Abstract
Background:
Glucose is an essential molecule in energy metabolism. Dysregulated glucose metabolism, the defining feature of diabetes, requires active monitoring and treatment to prevent significant morbidity and mortality. Current technologies for intermittent and continuous glucose measurement are invasive. Noninvasive glucose measurement would eliminate this barrier toward making glucose monitoring more accessible, extending the benefits from people living with diabetes to prediabetes and the healthy.
Methods:
A novel spectroscopy-based system for measuring glucose noninvasively was used in an exploratory, prospective, single-center clinical study (NCT06272136) to develop and test a machine learning-based computational model for continuous glucose monitoring without per-subject calibration. The study design blinded the development investigators to the validation analyses.
Results:
Twenty subjects were enrolled. Fifteen were used for the development set, and five in the validation set. All study participants were adults with insulin-treated diabetes and median glycated hemoglobin (HbA1c) of 7.3% (interquartile range [IQR] = 6.7-7.7). The computational model resulted in a mean absolute relative difference (MARD) of 14.5% and 96.5% of the paired glucose data points in the A plus B zones of the Diabetes Technology Society (DTS) error grid. The correlation between the average model sensitivity by wavelength and the spectrum of glucose was 0.45 (P < .001).
Conclusions:
Our findings suggest that Raman spectroscopy coupled with advanced computational methods can enable continuous, noninvasive glucose measurement without per-subject invasive calibration.
Introduction
Glucose is a tightly regulated energy source for human cells, fueling many cellular processes and metabolic functions. Blood glucose concentration is affected by many factors, including food, satiety, exercise, sleep, hormones, and mood. 1 Daily blood glucose fluctuations are a normal response to dietary intake and energy expenditure, but an excess caloric intake, particularly rich in sugars and starches, combined with a sedentary lifestyle, are proven risk factors for developing dysglycemia and, eventually, diabetes mellitus (DM). 2 DM is a notable health concern worldwide, currently affecting approximately half a billion people, projected to reach 783 million by 2045 3 with significant morbidity and mortality primarily caused by micro- and macrovascular complications.
The real-time tracking of glucose via continuous glucose monitors (CGMs) has improved the management of DM, particularly in type 1 and insulin-treated type 2 DM, where reductions of glycated hemoglobin (HbA1c), increases in time spent in the target glucose range, and reductions in the number of hypoglycemic episodes have been demonstrated.4-7 More recent data show that benefits are also seen in insulin-naive type 2 DM,7,8 whereas the value of CGM in prediabetes 9 and people without dysglycemia10,11 is an area of active research. Supporting this trend, the Food and Drug Administration (FDA) has recently cleared the use of CGMs for people not diagnosed with diabetes. 12
Today’s CGMs are minimally invasive, relying on a needle-based applicator to insert a microneedle filament, which uses an electrochemical approach to quantify the glucose concentration in the interstitial fluid. Since their market entry, CGM devices have steadily increased their accuracy and lifetime, reduced their footprint, and expanded their connectivity to phones, smartwatches, and pumps. Although CGMs have greatly improved the quality of care for people living with insulin-treated DM, noninvasive CGM technology can eliminate the discomfort and risks associated with minimally invasive monitoring, thus eliminating a significant barrier to adoption. 13 However, to eliminate this barrier, a noninvasive monitoring approach should also not require per-person invasive calibration to continuously measure glucose accurately. Many technologies and approaches have been proposed and pursued, but a noninvasive method for glucose measurement without per-person calibration has been elusive. 14
We here report the first results of an exploratory, prospective, single-center, multiple sequential-cohort study designed to assess the ability of a novel noninvasive investigational device to measure glucose without per-person calibration in adults living with type 1 or type 2 DM.
Methods
This study (NCT06272136) was conducted at Profil (Neuss, Germany) under consideration of the Declaration of Helsinki and in compliance with the Guideline for Good Clinical Practice and the applicable national regulations and provisions. The study protocol was approved by the Ärztekammer Nordrhein (Düsseldorf, Germany) Ethics Committee and exempted from approval by the German Federal Institute for Drugs and Medical Devices (BfArM). Data shown here are from the cohorts I and II participants, recruited between February 21 and June 4, 2024.
Study Devices
The investigational device (“Clinical Demo 2.0”, Liom Health AG, Pfaeffikon SZ, Switzerland) is a stand-alone device with a single millimeter-size exit glass window allowing measurement on the palmar side of the subject’s wrist (Figure 1). The device employs Raman spectroscopy (<3500 cm−1) to capture the signature of the glucose molecule in the human skin in contact with the window. During the measurements, the skin is continuously exposed to a sub-millimeter near-infrared light spot at power levels below 100 milliwatts. The device collects and captures with a spectral analyzer the near-infrared light response signals from the epidermis and dermis, which contain the instantaneous and specific signatures of the elements composing the skin tissues and liquids. The measured signals are sorted by wavenumbers, and then the corresponding amplitudes are recorded before being processed and analyzed offline with our computational procedure for glucose concentration estimation. The optics of the device were configured such that the dermis is measured, focusing primarily on measurements of interstitial fluid.
