Abstract
Background:
People with diabetes benefit from continuous glucose monitoring (CGM) systems. A downside of these valuable aids for diabetes management is the generation of a tremendous amount of waste. This study aimed to quantify this CGM-related waste.
Method:
Twenty-four used CGM sensors from two different manufacturers (8× FreeStyle Libre 2, 11× FreeStyle Libre 3, and 5× Dexcom G7) were dismantled manually and separated in case, circuit board, and battery. Each component as well as included packaging, applicator, and leaflet were weighed separately.
Results:
Packaging, applicators, and leaflets accounted for most of the waste (FL2: 93.4 g; FL3: 58 g; G7: 108.1 g). The plastic case contributed mainly to the total sensor weight (FL2: 1.9 g/63% of 3.3 g; FL3: 0.5 g/49% of 1.1 g; G7: 1.9 g/59% of 3.2 g), whereas the weight of the electronic circuit board and battery varied (FL2: 0.8 g/25%, 0.4 g/12%; FL3: 0.2 g/17%, 0.4 g/34%; G7: 0.7 g/22%, 0.6 g/19%). Extrapolating these data based on annual worldwide usage of around 230 million glucose sensors, approximately 20,000 tons of packaging, applicators, and leaflets and 580 tons of glucose sensors are disposed of, including about 340 tons of casings, 130 tons of circuit boards, and 110 tons of batteries.
Conclusions:
Our data highlight the potential for optimized resource utilization by reduction of packaging, sensor size, longer application periods, implementation of multiuse applicators, and the need for recycling options.
Introduction
Today continuous glucose monitoring (CGM) systems are standard of care in the treatment of people with type 1 diabetes mellitus and frequently used in people with type 2 diabetes. In daily clinical practice, technical aspects of the CGM systems and the glucose sensors used are of secondary importance. The choice of a given CGM system is driven by the preferences of the respective person with diabetes, costs, compatibility with insulin pumps, and so on. Although the waste generation associated with diabetes management has become a topic of interest for users, health care professionals, and manufacturers,1,2 relatively little is known about the waste footprint of CGM systems. Glucose sensors are worn for 7 to 14 days (depending on the type of CGM system), adding up to dozens of sensors used per year by each person with diabetes. Each sensor is packed in cardboard packaging which additionally contains an applicator device and an informational leaflet. As estimated by the available data of CGM users, on a worldwide perspective at least 230 million glucose sensors are used annually and subsequently disposed of, with a steep increase in the numbers per year. 3
Briefly, the glucose sensor of the CGM systems used most widely (>99%) consists mainly of a plastic case, an electronic circuit board, and a battery, ignoring expensive and critical parts like the needle-type glucose sensor itself. The batteries in the glucose sensors are mostly zinc-silver oxide or lithium-ion units (for an overview of batteries in diabetes technology refer Avari et al 4 ). The batteries are needed to power the glucose measurement process itself, storing the data, and transmitting them to a receiver, which today is most often a smartphone, or a handheld receiver offered by the manufacturer of the given CGM system.
This study aimed to quantify the amount of waste associated with the usage of CGM systems. Therefore, the glucose sensors of three different CGM systems widely used (FreeStyle Libre 2 [FL2], FL3, and Dexcom (DC) G7 (G7)) were manually dismantled and the different parts as well as their cardboard packaging, paper leaflets, and applicator devices were weighed. These data were extrapolated employing the number of sensors used worldwide to estimate the amount of generated waste.
Materials and Methods
Glucose Sensors
Twenty-four glucose sensors (FL2: n = 8; FL3: n = 11; G7: n = 5) were collected by people with diabetes (PwD) of a specialized diabetes practice in Germany during a previous study 5 (Figure 1).
