Abstract
Background:
Smart systems for multiple daily injections (Smart MDI) integrate continuous glucose monitoring, connected insulin pens, smartphone apps, and cloud-based data storage to provide bolus and corrective dose suggestions, reminders/alerts, automatic tracking and sharing of insulin therapy, and glycemic data to users, caregivers, and providers. This is an expert consensus on the clinical value of Smart MDI and critical points for implementation in adults and children/adolescents with diabetes.
Methods:
A nominal group technique combined with the estimate-talk-estimate approach was employed to achieve consensus among panel members from the Italian Intersociety Technology and Diabetes Study Group with expertise in pediatric and adult diabetes care.
Results:
The expert consensus indicated that glycemic profiles can be improved by using bolus dose suggestions based on glucose values, planned meals, the insulin-to-carbohydrate ratio, correction factors, and consideration of insulin-on-board. Automatic remote sharing of patient data on glycemia and insulin therapy allows clinicians to make more appropriate and timely therapeutic recommendations based on objective data. Dose tracking, bolus reminders/alerts, and reduced hypoglycemia and associated anxiety achieved through Smart MDI may improve adherence.
Conclusions:
Smart MDI can reduce treatment burden while improving the daily experiences and glycemic outcomes for adults and children/adolescents with type 1 or type 2 diabetes. However, high-quality clinical data are lacking, and more evidence is needed to compare the effects of Smart MDI and other advanced insulin delivery systems on glycemic and patient-reported outcomes.
Introduction
Maintaining glucose values in the target range delays or prevents the onset of microvascular and macrovascular complications in individuals with diabetes.1-3 However, evidence from large retrospective database studies suggests that many people struggle to achieve American Diabetes Association (ADA) and International Society for Pediatric and Adolescent Diabetes (ISPAD) glycated hemoglobin A1c (HbA1c) targets.4-7
This is at least in part because managing intensive insulin therapy regimens requires constant vigilance and is associated with a significant treatment burden and stress.8-11 Together with the demands of daily life and fear of iatrogenic hypoglycemia, these factors can result in suboptimal adherence and outcomes.12-14 Digital technologies have improved the management of diabetes and reduced the treatment burden for individuals with diabetes and health care providers (HCPs).15,16 Clinical studies have confirmed the value of continuous glucose monitoring (CGM) in increasing time in range while reducing hypoglycemia.17-20 Insulin pens provide convenient and accurate insulin delivery, and many can record and display the last dose administered and time of injection. Connected insulin pens are advanced devices that synchronize with mobile apps for dose tracking, dose reminders, missed dose alerts, calculating bolus and correction doses, and to interface with other devices such as CGM or wearable fitness tracking devices. The clinical evidence supports the benefits of connected insulin pens,21,22 improving glycemic outcomes while reducing treatment burden and enhancing the quality of life of people living with diabetes. 23
A smart insulin pen system for multiple daily injections (Smart MDI) combines these components with an associated companion app and a cloud-based data storage platform (Box 1). 24 Advanced diabetes ecosystems for intensive insulin therapy include the functions of real-time CGM, connectivity, digital health software for decision support, bolus dose calculation and associated reminders/alerts, and automatic presentation and archiving of data on glycemia and insulin doses for access by the diabetes care team for optimizing individual care plans. However, although available and used in real-world settings, there are little clinical data to guide practitioners. Therefore, the aim of this consensus was to identify the clinical utility of a Smart MDI system and critical points for successful implementation in both the adult and pediatric populations with diabetes. We chose a consensus methodology as an initial review of the literature revealed only limited robust clinical data, despite real-world use, precluding the formulation of evidence-based guidelines.
Characteristics of an Ideal Smart Multiple Daily Injection (Smart MDI) System.

