidence as moderate, the platform clearly has the potential to reduce technical barriers to customization.
Specialists suggested several improvements to enhance the platform’s functionality and usability (Table 5). The most requested addition was certification for clinical use, noted by 56.25% of specialists, with one specifically requesting certification for use during pregnancy. Cost-effective supplies were also highlighted by 50% of specialists, reflecting an emphasis on affordability. Integration of AI was another priority, with 31.25% of specialists expressing interest in AI capabilities to support more advanced functionality. Other suggestions like waterproof design and an ergonomic case underscored the need for practical features that enhance usability and comfort.
Additional Features Suggested After Introducing the Platform.

Discussion
The development of this cost-effective, modular infusion platform marks a significant step forward in diabetes management by offering an accessible, customizable solution tailored for hands-on learning and innovation. Drawing inspiration from open-source initiatives like OpenAPS, the platform empowers specialists to actively contribute to its development. Survey feedback underscores its educational value by bridging the gap between technical knowledge and clinical application, fostering innovation. Table 6 provides a comparative overview, positioning this platform alongside existing initiatives and highlighting its distinctive contributions.
Comparison of This Platform With Other Initiatives.

Specialists expressed a strong interest in understanding the technical workings of infusion pumps, a topic often underexplored in clinical training. While these endocrinologists were familiar with pump operation, many lacked knowledge of components, like the microcontroller’s role in real-time dosing or the motor’s precision in flow regulation. After the hands-on session, 88% of participants expressed high or very high interest in learning and understanding more about these engineering principles, indicating the platform’s ability to transform passive users into engaged learners. By providing continued educational resources and workshops, the platform can further empower specialists to customize and troubleshoot devices, fostering clinician-led innovation.
The integration of CGMs through BLE and additional communication interfaces like USB was perceived as highly beneficial. These features enable closed-loop applications that autonomously leverage real-time CGM data while using smartphone or cloud-based processing capabilities to minimize hypo- and hyperglycemia risks. As specialists become proficient in customization, they can develop patient-specific workflows to overcome limitations in commercial devices, fostering clinician-led advancements.
Ease of use remains pivotal for adoption. Specialists noted that traditional pumps often overwhelm patients with limited technical skills. This platform’s intuitive interface—featuring menu-navigation buttons, clear status LEDs, and optional LCD integration—was recognized for its simplicity and adaptability. These design features make it accessible to both specialists and patients, promoting self-management and broader engagement with infusion technology.
User feedback highlighted areas for enhancement, including adding a graphical interface for real-time visualization of dosing adjustments and expanded data storage for long-term comparisons of control strategies. Training materials, such as guides and tutorials, were also suggested to ease the learning curve, further solidifying the platform’s value as an educational and research tool.
Affordability is another critical advantage, with the platform’s estimated material cost under $200 USD making it accessible in low-resource settings where commercial pumps exceed $4000 USD. This affordability also positions the platform for broader applications, such as managing long-term pain or oncology treatments. However, transitioning to clinical-grade functionality will require rigorous testing to ensure safety and reliability, indicating a dual role as both an educational and potential clinical tool.
The platform’s modularity and open design provide a scalable framework for advancing low-cost insulin pump initiatives worldwide. Leveraging cost-effective, widely available components and adhering to medical-grade standards, it is adaptable to resource-constrained settings. Developers can customize its modular PCB design to accommodate alternative hardware configurations and integrate region-specific glucose-monitoring technologies through its open firmware.
In addition, the platform fosters global collaboration among engineers, medical professionals, and researchers to prototype and refine insulin delivery systems tailored to local needs. By addressing gaps in hardware flexibility, user education, and clinical training, it complements existing efforts. For example, integrating its modularity with OpenAPS’s advanced closed-loop software could yield comprehensive, affordable, and user-friendly solutions.
Challenges and Risks
Although primarily designed as an educational tool, the platform allows users to explore advanced challenges like closed-loop system stability and adaptive algorithms. Closed-loop systems inherently require addressing issues like CGM sensor delays, algorithm inaccuracies, and hardware malfunctions, all of which can be explored through the platform’s customizable architecture. In addition, the incorporation of bioinspired algorithms or AI offers the potential to mimic natural glucose-insulin dynamics more effectively. However, challenges such as computational efficiency, sensor precision, and data security must be carefully managed to ensure feasibility and safety.
Adhering to ISO 14971 risk management principles, 26 incorporating safeguards into the platform’s design is critical. Essential safety features include alarms for infusion set obstructions, battery depletion, and delivery malfunctions. Advanced mechanisms like motor shutdown in the event of over-infusion and pressure equalization to mitigate environmental impacts can align the platform with safety practices observed in commercial pumps. These features offer a baseline for users to experiment with and develop safety-critical systems.
Limitations
This study has limitations, notably the small sample size restricted to specialists from a single institution in Mexico, which limits generalizability. Future studies should involve broader participation across regions and institutions to better evaluate adaptability. The reliance on self-reported survey data may also introduce bias, as participants could overestimate their confidence or interest. Objective metrics, such as performance-based evaluations during platform use, would complement survey results and strengthen findings in future stages of development.
Moreover, the controlled testing environment, while useful for detailed feedback, lacks the variability of real-world conditions. Long-term field testing under diverse clinical and environmental scenarios will be necessary to assess the platform’s robustness and reliability.
Conclusion
This study introduces a cost-effective, modular infusion platform designed to give diabetes specialists hands-on experience with device mechanics, customization, and control algorithms. Inspired by open-source projects like OpenAPS, the platform transforms specialists from passive users to active developers, bridging gaps in clinical training and fostering a deeper understanding of infusion technology. Survey feedback demonstrates its effectiveness in increasing specialists’ interest in customizing and understanding insulin pumps.
The platform’s modularity and adaptability for closed-loop systems underscore its potential for clinical application. With an estimated material cost under $200, it offers a viable solution for low-resource settings where traditional pumps are prohibitively expensive. Further development of a user-friendly interface, enhanced connectivity, and risk management could enable eventual patient use, allowing individuals greater independence in diabetes management.
While the platform has strong educational value, additional studies with larger samples and operational validation are necessary for its transition to a clinical-grade device. Continued refinement and training resources will improve usability, safety, and effectiveness. By bridging education and clinical application, this affordable and flexible platform has the potential to democratize access to advanced diabetes technology, ultimately transforming specialist training and patient care in support of accessible, personalized diabetes solutions.
Footnotes
Abbreviations
AI, artificial intelligence; BLE, bluetooth low energy; CGM, continuous glucose monitor; DIY, do-it-yourself; EEPROM, electrically erasable programmable read-only memory; GPIO, general purpose input-output; I2C, interintegrated circuit; MPC, model predictive control; OOP, object-oriented programming; PCB, printed circuit board; PID, proportional-integral-derivative; RTC, real-time clock; UART, universal asynchronous receiver-transmitter; USB, universal serial bus.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by CONACyT (grant no. CVU 682831).