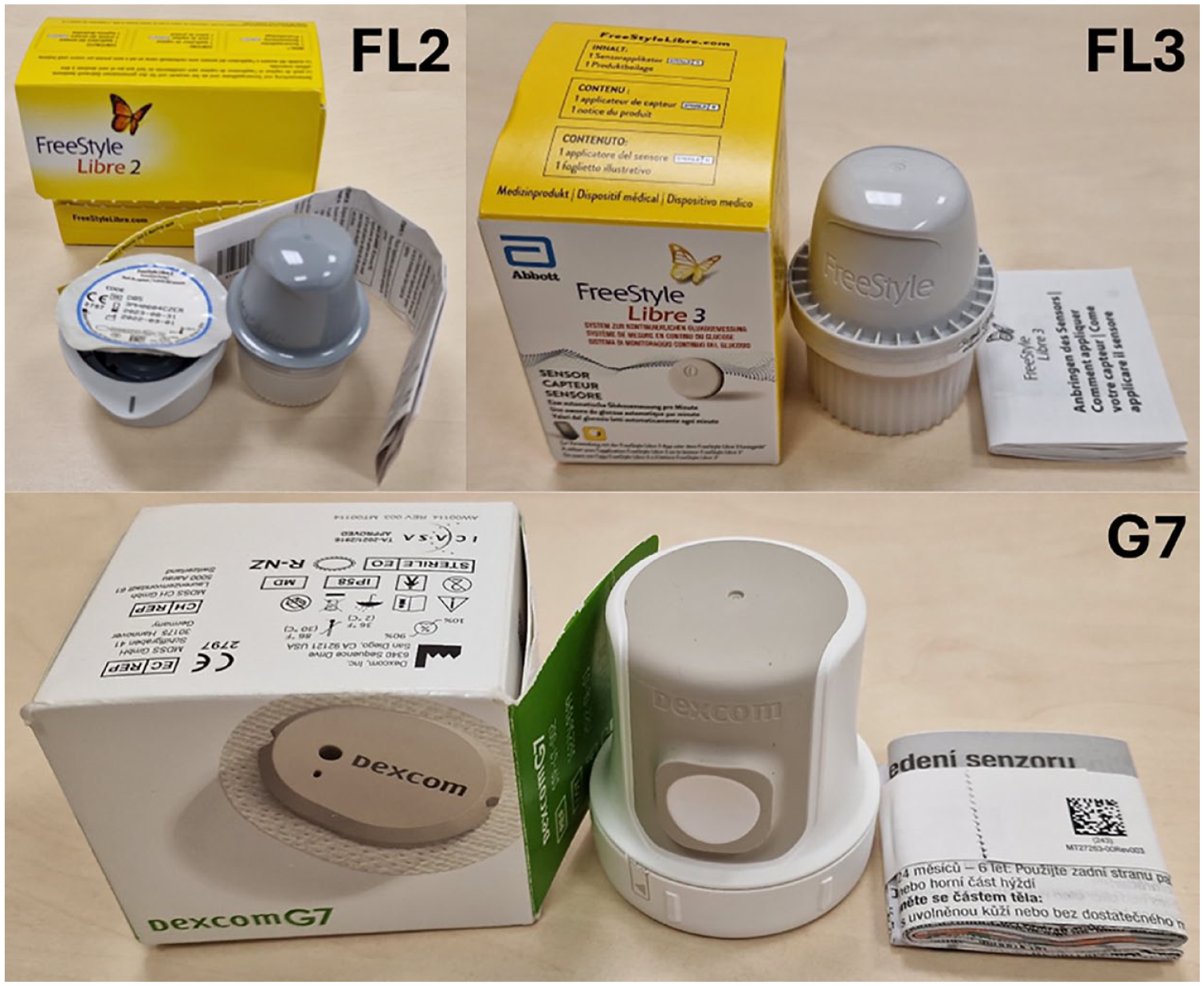
Analyzed glucose sensors with their respective packaging, applicators, and leaflets. Packaging is made of cardboard, applicators mostly of plastic, and leaflets of paper. Top left: FreeStyle Libre 2, top right: FreeStyle Libre 3, bottom: Dexcom G7.
Each sensor was manually dismantled, employing a vice, slotted screwdriver, and hammer to enable measurement of the plastic case, the electronic circuit, and the battery (FL2: SR626W, Seizaiken, Japan; FL3: SR716SW, Seizaiken, Japan 6 ; G7: CR1216, Maxell, Japan 7 ) being part of each sensor (Figure 2). Each part as well as the packaging, the applicator, and the leaflet with printed information were weighed with a laboratory scale (DIPSE Joshs MR1, SSR, Oldenburg, Germany).