Methods
To overcome some of the negative aspects of group dynamics, the nominal group technique (NGT) was combined with the estimate-talk-estimate (E-T-E) approach to achieve consensus among members of a panel with expertise in pediatric and adult diabetes care. The NGT is a formal method developed to encourage equal participation in arriving at a consensus.25,26 The steps in the NGT procedure are presented in Figure 1. The nominal group consisted of current or former members of the Italian Intersociety Technology and Diabetes Study Group. Inclusion criteria were that all had first-hand experience using the Smart MDI system and were internationally recognized experts in the field.
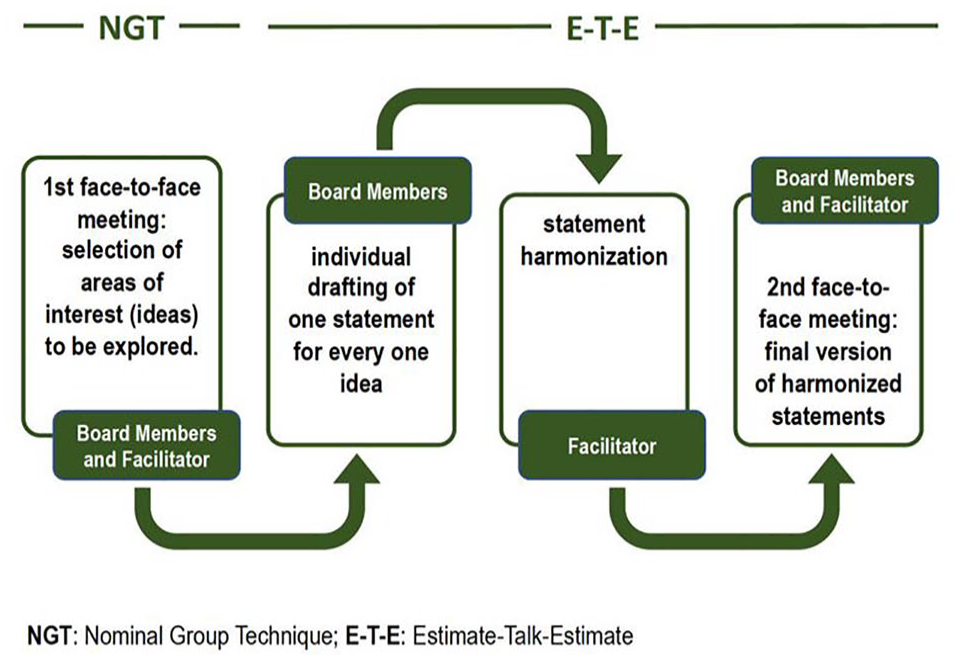
Overall process used to obtain consensus [from
During the first of the three face-to-face meetings, each member of the board worked independently in silence to identify a list of areas of interest (ideas). Participants then presented their ideas aloud, without discussion, in a round-robin feedback session, while a senior clinical epidemiologist (GP) trained in developing group consensus (the facilitator) recorded them. The facilitator organized and categorized these into nonredundant ideas that were then anonymously ranked by the board to assign priority. Working independently in the period between the first and second meetings, board members drafted one statement for each idea using the E-T-E approach, 28 which combines an independent nominal group activity with a face-to-face interaction phase. 29 These statements were then harmonized by the facilitator before discussion in the second face-to-face meeting, in which board members and the facilitator reviewed, discussed, and modified the statements to arrive at a final agreement.
Working with the support of a medical writer, board members drafted introductory materials and the final statements with supporting evidence from the literature, which they discussed at the third face-to-face meeting and further elaborated through rounds of revision online, arriving at the final consensus presented here. The literature was searched in the PubMed, EMBASE, and Google Scholar Databases using the search terms Smart MDI system AND diabetes AND connected pens. All panel members were from the same country (Italy); however, this consensus is generalizable internationally because all panel members are internationally recognized experts in the field, and the supporting literature was from an international evidence base.
Results
Ten statements were drafted, discussed, modified, and approved by the board of experts. Each statement is presented below, along with the available supporting evidence.
Support for Carbohydrate Counting, With Glycemia-Based Bolus Recommendations

Optimization of insulin therapy is crucial in diabetes management but is complicated by the need for careful calculations to balance basal insulin (long-acting) with bolus insulin (rapid-acting, ultra-rapid-acting) to maintain glucose levels in the target range. Factors influencing the increase in postprandial glucose include meal composition, in particular the amount and type of carbohydrates, in addition to portion size. 30 Administering properly timed prandial bolus insulin based on carbohydrate counting, pre-prandial glucose values, insulin-to-carbohydrate ratio (ICR), and active insulin on board (IOB) can allow flexible meal planning while maintaining glucose levels in a target range. 31 However, many people with diabetes struggle to appropriately estimate their prandial insulin doses, 32 and poor numeracy skills can result in errors in calculating insulin doses. 33 Conversely, automated bolus calculators can help people with diabetes optimize their insulin dosing and improve clinical outcomes.34-36 The evidence suggests that the use of such calculators can significantly improve the postprandial glycemic profile 37 and quality of life. 38
A dedicated app equipped with a bolus calculator that considers parameters, ICR, insulin sensitivity factor (ISF), IOB, and glycemic targets could benefit glycemic control, especially in the postprandial phase.39,40 Integration of a smart insulin pen with an insulin bolus calculator could thus support the implementation of appropriate insulin dosing while facilitating the adoption of this procedure by people with diabetes using MDI. 41 However, some users might find the setup of these devices challenging or the apps difficult to use, especially the elderly.
Improvements in Doctor-Patient Communication