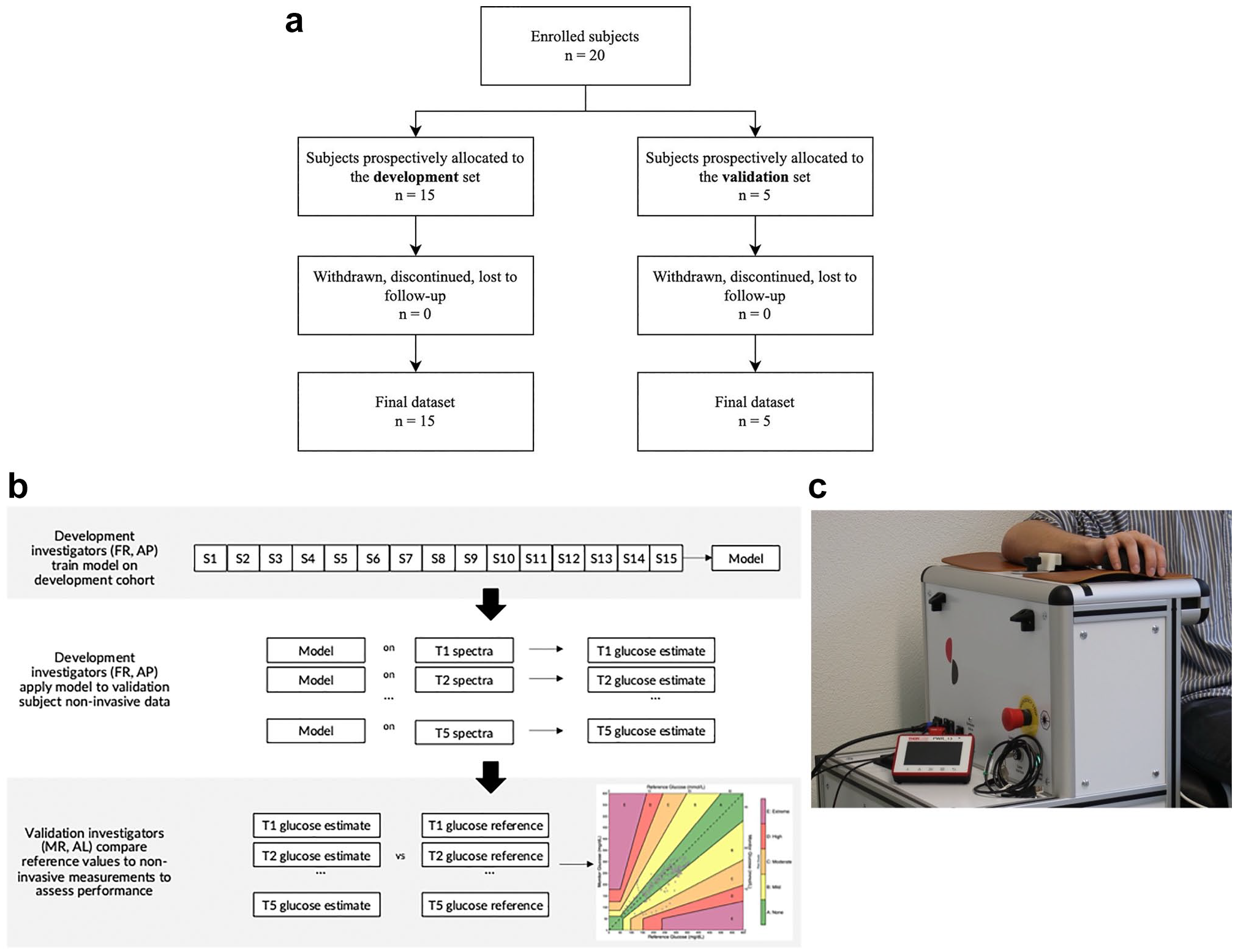
(a) Study diagram. (b) Analysis design. (c) Investigational device with the subject’s wrist positioned on the top. A laser interrogates the palmar side of the wrist.
Participants
Study participants were consenting adults living with type 1 or insulin-treated type 2 diabetes for at least one year and meeting all the inclusion and exclusion criteria (Supplemental Table 1). At the screening, their HbA1c was below or equal to 9.5%. They were to have a skin tone in the Fitzpatrick scale type 1 to 4, 15 and a wrist exempt from injury, infection, tattoo, or any atypical skin conditions.