Disassembled glucose sensors. Representative images of dismantled glucose sensors showing the plastic casing, electronic circuit board, and battery. From top to bottom: FreeStyle Libre 2, FreeStyle Libre 3, and Dexcom G7. The black bar indicates 2 cm.
Extrapolation of Data
The obtained data were extrapolated using available data on CGM users worldwide. According to Kelly Close’s recent CGM memorandum, 3 approximately 5.7 million people are using the FL system and 2.3 million users of DC. These numbers were multiplied by the minimal number of sensors required yearly (26 FL sensors, 37 DC sensors), resulting in 148.2 million FL sensors and 85.1 million DC sensors (233.3 million sensors combined) per year. The mean weight of the measured components, that is, packaging, applicator, leaflet, and the total weight of the sensor, case, circuit board, and battery were multiplied by the number of sensors. Since there are no data available on the distribution of FL 2/3 and DC G6/7, the number of FL sensors was distributed equally, that is, divided by 2, on the FL2 and 3 system. As we only studied the G7, we used the total amount of DC sensors for the calculation.
Statistics
GraphPad Prism 7 (GraphPad Software, San Diego, CA, USA) was employed for statistical analyses. The Shapiro-Wilk’s test was used to test the data for normal distribution. A nonparametric test (Kruskal-Wallis) was used for statistical analysis. Data are given as mean ± SD. A P value of <.05 was considered significant.
Results
We found considerable differences in packaging, applicator, and leaflet weight between the three CGM systems (Figure 1, Table 1). Packaging as well as applicator weight was similar for the FL2 and G7 sensors, while the FL3 was packed with the least cardboard packaging (FL2: 17.9 g; FL3: 9.4 g; G7: 17.1 g) and had the lightest applicator (FL2: 71.6 g; FL3: 44.3 g; G7: 71.4 g). Thus, a marked reduction in packaging (−8.5 g) and applicator (−27.3 g) weight was found for the FL3 system in comparison with its predecessor. Of note, both the applicator and the plastic packaging of the FL2 sensor itself were weighed together as they were considered as a unit. Thus, the reduction in plastic weight is caused by the re-design of the applicator which contains the sensor and does not require separate sensor packaging anymore. The G7 sensor included the heaviest leaflet (FL2: 3.9 g; FL3: 4.3 g; G7: 19.6 g).
Weight of Packaging, Applicator, and Leaflets Which are Distributed With Each Single Sensor.
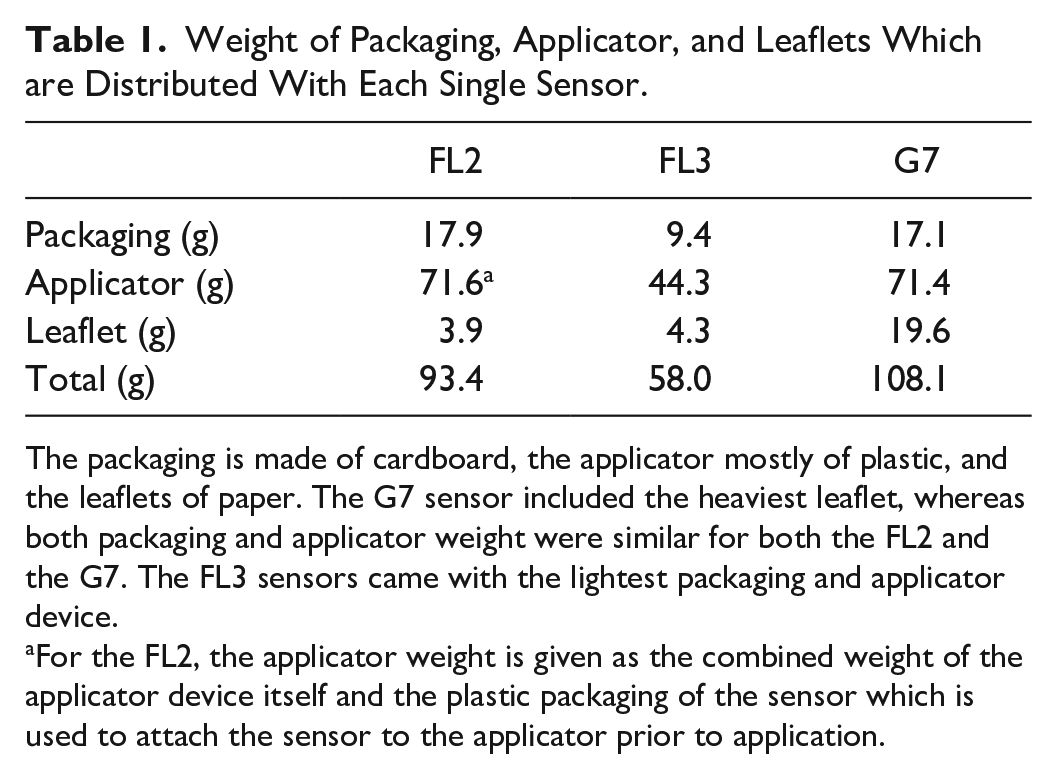
The packaging is made of cardboard, the applicator mostly of plastic, and the leaflets of paper. The G7 sensor included the heaviest leaflet, whereas both packaging and applicator weight were similar for both the FL2 and the G7. The FL3 sensors came with the lightest packaging and applicator device.