The real-time, remote sharing of glycemic data and information on insulin therapy between people with diabetes and diabetologists represents a significant advance in diabetes care. This type of exchange enables diabetologists to make more informed and timely therapeutic recommendations based on objective data.42,43 In addition, a Smart MDI system may extend such benefits by facilitating personalized care through continuous monitoring, particularly in those individuals who may require closer monitoring and attention (eg, people just starting insulin therapy, infants and young children, students studying away from home, younger people with diabetes transitioning to adulthood, nursing home residents, and those with cognitive impairment).42,44 Recognizing patterns in glycemic data allows for early detection of hypoglycemia, prompting timely interventions and notifying caregivers and family members when necessary. Sharing of treatment data creates the opportunity for more effective remote and in-person HCP consultations to identify and resolve treatment issues. 45 Telemedical interventions are also associated with a small but significant improvement in diabetes control.46-48 Overall, these technological advances contribute to a proactive and collaborative approach to diabetes management, enhancing precision in therapeutic decisions and promoting a supportive network for individuals managing diabetes. Prescribers may need initial training to support health care provider uptake and to fully benefit from enhanced communication.
Improved Adherence to Insulin Therapy

Edelman and Polonsky 13 reported that more than 50% of people with diabetes do not meet glycemic goals and that real-world decreases in HbA1c levels were significantly lower than those reported in randomized clinical trials, highlighting poor adherence to medication as a cause of this discrepancy. Missed insulin injections are an important reason for suboptimal outcomes, where HbA1c levels are correlated with adherence to therapy. Randløv and Poulsen 49 reported that missing basal insulin injections twice a week was associated with a 0.3% increase in HbA1c.
Adherence can be improved through a comprehensive insulin administration tracking system that monitors insulin dosage and timing while identifying missed boluses and informing people with diabetes and HCPs on corrective actions. A Smart MDI system has options for tracking compliance50-52 and can improve adherence through reminders, alerts, and alarms related to bolus administration and glycemic values. These features serve as timely cues for people with diabetes, fostering a proactive approach to diabetes management while reducing glycemic excursions. 53
Structured Educational Path

Effective use of a Smart MDI system requires the integration and implementation of a structured educational pathway, which is essential for empowering people with diabetes with the knowledge and complex skills needed to navigate the system successfully.54,55
Beyond its technical capabilities, a Smart MDI system actively engages people with diabetes by suggesting (teaching) context-dependent positive behaviors. Appropriate behaviors may include corrections for underestimated or omitted insulin injections and consideration of active IOB.52,56 This real-time guidance promotes informed decision-making and a preemptive approach to glycemic control. Furthermore, retrospective assessment of recording glucose and therapy data may educate people with diabetes on the effects of medication, nutrition, and physical activity on glucose levels, potentially enhancing awareness and fostering a sense of self-efficacy in managing their disease. The inclusion of structured education in a Smart MDI system could contribute to more personalized and effective diabetes care, ultimately improving patient outcomes and overall quality of life.57-60
Prevention of Hypoglycemic Events and Related Anxiety

Hypoglycemia (<70 mg/dL) can be life-threatening if severe and can lead to fear of future hypoglycemic episodes. Fear of hypoglycemia contributes to treatment nonadherence and suboptimal clinical outcomes when people with diabetes develop avoidance behaviors and other management behaviors that interfere with proper insulin treatment.61,62 It is also associated with anxiety and reduced quality of life in people with diabetes and their caregivers.63,64
The CGM reduces the risk of hypoglycemia in people with diabetes using MDI,65,66 and the use of a bolus calculator reduces the occurrence and fear of hypoglycemia. 67 Improving confidence in a suggested bolus dosage increases the likelihood that a patient will administer the correct dose. 34 Assessment of active IOB plays a crucial role in reducing the risk of hypoglycemia associated with insulin stacking. 68 A Smart MDI system tracking IOB and suggesting the correct insulin bolus dose based on current glucose levels and active insulin may thus reduce the likelihood of hypoglycemic events. This technical capability can alleviate fear and anxiety related to hypoglycemic events, contributing to a more confident and empowered therapeutic approach.
Patient Selection