Study Procedures
Enrolled subjects were required to fast for at least 8 hours before the measurement procedures. An indwelling catheter for blood sampling and intravenous insulin injection was inserted into one arm, and the investigational device was fixed to the other arm. Study participants with fasting plasma glucose above 150 mg/dL received an intravenous insulin bolus for plasma glucose lowering. Data collection with the investigational device started 30 minutes before the baseline plasma glucose measurement. For comparison purposes, venous blood samples were collected using the indwelling catheter during the intervention for 5 hours. Time windows for blood sampling were every 5 minutes from intervention until post-meal plasma glucose drop to <150 mg/dL, and from then every 15-minute interval until the end of the intervention. The meal test period started with plasma glucose below 150 mg/dL, and the participant received a predefined carbohydrate-rich meal without insulin administration at the start of meal intake. Plasma glucose values were monitored until the level rose above 300 mg/dL. At plasma glucose above 300 mg/dL, an individualized insulin bolus dose was administered subcutaneously in the abdominal area. In case plasma glucose did not rise above 300 mg/dL after meal intake, the insulin bolus was administered as soon as plasma glucose stopped rising and reached a plateau. An additional insulin bolus was administered in case plasma glucose did not drop below 150 mg/dL after insulin bolus administration. A follow-up visit was made five to eight days after the intervention day for safety purposes. Adverse events, including device-related events, were collected from enrollment until the end of the post-intervention follow-up.
Sample Size
As this was an exploratory clinical investigation to evaluate the ability and the accuracy of the investigational device to measure glucose noninvasively and to capture preliminary information on the investigational device to adequately plan further steps of clinical development, no sample size estimation was performed, and no formal hypothesis testing was planned.
Baseline and Procedural Characteristics
Baseline characteristics, including demographics and information on diabetes management, are presented as medians (interquartile range—IQR) for continuous variables and counts (%) for categorical variables. Procedural characteristics are also presented as medians (IQR) and comprise durations and glucose levels in different glycemic states. In addition, we also report rates of absolute changes, which we categorized into “low” (less than 1.0 mg/dL/min), “moderate” (between 1.0 and 2.0 mg/dL/min), “high” (between 2.0 and 3.0 mg/dL/min), and “very high” (above 3.0 mg/dL/min).
Computational Model Development and Validation
Among the 20 subjects of this first study results, 15 were allocated to the training data set, although the remaining five were allocated to the validation data set. No subject was withdrawn, discontinued, or lost to follow-up (Figure 1a). The data used for algorithm training and development was not eligible for evaluating the study objectives, and the data split was predefined prior to the study start. The computational procedure is a differentiable regression model which was developed based on the data of this development cohort. The procedure processes the raw signals via simple frequency transformation and filtering to ensure that the data are standardized before being analyzed by a model for estimating glucose. The development investigators (FR and AP) were blinded to the reference data used to evaluate this study’s primary and secondary outcomes. A predefined, mutually exclusive data split was used to evaluate the primary and secondary endpoints of the study: the first 15 subjects were assigned to the development set, and the subsequent five to the validation set. The analysis of the validation set was performed in silico, after study completion, by two independent statisticians from another group not involved in the clinical intervention or data collection (MR and AL) (Figure 1b). All analyses were performed using the intention-to-treat data set using the statistical software R version 4.4.0. No imputation for missing data was performed, although obviously erroneous data points based on spectral quality were excluded from the analyses.
Point Accuracy Assessment
The primary analysis was performed by visualizing both the Clinical Demo 2.0 values and the reference plasma glucose measurements over the time of intervention in individual graphs. The determination of the accuracy of the glucose estimations against venous plasma glucose measurements was assessed by computing both the overall mean absolute relative difference (MARD) and the MARD at the participant level. A Diabetes Technology Society (DTS) error grid was generated for the full validation data set for point accuracy. 16 We report the proportion of data points for each of the five zones for the full validation dataset and separately per participant.
In Vitro Measurement of Glucose
A 64-mM glucose solution was prepared in a UV cuvette using the same setup as in the clinical investigation to collect a clean Raman spectral signature of the glucose molecule. The spectra were cropped in the wave number range 227 to 3668 cm−1 and denoised.
Results
The study population consisted of ten women and ten men. The median age was 41 years (IQR = 31-45), the median body mass index (BMI) was 25.8 kg/m2 (IQR = 22.9-28.6), and the median HbA1c was 7.3% (IQR = 6.7-7.7). Participants had type 1 DM for more than one year and skin type II (25%), III (40%), or IV (35%) according to the Fitzpatrick scale 15 (Table 1). Among participants, the time from the start of measurement procedures to hyperglycemia ranged from 5 to 75 minutes, and the median time in hyperglycemia was 260 minutes (IQR = 197.5-274.3). Median plasma glucose values were 117 mg/dL (IQR = 104-135) at the start of the procedure and similar at discharge (119 mg/dL; IQR = 85-180). Median plasma glucose during hyperglycemia “(defined as glucose values above 180 mg/dL)” was 256 mg/dL (IQR = 240-279). The lowest measured plasma glucose value among all interventions was 37 mg/dL, and the highest was 406 mg/dL. Distributions across the different rate of change (RoC) categories were comparable in the two cohorts: in both groups, 16% of the measured datapoints were during very high RoC, approximately 15% in high, 31% in moderate, and 37% during low RoC (Table 2). The mean absolute RoC was 1.73 mg/dL/min (IQR = 0.60-2.40) for the validation cohort, and 1.75 mg/dL/min (IQR = 0.60-2.40) for the development cohort.