For the FL2, the applicator weight is given as the combined weight of the applicator device itself and the plastic packaging of the sensor which is used to attach the sensor to the applicator prior to application.
The total weight of each glucose sensor before disassembly was 3.3 g (FL2), 1.1 g (FL3), and 3.2 g (G7). The process of dismantling was tedious as the adhesives used made a precise separation of the components difficult if not impossible. This accounts for slight differences in the measured weight of both the case and the circuit board of a given glucose sensor, that is, small plastic parts were lost or stuck to another part in the process of dismantling. The G7 sensor was particularly difficult to disassemble into its main parts (Figure 2).
The plastic case contributed the most to the total weight of all three sensors (Figure 3, Table 2). The weight of the built-in circuit board and battery varied, especially when the FL3 was compared to its predecessor and the G7. The circuit board contributed 25% (0.75 ± 0.04 g) to the total weight of the FL2, 17% (0.18 ± 0.02 g) for the FL3’s, and 22% (0.71 ± 0.02 g) for the G7’s circuit board. The batteries of the FL sensors were significantly lighter than that of the G7 but contributed mostly to the total weight of the FL3 due to its slim and lightweight design (FL2: 12% (0.37 ± 0.01 g); FL3: 34% (0.36 ± 0.01 g); G7: 19% (0.64±0.01g)). The circuit board and case were significantly lighter in the FL3 when compared to the FL2 and the G7.

Weight of the glucose sensors’ case (a), circuit board (b), and battery (c). The mean weight of each component is given by a horizontal line. Varying measurements for the case and the circuit board resulted from the fact that the components could not always be separated exactly the same way when they were dismantled. In addition, some small pieces of plastic splintered off during the dismantling process. This concerned the G7 most markedly. P*** < .0002. **P < .005, *P < .05, n = 8-11, Kruskal-Wallis test.
Total Weight and Relative Weight Distribution of the Glucose Sensors’ Main Components Given as Mean ± SD (Percentage of Total Sensor Weight).

The respective percentage was calculated by the relation of the weight of the disassembled parts to the total weight of all three components’ weight and not to the measured weight of the intact sensors. Thus, it was accounted for the slight differences in weight due to lost plastic parts and incomplete separation of the respective parts due to the adhesives. The FL2 glucose sensor was the heaviest, its successor the most lightweight sensor analyzed. The case contributed mostly to the total weight of each sensor, whereas the proportion of the circuit board and battery varied between the glucose sensors.
Extrapolating these data by employing the number of CGM sensors disposed of yearly worldwide indicates the generation of approximately 3500 tons of cardboard packaging, 15 000 tons of plastic applicators, 2000 tons of paper leaflets, 340 tons of plastic casings, 130 tons of electronic circuit boards, and 110 tons of batteries, that is, 20 000 tons of indirectly sensor-related waste and 580 tons of direct sensor-associated waste in total, per year (Tables 3 and 4).
Estimated Amounts of Cardboard Packaging, Plastic Applicators, and Paper Leaflets Generated With Every Sensor of Three Worldwide Often Used CGM Systems.

Each part was measured and extrapolated using available data on the number of glucose sensors annually used worldwide.
Estimated Amounts of Plastic Cases, Electronic Waste, and Batteries Generated by Disposing of Three Worldwide Often Used Glucose Sensors in the Waste.