The ADA Standards of Medical Care in Diabetes recommend that the “type(s) and selection of devices should be individualized based on a person’s specific needs, preferences, and skill level.” 69 A Smart MDI system is a viable consideration for people with diabetes who face challenges in achieving therapeutic goals with traditional MDI and who are unable or prefer not to use an automated insulin delivery (AID) system or continuous subcutaneous infusion device.20,70,71 A Smart MDI system may provide such people with the benefits of advanced technologies while meeting their preferences and lifestyle choices. The versatility of a Smart MDI system makes it applicable to a broad range of people with diabetes, including both the pediatric and adult populations.
We suggest that a Smart MDI system offers a user-friendly and technologically advanced option for individuals with type 1 diabetes starting from the age of two years and for those with type 2 diabetes who are receiving intensive insulin therapy. These people with diabetes can benefit from real-time CGM, automatic recording and transmission of insulin delivery times, doses, and corresponding interstitial glucose data, and the decision-support capabilities of a Smart MDI system. Elderly individuals who may have limited engagement with technology or younger children who may not be able to manage a personal smartphone could face challenges in using Smart MDI systems without supervision (eg, during school activities for children). These special groups of people with diabetes require a cautious approach, as the use of such technologies could be difficult. Support from a third party (parents, spouse/partners, legal guardians) could be a great help to these individuals.
Need for Adequate Scientific Evidence

Diabetes technology is a rapidly evolving area, and much of the evidence is from small studies of short duration. Only a few well-designed randomized controlled trials assess clinical effectiveness.24,72 Current knowledge about the efficacy and safety of Smart MDI systems is based predominantly on noncomparative studies that have assessed various components of a Smart MDI system in people with type 1 diabetes17,18,21,22 or type 2 diabetes.36,73 However, more evidence on a complete Smart MDI system is needed, including studies that compare its safety, clinical efficacy, and real-world effectiveness to traditional MDI and other technologies. Studies should also capture patient-reported outcomes, treatment burden, and patient confidence in self-management, as well as HCP perspectives using this system. Following the acquisition of sufficient scientific evidence, pharmacoeconomic studies should evaluate the economic feasibility and sustainability of Smart MDI systems within the health care landscape. Studies of individual system components suggest an economic benefit, as well as a clinical benefit.24,74,75 Cost-effectiveness is likely to vary according to the health care system structure, and a systematic approach is vital to informing decisions on establishing Smart MDI systems as sustainable tools for diabetes management in routine clinical practice.
Personalization

Achieving precise and personalized diabetes therapy can be challenging due to the complex and diverse nature of individual patient needs. 76 However, a Smart MDI system has numerous customizable options that can be adapted to the needs of individual people with diabetes. For example, insulin dose calculator parameters can be adjusted to suggest fixed doses before pre-announced meals, doses based on the meal estimation method, doses based on carbohydrate counting, or doses based on integration of the carbohydrate count, current glucose value, and IOB. Other parameters to personalize include the patient’s ISF and ICR, as well as the type of insulin and its duration of action, glycemic targets, and maximum doses. Settings for predictive alerts and hypoglycemia/hyperglycemia threshold alarms can also be customized to individualize the balance between safety/efficacy and possible alarm fatigue. As noted above, for some individuals less technologically proficient, such extensive customization may represent a barrier to use.
Insulin Storage
Insulin is sensitive to high and low temperature extremes, 77 such as if left in an automobile in the sun or in an excessively cold part of a refrigerator. Another advantage of a Smart MDI System is its ability to alert users, through alarms and reminders, when their insulin reservoir is low, has reached its recommended shelf life, or has been stored under extreme temperatures that could affect insulin performance. 78
Influence on Quality of Life and Other Patient-Reported Outcomes