Baseline Characteristics.
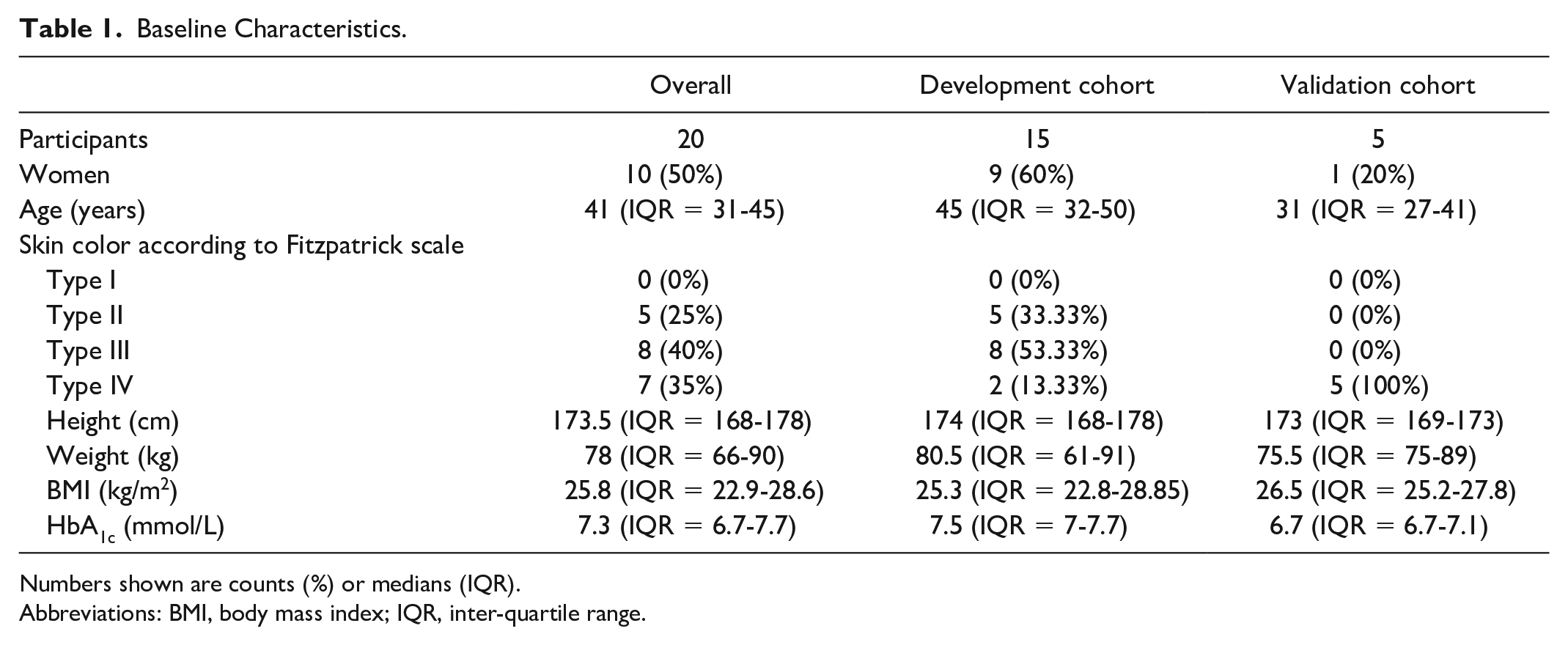
Numbers shown are counts (%) or medians (IQR).
Abbreviations: BMI, body mass index; IQR, inter-quartile range.
Procedural Characteristics.