Each part was measured and extrapolated using available data on the number of glucose sensors annually used worldwide.
Discussion
The success of systems for CGM is driven by the benefits PwD experience in daily life experience by using them,8,9 especially when combined with an insulin pump and an algorithm as part of a system for automated insulin delivery (AID). 10 The downside is that millions of glucose sensors are disposed of—most often in general waste—worldwide annually. In addition, sensors are commonly distributed individually packed in cardboard together with single-use applicators and enclosed leaflets. Data presented here on the weight of key components of the glucose sensors indicate clear differences in the composition and weight distribution (driven to a good extent by the battery type used) of three widely used CGM systems. The reduction in weight from one generation of the glucose sensors of their CGM system to the next one (FL2 vs FL3) can also be interpreted as attempts by the manufacturer to reduce waste production. Similarly, the amount of packaging, leaflets, and applicators differed considerably between the three analyzed CGM systems.
Our study has limitations. The manual separation of the glucose sensors was difficult, imprecise, and prone to injury. The adhesives used to make the sensors tight complicate the disassembly process and, for the G7 sensor, render the complete separation nearly impossible, which can be considered also hamper attempts to recycle such glucose sensors. Despite these limitations, this study provides a first insight into the amount and type of waste generated by glucose sensors.
It is not publicly known how many glucose sensors are used worldwide per year by PwD and other users; however, estimates indicate that around 5.7 million PwD use an FL CGM system and 2.3 million a DC CGM system. 3 Since at least 26 glucose sensors are required if a PwD is using an FL CGM system all year long and 37 glucose sensors with the DC CGM system, this estimation sums up to 148 200 000 glucose sensors for the FL2/FL3 and 85 100 000 for G7 (233 300 000 sensors in total), not accounting for additional sensors required as a replacement for faulty, damaged, or lost sensors. These numbers were used to extrapolate the amount of waste generated by glucose sensors annually. For the sake of feasibility, the total number of FL glucose sensors was divided by two to take into account that FL2 and FL3 sensors were examined. According to respective calculations, the waste generated indirectly by packaging and applicators accounts for most of the waste worldwide per year associated with the usage of the three CGM systems examined for this study. Our extrapolation indicates approximately 3500 tons of cardboard packaging, 15 000 tons of plastic applicators, and 2000 tons of paper leaflets, that is, 20 000 tons in total, per year (Table 3). As demonstrated by the comparison between the FL2 and FL3 systems, reduction in packaging, leaflets, and applicators yields a huge potential for reducing waste. It is favorable to reduce packaging to a minimum, that is, omit printed manuals and other content, which is not required for using the sensor, although national law regulations must be considered. A lot of plastic is wasted when single-use sensor applicators are disposed of with each new sensor. Reusable devices would have a significant impact on the amount of plastic waste. As an example, the Medtronic applicator MMT-7510 of the iPro2 CGM system (off market) was reusable.
Our extrapolation suggests that around 580 tons of waste is generated by the sensors directly. This separates into approximately 340 tons of plastic casings, 130 tons of electronic circuit boards, and 110 tons of batteries (Table 4). Batteries are of special interest since batteries are usually discarded with a residual amount of power. It is estimated that in general, around 5% to 10% of a battery’s total energy remains after usage.11,12 The electronic board and the sensor itself contain valuable resources that are worth recycling.
Continuous glucose monitoring systems are mainly used in outpatients. Their role in inpatient care was studied by several authors.13-15 Their data indicate an improvement in glucose regulation, avoiding hypoglycemia, and more efficient treatment, which also saves health care professionals’ time. However, it can be assumed that due to single-use transmitters and a mostly shorter duration of the hospitalization than the sensor period, resource usage is not optimal in an inpatient setting.