Diabetes is associated with increased anxiety and reduced QoL for both people with diabetes and caregivers.8-10 Several components of a Smart MDI system have been shown to improve QoL in people with type 1 and type 2 diabetes receiving intensive insulin therapy, including CGM79,80 and connected insulin pens. 81 Moreover, sharing of CGM data is associated with improved QoL in caregivers who are involved in diabetes management. 80 Although several components of a Smart MDI system are known to improve QoL, formal studies with the entire system using validated QoL instruments are needed to determine the overall effect.
Conclusions
Increasing the time in target glycemic range while avoiding hypoglycemic episodes is crucial for improving the health and QoL of people with diabetes. Only a minority of people with diabetes achieve ADA-recommended HbA1c targets. Smart MDI systems could be instrumental in achieving these therapeutic goals in people with diabetes who choose to administer intensive insulin therapy with MDI. Indeed, a Smart MDI system may improve adherence, glycemic control, and safety in people with diabetes receiving intensive insulin therapy while reducing treatment burden and addressing several challenges faced by those using MDI.
The Smart MDI system’s capacity for precise and personalized insulin dosing may help reduce glucose fluctuations, positively impacting the daily experiences of those living with diabetes. In addition, it can improve doctor-patient communication, relieving the patient of the burden of maintaining and transmitting an electronic diary. Retrospective data analysis helps with insulin dose adjustment while enhancing the patient’s awareness of disease management and factors that influence their glycemia.
Overall, there is a lack of evidence-based clinical data on Smart MDI use, precluding the formulation of evidence-based guidelines at this time. Nevertheless, this consensus is strengthened by using recognized methodologies for consensus building and the expertise of participants. In addition to the lack of high-quality clinical data, the study is also limited by participants only attending from one country (Italy). More research is needed to fully define the effectiveness of this technology, its impact on patient/caregiver QoL, and pharmacoeconomic parameters. Structured education is needed for people with diabetes and health care providers to obtain all the potential benefits offered by the Smart MDI system.
Although the relatively small amount of data on Smart CGI is generally positive, several barriers will need to be overcome. Specifically, device interoperability (ie, device-agnostic systems) is needed to allow flexibility in tailoring the system to individuals with diabetes through device selection, while implementations of standardized device data and data-sharing protocols will facilitate the insertion of data from different sources into electronic health records. Most importantly, the utility of these devices requires robust testing in well-designed clinical studies to compare the effects of Smart MDI and other advanced insulin delivery systems on glycemic and patient-reported outcomes.
Footnotes
Acknowledgements
The authors thank Richard Vernell for editorial assistance, funded by Medtronic.
Abbreviations
SmartMDI, Smart Multiple Daily Injections; HbA1c, glycated hemoglobin A1c; HCPs, health care providers; CGM, continuous glucose monitoring; NGT, nominal group technique; E-T-E, estimate-talk-estimate; ICR, insulin-to-carbohydrate ratio; IOB, insulin on board.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FG has served as an advisor for AstraZeneca; a research investigator for Eli Lilly; a speaker for AstraZeneca and Eli Lilly; a consultant for Abbott, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Medtronic, Merck Sharp & Dohme, Novo Nordisk, Roche Diabetes Care, and Sanofi; and has received grants from Eli Lilly, LifeScan, and Roche Diabetes Care. RB has served on Speaker’s Bureau: Eli Lilly, Sanofi, Menarini, Medtronic, Abbott, Theras, Movi; Board Member/Advisory Panel: Sanofi, Medtronic, and Ypsopump; and consultant for Eli Lilly, Sanofi, Abbott, Medtronic, and Movi. FC has been a speaker for LifeScan; advisory board member for Medtronic. CI has participated in advisory boards for Novo Nordisk, Lilly, Roche Diabetes Care Italy, Abbott, Menarini, Ascensia, Senseonics, and Medtronic; and received speaking fees from Novo Nordisk, Roche Diabetes Care Italy, Abbott, Ascensia, Lilly, and Boehringer Ingelheim Pharmaceuticals. AL participated in an Advisory Board for Medtronic and was/is a consultant for Eli Lilly, Menarini, Abbott, and Medtronic. CM has participated in an advisory board for Medtronic. GP has participated as a facilitator in an advisory board for Medtronic. DP has participated in an advisory board for Medtronic. IR has participated in Speaker’s Bureau for Eli Lilly, Sanofi, Menarini, Medtronic, Abbott, and Theras; Advisory Board Member: Sanofi and Medtronic; and consultant for Eli Lilly, Sanofi, Menarini, Abbott, and Medtronic. EZ has participated in an Advisory Board for Medtronic. AS has spoken for Sanofi and Abbott, has received support for attending meetings from Movi, and has served on advisory boards for Medtronic and Movi.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The advisory board was funded by Medtronic Italy spa, Milan, Italy. However, Medtronic had no role in steering or suggesting any of the content in this paper. The consensus reached by the Expert Committee depicts solely their expertise and does not necessarily represent the opinion of Medtronic.