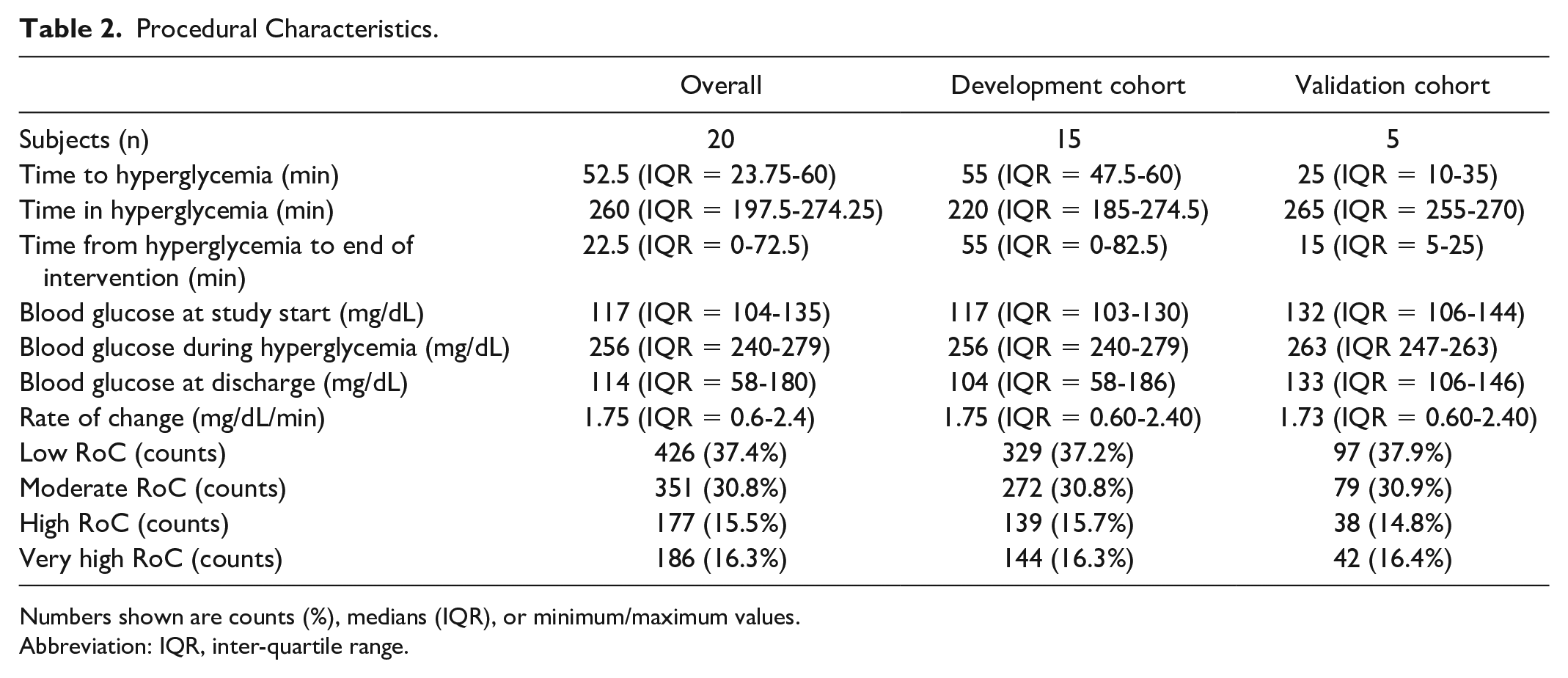
Numbers shown are counts (%), medians (IQR), or minimum/maximum values.
Abbreviation: IQR, inter-quartile range.
The MARD in the validation cohort was 14.5%, ranging from 11.1% to 22.0% at the individual level. The time series of the development and validation cohorts were similar (Supplemental Figure 1). The individual time series of the five subjects of the validation cohort visually depicts the correlation between the reference and test device glucose values (Figure 2). The MARD in hyperglycemia (above 180 mg/dL) was 12.4% (range at the individual level: 7.0%-19.9%) and the MARD in euglycemia (70-180 mg/dL) was 25.9% (range at the individual level: 12.1%-40.8%). The MARD during low RoC was 14.9%, 11.6% during moderate, 11.2% during high, and 19.6% during very high RoC.