Continuous glucose monitoring systems are a crucial pillar of modern diabetology. While they only contribute a small percentage to the total amount of waste generated worldwide, 16 they comprise a good percentage of that generated for diabetes therapy. Accordingly, there is an interest in how to reduce the footprint of therapy and conserve resources—especially since they are usually discarded in general waste and not properly disposed of. 5
In the European Union (EU) and Switzerland, manufacturers of medical products are subject to “extended producer responsibility” (EPR). For example, the manufacturers of batteries are obliged to ensure proper disposal and recycling of their products. The new EU Battery Regulation has been in force since February 18, 2024. It was published by the EU Commission in the Official Journal on August 17, 2023, and came into force in all member states with six months’ notice. As a component of the European Green Deal, it is intended to regulate and simplify the assessment and standardization and thus the legal framework of battery types and the associated obligations in the EU countries. In addition to registration and labeling obligations, this also includes the take-back of batteries and the promotion of environmentally friendly recycling (or, if applicable, participation in an approved battery-return system). For example, in Switzerland “Inobat” 17 acts as a battery collection point. Each municipality has its collection point where batteries and electronic devices including medical devices can be disposed of.
As another practical example, German law obliges manufacturers of electrical or electronic equipment to take responsibility for the entire life cycle of their products, that is, under EPR this means that a manufacturer is legally required to ensure the safe disposal of its equipment, including the collection, treatment, and recycling of old/broken products. This falls under the responsibility of the WEEE Directive (Directive on Waste Electrical and Electronic Equipment) 2012/19/EU, revised version 2024/884/EU. Manufacturers are obliged to register for a WEEE number.
A medical device manufacturer must register with the relevant national authorities in each country in which they distribute or sell devices, irrespective of health regulations, in accordance with the requirements of the aforementioned regulations.
The composition of the analyzed sensors, that is, the type of plastic in the components, is of further concern. Different materials and especially different types of plastic are more suitable or not suitable at all for recycling. We have no information regarding the plastic used which would determine the feasibility of recycling CGM systems. Another topic of interest is the equivalent of CO2 emissions generated by CGM therapy and the amount that could be saved by decreasing sensor size, increasing the usage period, or employing multiuse applicators, which are mainly made of plastic. In addition, production, delivery, and disposal would have to be taken into account. Perugini et al 18 thoroughly discuss the environmental impact of plastic in their review from 2005 and particularly compare the estimated emissions of different methods of plastic disposal and recycling. Accordingly, the CO2 equivalent in kg per kg plastic by the example of polyethylene terephthalate (PET), a widely used material, ranges from 1.4 (mechanical recycling) to 7.3 (combustion). Other studies report similar emission equivalents. 19 However, converting the amount of CGM plastic waste into CO2 emission equivalents is not feasible in a serious manner, as the exact knowledge of the materials used and the life cycle of the devices, that is, recycling, combustion, and so on, is not known to the public.
The glucose sensors used for the analysis presented in this manuscript were collected while performing a real-world study in a specialized diabetes practice in a mid-size town in Germany. The waste disposal behavior of PwD was analyzed, and the therapy-related waste was quantified. 5 Although glucose sensors made only up for a relatively small quantity of the total amount of disposed aids used for diabetes therapy, that is, needles and insulin pens, their composition is by far more complex, and they comprise valuable materials. Interestingly, most PwD that participated in this study were specifically asked to, dispose of the used glucose sensors in general waste and not in special collection sites, where at least, parts of the sensors can be separated/recycled. However, the PwD that participated in the study mentioned before clearly expressed their demand for more sustainability of the products used by them for diabetes therapy. Also, in other surveys performed recently in Germany among the PwD, this strong wish was expressed.
Conclusion
In conclusion, hundreds of tons of plastic waste and electronic waste/batteries as well as thousands of tons of packaging and applicators are disposed of annually in the form of CGM systems. Fully acknowledging the benefits that PwD has by using CGM, these data highlight huge potential to enhance the sustainability of diabetes management by offering good recycling options for the glucose sensors, reduction of packaging, and employing multiuse aids whenever possible.
Footnotes
Acknowledgements
The authors thank Prof. Dr. Christian Unsöld, TUN Consulting, for his expert advice, and Ms Birte Hussmann, Working Group Experimental Diabetology and Islet Cell Biology, for her assistance.
Abbreviations
CGM, continuous glucose monitoring; DC, Dexcom; EPR, extended producer responsibility; EU, European Union; FL, FreeStyle Libre; FL2, FreeStyle Libre 2; FL3, FreeStyle Libre 3; G7, Dexcom G7; PET, polyethylene terephthalate; PwD, people with diabetes; WEEE, waste electrical and electronic equipment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