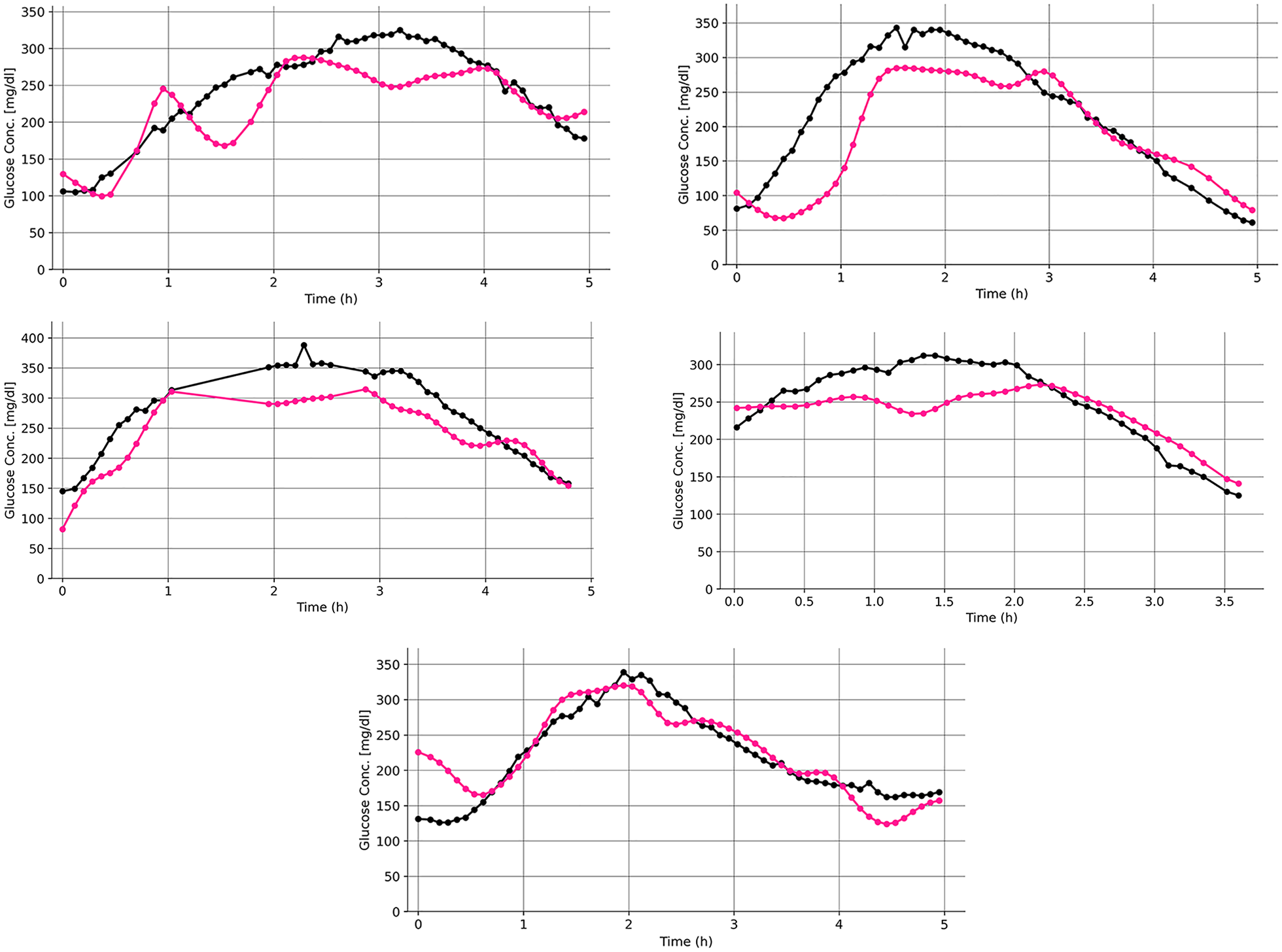
Time series of the five subjects of the validation cohort.
In the validation set, 78.8% of matched pairs were in zone A, 17.7% in zone B, and 3.5% in zone C, while none were in zones D and E, as per the DTS error grid 16 (Figure 3). Four of the five participants had estimated glucose values that were in zones A or B only, and the individual proportion of data points in zone A only ranged from 63.2% to 88.6% (data not shown).
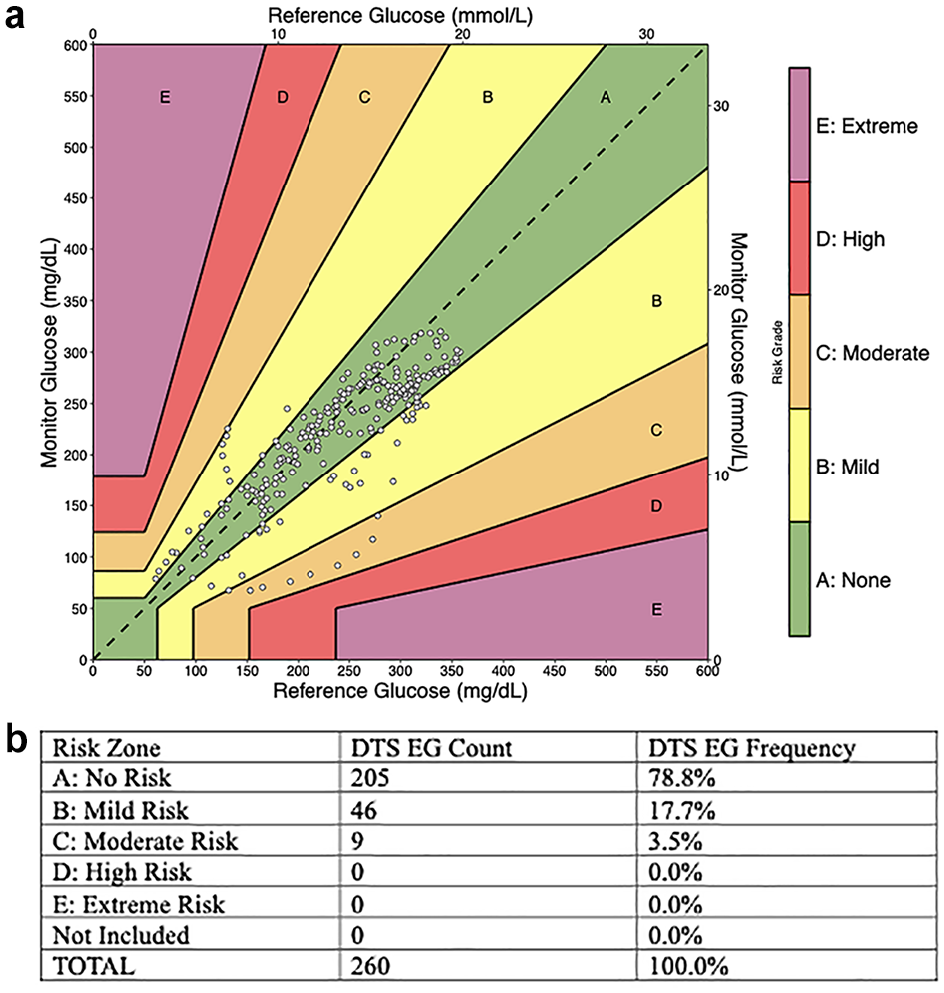
(a) Diabetes Technology Society (DTS) error grid. (b) Number and percent of paired data points in the different zones of the DTS error grid.
Owing to the nature of these noninvasive measurements, there is a question of whether the estimates of glucose are based on a correlate of glucose or based on a measurement of glucose itself. To investigate this, we evaluated our model’s average glucose estimate sensitivity to changes in Raman spectra (ie, the gradient of the independent variable of glucose with respect to the dependent variables of Raman spectrum intensities) and compared this to the glucose spectrum as evaluated from a series of aqueous glucose solutions (Figure 4). To provide robust estimates of glucose, the model must not only be sensitive to glucose but also insensitive to other Raman signals unrelated to glucose estimation. In other words, as nontarget biomarkers induce changes in the spectrum, the output of the model must be insensitive to these directions of change. This phenomenon is also observed in similar approaches to model interpretation in explanation vectors for classification models. 17 Owing to this dual role, it is expected that the correlation coefficient has a moderate value. We found a Pearson correlation coefficient of 0.45 (P < .001).
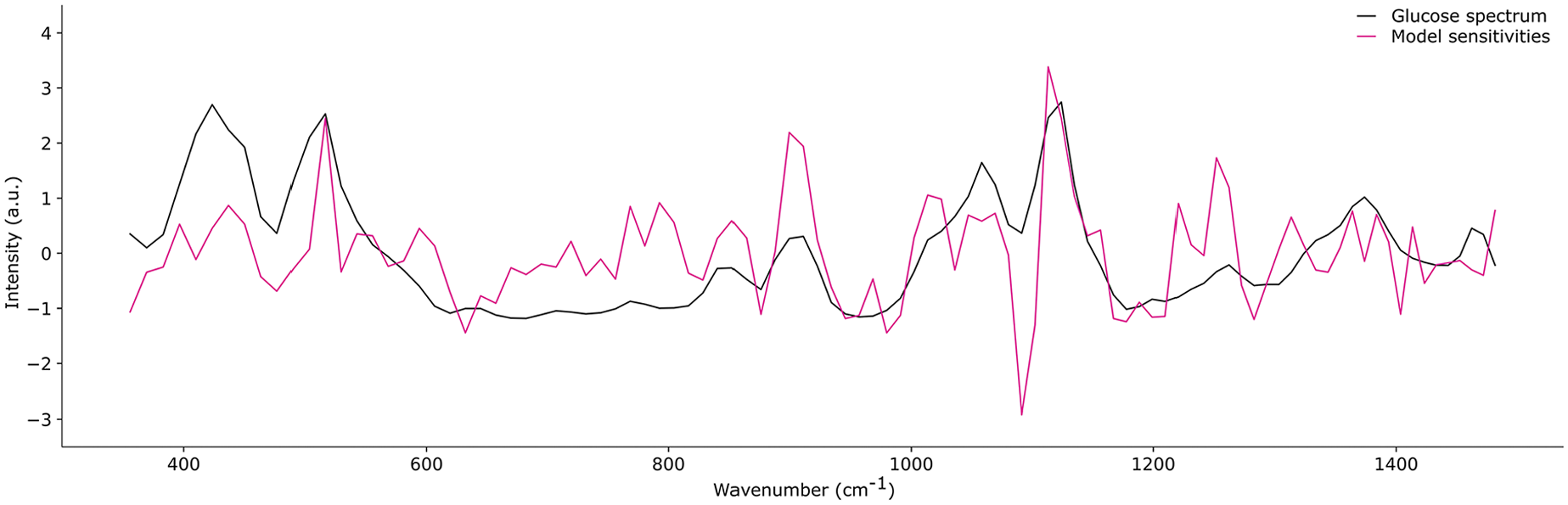
Model sensitivity compared with glucose spectrum.
In summary, 96.5% of the matched pairs between estimated and reference plasma glucose values were in zones A and B of the DTS error grid, which represent the zones where errors are expected to have little or no effect on clinical outcome. The accuracy was relatively consistent across different RoC, ranging from an MARD of 11.2% during high and 19.6% during very high RoC.
Discussion
Our study’s findings suggest that measuring glucose noninvasively without invasive calibration is possible and confirm that optical systems combined with advanced computational models are highly promising.
The strengths of this study are (1) the prospective, external validation design where a pre-defined subset of the study subjects is used to assess the performances of the computational model issued from a development cohort, (2) the focus on developing a calibration-free computational model, and (3) the study procedure that consisted of rapid and large glucose excursions in a population of people living with profound defects in their glucose metabolism. There is a rich literature of approaches and attempts to measure glucose noninvasively, and the typical approach is to develop a computational model with per-person calibration. This fact is often implicit in the design of the development and validation measurements. In this case, the development and validation measurements were partitioned by subject such that no subjects were present in both the development and validation cohorts. In addition, the evaluation of the validation cohort estimates was done in an independent fashion such that strictly separate investigator groups performed model development and model evaluation. The study population has large and rapid glucose changes that make glucose monitoring especially challenging.
Limitations include the modest sample size of the validation cohort, the lack of subjects with darker skin (types V and VI on the Fitzpatrick scale), the lower glucose peak than expected, and the limited data in hypoglycemia. As the glucose profiles used for training and validation had a very similar shape, it could be that the model learned to output the shape of the profile, independent from the input. Therefore, we developed a similar model with data from only a subset of the subjects and only for time periods of strictly increasing glucose values. This model, applied to all time periods for the remaining subjects, accurately tracked the full trajectory despite never being exposed to decreasing glucose values (data not shown).
Overall, the performance is comparable to first-generation CGMs 18 but unlike those systems, this device does not require per-person calibration. Today’s best-in-class CGMs are factory-calibrated and do not require users to perform invasive measurements, such as fingerstick blood glucose measurements, unless CGM results do not match user symptoms. Their success, combined with their limitation of invasiveness, has long spurred interest in developing noninvasive methods to sense glucose continuously. The glucose molecule’s well-characterized optical properties have led to multiple attempts to develop spectroscopic sensors. Although numerous proofs-of-concept have been shown in laboratory conditions, this is one of the first to demonstrate state-of-the-art performance without invasive calibration. 19 In addition, to have the potential for a convenient, comfortable wearable form factor, such technology should have the potential for miniaturization while still demonstrating adequate accuracy and robustness in real-life conditions.14,20,21 The convergence of recent developments in the fields of photonics, electronics, and computational sciences, especially regarding miniaturization, opens new opportunities to bring continuous, noninvasive glucose sensing to the wrist eventually. The results presented here lay down the foundations toward the development of such a technology.
Conclusions
This study shows that the Raman spectroscopy-based technology embedded in the investigational device and the associated machine-learning computational model bear great promise to enable the development of a wearable, noninvasive glucose monitor that does not require per-person calibration. Future efforts will focus on miniaturizing the technology to a smartwatch-like form factor to ensure user convenience and further developing and testing the computational model to ensure adequate accuracy is also achieved in the real-life utilization of a CGM, in a more diverse population, including the assessment of the system’s performance in healthy and prediabetes populations.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251313811 – Supplemental material for A Prospective Pilot Study Demonstrating Noninvasive Calibration-Free Glucose Measurement
Supplemental material, sj-docx-1-dst-10.1177_19322968251313811 for A Prospective Pilot Study Demonstrating Noninvasive Calibration-Free Glucose Measurement by Martina Rothenbühler, Aritz Lizoain, Fabien Rebeaud, Adler Perotte, Marc Stoffel and J. Hans DeVries in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors are grateful for all the work and continuous support from many colleagues at Liom Health AG, Profil GmbH, and DCB Research AG, without which this work would not have been possible.
Abbreviations
CGM, continuous glucose monitor; DM, diabetes mellitus; DTS, Diabetes Technology Society; IQR, interquartile range; BfArM, German Federal Institute for Drugs and Medical Devices; MARD, mean absolute relative difference; RoC, rate of change; HbA1c, glycated hemoglobin.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AP and FR are the employees of Liom Health AG.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Liom Health AG.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
